Clear Sky Science · en
Repurposing metformin for treating osteoarthritis via leveraging Nrf2 signaling
Why a diabetes pill might help aching joints
Osteoarthritis is a leading cause of pain and disability as we age, yet current treatments mostly mask symptoms rather than slow the disease. This study asks a provocative question: could metformin, a cheap and widely used drug for type 2 diabetes, be repurposed to protect worn-out joints by taming a form of cellular “rust” known as oxidative stress?
The problem of wear, tear, and cellular rust
Osteoarthritis is more than simple wear and tear. In painful joints, the smooth cartilage that cushions bones breaks down, the underlying bone remodels, and inflammation smolders in the surrounding tissue. The authors focus on oxidative stress—an imbalance between reactive oxygen species (ROS), which damage cells, and the body’s antioxidant defenses. Excess ROS in joint cartilage can trigger inflammatory molecules, speed up cartilage breakdown, and push cartilage cells toward death or premature aging. Because many of these processes are driven by chemistry shared across tissues, a drug that calms oxidative stress elsewhere in the body might also help diseased joints.
An old drug with new possibilities
Metformin has been prescribed to millions of people with type 2 diabetes for more than 60 years. Beyond lowering blood sugar, it has been linked to anti-inflammatory, anti-aging, and antioxidant effects. The researchers began with a systematic review of past work, which strengthened the idea that oxidative stress is central to osteoarthritis and that metformin can counter oxidative damage in several chronic diseases. They then set out to test, in animals and cells, whether metformin could relieve osteoarthritis and through which biological switches it might act.
Testing metformin in arthritic mice
In one set of experiments, the team induced osteoarthritis in the knee joints of adult mice by injecting a chemical that injures cartilage. Some mice received daily metformin by mouth for eight weeks. Compared with untreated animals, metformin-treated mice showed reduced sensitivity to pain in behavioral tests and tended to move more steadily across a narrow beam. Imaging of the knee by micro–CT scans and detailed tissue staining revealed that metformin modestly slowed cartilage erosion and helped preserve key structural molecules, while limiting enzymes that normally chew up cartilage. Blood tests showed lower levels of a damage marker linked to oxidative stress and higher activity of one of the body’s natural antioxidant enzymes.
Zooming in on cartilage cells and a key defense switch
To understand how metformin works at the cellular level, the authors turned to cultured cartilage cells exposed to hydrogen peroxide, a source of ROS that mimics oxidative stress. Under this assault, cells shrank, survival dropped, inflammatory genes and cartilage-degrading enzymes surged, and markers of programmed cell death rose. Adding metformin protected the cells: they survived better, looked healthier, made fewer inflammatory and destructive molecules, and generated fewer ROS. The researchers traced this protection to a molecular switch called Nrf2, a master regulator of antioxidant defenses. Metformin promoted Nrf2 movement into the cell nucleus and boosted downstream proteins such as HO-1 that help neutralize ROS. When Nrf2 was deliberately reduced in cells, metformin’s benefits largely faded, suggesting this pathway is crucial for its protective action.
