Clear Sky Science · en
HIV Vpr induces demethylation of the SNCA antisense promoter, leading to neurocognitive impairment
Why HIV Can Still Affect the Brain
People living with HIV are living longer, healthier lives thanks to modern antiretroviral drugs. Yet many still experience problems with memory, concentration, and movement, even when the virus in their blood is well controlled. This study explores why that happens by zooming in on a small viral protein, called Vpr, and a brain protein, alpha‑synuclein, that is also central to Parkinson’s disease. Understanding how these two molecules interact may reveal why HIV can accelerate brain aging and suggest new ways to protect the brain.
A Brain Protein at the Crossroads of HIV and Parkinson’s
Alpha‑synuclein is a protein that helps nerve cells communicate, especially in brain regions that control movement and memory. When too much alpha‑synuclein builds up, it clumps into aggregates that damage synapses, strain energy‑producing mitochondria, and fuel inflammation. Such clumps are a hallmark of Parkinson’s disease. The authors show that alpha‑synuclein also builds up with normal aging in mice, and that HIV’s Vpr protein pushes its levels even higher in nerve‑like cells. This places alpha‑synuclein at the crossroads between HIV‑related thinking problems and classic movement disorders.
How a Viral Protein Rewrites Cellular “Punctuation”
Every cell uses chemical tags on DNA—often described as molecular punctuation marks—to turn genes on or off. In this work, the team focused on a little‑known control switch inside the alpha‑synuclein gene called the antisense promoter. In healthy cells, this switch is heavily tagged with methyl groups, keeping it relatively quiet. The researchers found that Vpr strips away these tags at specific sites in this region, a process known as demethylation. Once those marks are removed, the antisense promoter becomes more active and drives extra production of alpha‑synuclein, setting the stage for harmful aggregation.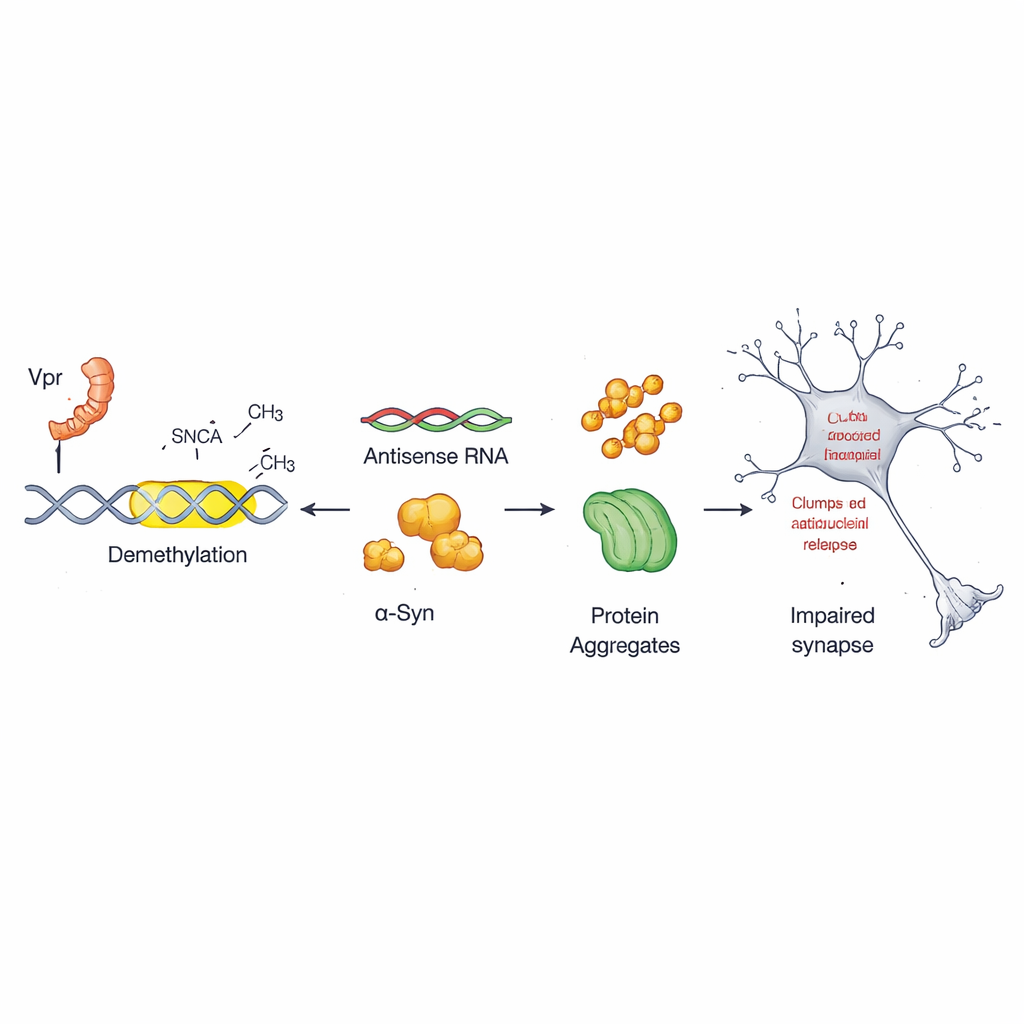
Evidence from Cells, Mice, and Human Brains
Using cultured human nerve‑like cells and primary mouse neurons, the authors showed that adding Vpr boosts alpha‑synuclein message levels and protein levels over time. A drug that broadly promotes DNA demethylation mimicked some of these effects, underscoring the role of epigenetic control. In contrast, a compound called DMOG, which blocks demethylating enzymes, prevented Vpr from fully activating the antisense promoter. The team then turned to human brain samples from people with and without HIV. Brains from HIV‑positive donors—both before and after antiretroviral treatment—had fewer methyl tags at the same DNA region and higher levels of alpha‑synuclein and its antisense transcript, especially in donors with HIV‑related dementia. This suggests that the virus leaves a durable epigenetic scar in the brain.
From Molecular Changes to Memory Problems
To connect these molecular shifts to behavior, the researchers examined how Vpr affects brain circuits in mice. When they applied Vpr to slices of mouse hippocampus, a region vital for memory, basic signal strength between neurons was unchanged, but the ability to strengthen connections—a process called long‑term potentiation—was reduced. In living mice, targeted injections of Vpr into the hippocampus led to poorer performance on a spatial memory task in which animals must remember the location of objects. Together, these experiments suggest that Vpr‑driven alpha‑synuclein changes are not just biochemical curiosities; they translate into weakened synapses and measurable memory deficits.
What This Means for People Living with HIV
This study proposes a clear chain of events: HIV releases Vpr, Vpr reprograms a key DNA switch controlling alpha‑synuclein, the protein accumulates and clumps, and neurons gradually lose their ability to communicate and support memory. Because similar alpha‑synuclein problems underlie Parkinson’s disease, the work hints that HIV and classic neurodegenerative diseases share overlapping mechanisms. Importantly, the findings highlight possible new strategies—such as drugs that stabilize DNA methylation at the antisense promoter or limit alpha‑synuclein buildup—to slow or prevent HIV‑related cognitive decline and movement problems.
Citation: Santerre, M., Wang, Y., Kalamarides, D. et al. HIV Vpr induces demethylation of the SNCA antisense promoter, leading to neurocognitive impairment. Sci Rep 16, 6078 (2026). https://doi.org/10.1038/s41598-026-35691-3
Keywords: HIV-associated neurocognitive disorders, alpha-synuclein, epigenetics, DNA methylation, Parkinson-like symptoms