Clear Sky Science · en
Proteomic atlas of human peritoneal tissue
Why the Lining of Your Abdomen Matters
The inside of the abdomen is wrapped in a delicate, glistening sheet called the peritoneum. When cancers spread there, the result—peritoneal carcinomatosis—is often painful, hard to treat, and ultimately deadly. Yet while doctors have learned a lot about tumors and cancer cells, the peritoneum itself has remained a kind of “black box.” This study opens that box by building a detailed catalog of the proteins that make up healthy human peritoneal tissue, offering clues to why some cancers find this surface such a welcoming place to grow.
Mapping the Hidden Landscape
The researchers set out to answer a basic but long-ignored question: what is the normal molecular makeup of the peritoneal lining? To do this, they collected tiny samples of non-cancerous peritoneal tissue from patients undergoing abdominal surgery for other reasons. Some samples were fresh frozen, while most came from standard pathology blocks preserved in wax—a common way hospitals store tissue. Using a highly sensitive technique called mass spectrometry, the team broke these tissues down into their component proteins and identified more than a thousand distinct proteins that together form the fabric of the peritoneal surface.
The Peritoneum: Simple but Strong
Compared with organs like the ovary or kidney, the peritoneum turned out to be relatively modest in its protein diversity. Each sample yielded far fewer unique proteins than typical visceral organs, which is consistent with the peritoneum’s simpler structure: a thin layer of specialized surface cells lying on top of supportive connective tissue. However, what the peritoneum lacks in variety, it makes up for in structure. Many of the most abundant proteins were structural “scaffolding” molecules, including several types of collagen and other components of the extracellular matrix—the protein-rich mesh that surrounds and supports cells. These sturdy fibers help the peritoneum withstand constant motion and friction inside the abdomen, but they may also create a tough barrier that can shield tumors from immune attack.
Proteins That Welcome or Fight Tumors
Beyond basic architecture, the atlas revealed proteins with known roles in cancer spread. The team found molecules that help cancer cells stick, invade surrounding tissue, and survive in new environments. Examples include fibronectin, laminin, periostin, tenascin, and heparan sulfate proteoglycans, all of which have been linked to tumor growth, blood vessel formation, and resistance to treatment. They also detected proteins involved in immune defense and inflammation, such as markers of neutrophils—a type of white blood cell—supporting the idea that these cells and their granular secretions may influence how tumors behave in the peritoneal cavity. In addition, the researchers identified several known cancer-promoting proteins (oncoproteins), tumor suppressors, and potential biomarkers that could be tracked as disease develops.
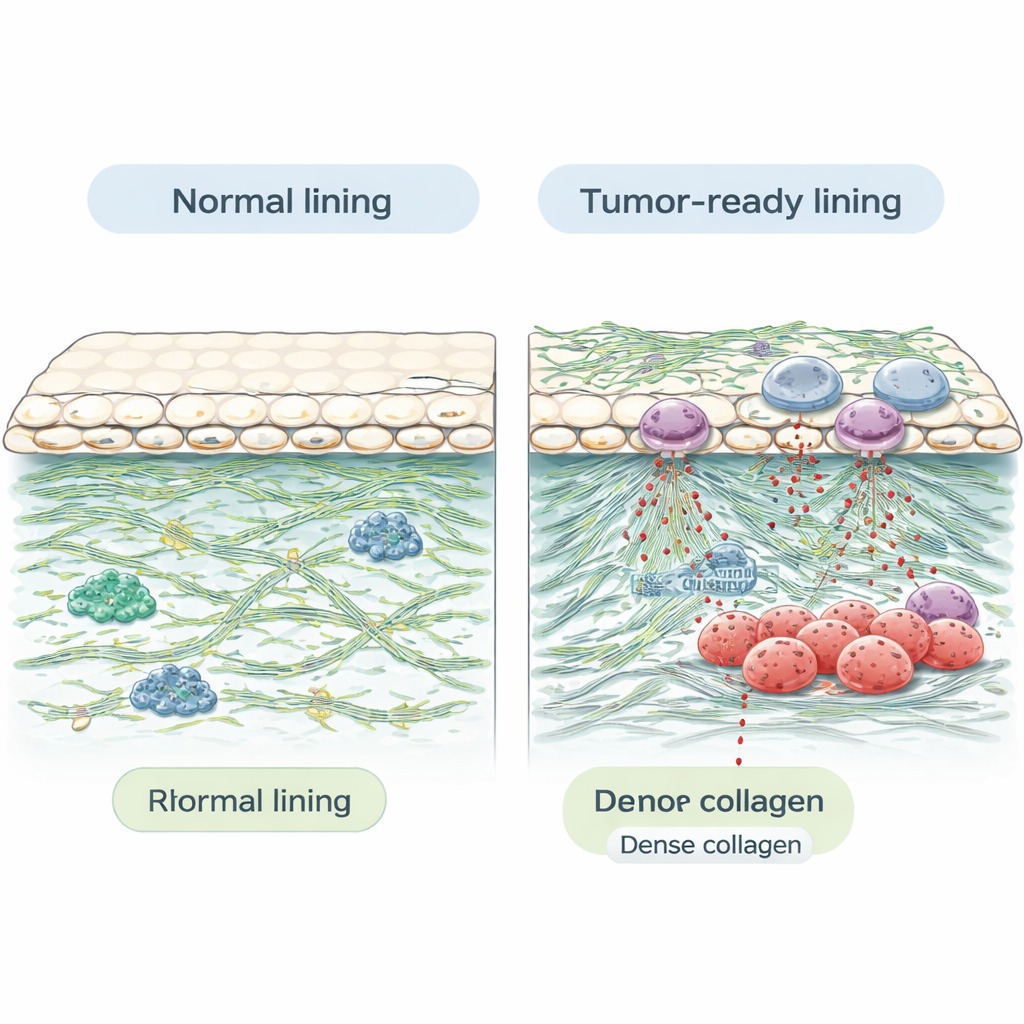
Clues for Rare Peritoneal Cancers
While most peritoneal tumors originate from elsewhere in the body, some cancers arise directly from the peritoneum, such as malignant mesothelioma and primary peritoneal carcinoma. These are rare and poorly understood diseases. By showing that key cancer-related proteins and pathways are already present in healthy peritoneal tissue, this atlas provides a starting point for studying how those molecules change as tumors form. Because the methods work well on routine, archived pathology samples, future researchers can look back at stored tissues to ask which protein changes predict who will develop peritoneal disease, and who might respond best to particular treatments.
What This Means for Patients
For people living with or at risk for abdominal cancers, this work does not yet translate into a new drug or test—but it lays crucial groundwork. By defining the “baseline” protein environment of the peritoneum, the study helps scientists see which elements of this inner lining may nourish cancer “seeds” and which might be harnessed to fight them. In the years ahead, comparing this healthy atlas with diseased peritoneal tissue could uncover new biomarkers for earlier detection, better ways to monitor response to therapy, and ultimately, strategies to change the peritoneal lining from fertile soil for tumors into a less hospitable landscape.
Citation: Zhang, Q., Sherry, C., Peng, X. et al. Proteomic atlas of human peritoneal tissue. Sci Rep 16, 7378 (2026). https://doi.org/10.1038/s41598-026-35687-z
Keywords: peritoneal carcinomatosis, tumor microenvironment, proteomics, extracellular matrix, peritoneal metastasis