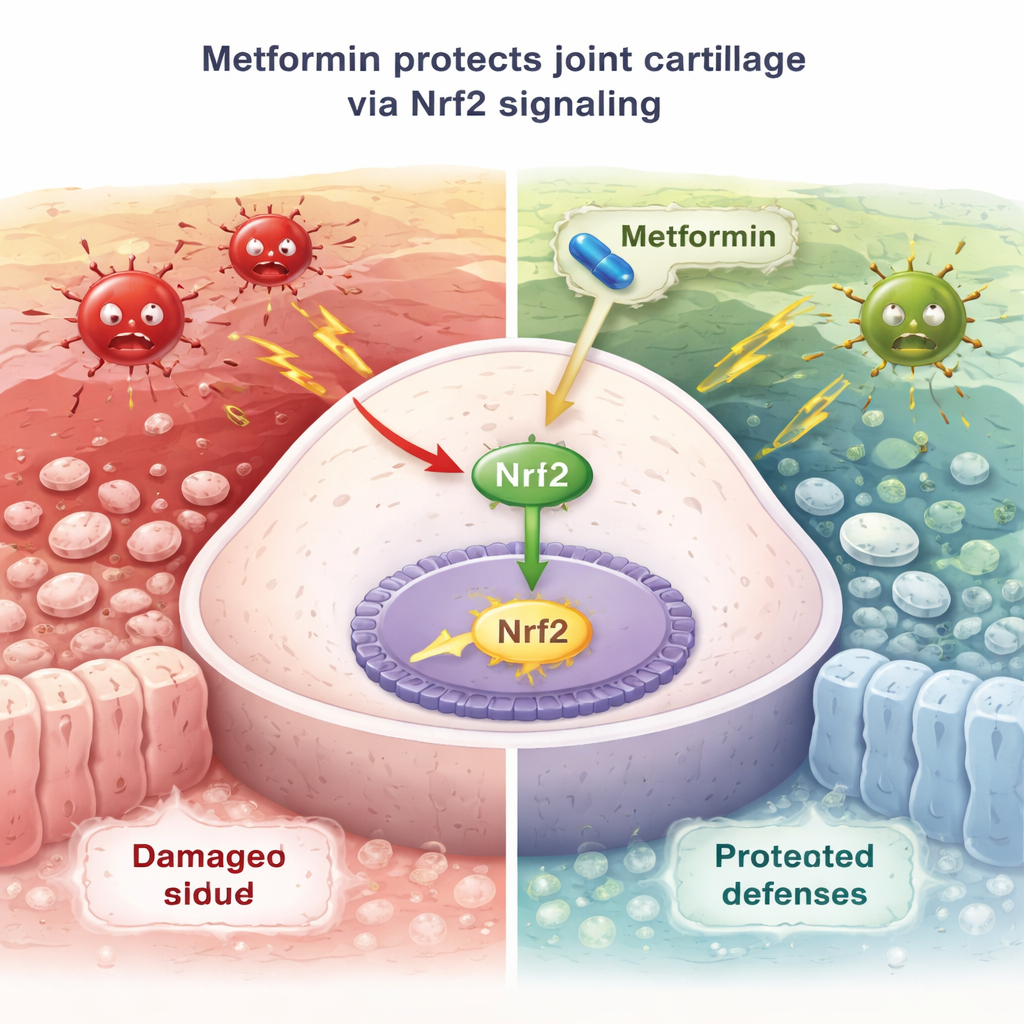
What happens when the key switch is missing
The team then repeated the osteoarthritis experiments in genetically engineered mice that lack Nrf2 altogether. In these animals, metformin no longer eased joint pain, did not meaningfully protect cartilage structure, and failed to improve oxidative stress markers in the blood, in stark contrast to its effects in normal mice. This underscored that Nrf2 is a central hub through which metformin shields joints from oxidative damage. Finally, the authors reviewed existing clinical studies in people with type 2 diabetes. Several retrospective cohort studies reported that patients taking metformin were less likely to undergo knee or hip replacement surgery—an indirect sign that their joint disease may have progressed more slowly.
What this could mean for people with sore knees
Taken together, the work suggests that metformin can reduce pain and slow some of the structural damage in osteoarthritis, at least in preclinical models, largely by turning on the Nrf2 antioxidant defense pathway in cartilage cells. For patients, this does not yet mean metformin should be used routinely for joint disease; the evidence in humans is still indirect, and the drug has its own risks and contraindications. However, because metformin is inexpensive, well studied, and already widely prescribed, these findings provide a strong rationale for rigorously designed clinical trials to test whether this familiar diabetes pill could one day become part of a disease-modifying strategy for osteoarthritis.
Citation: Zhong, Y., Chen, J., Shi, N. et al. Repurposing metformin for treating osteoarthritis via leveraging Nrf2 signaling. Sci Rep 16, 4966 (2026). https://doi.org/10.1038/s41598-026-35708-x
Keywords: osteoarthritis, metformin, oxidative stress, Nrf2 pathway, cartilage protection