Clear Sky Science · en
Investigation of Acoramidis-based ionic liquids as potential stabilizers of transthyretin using DFT calculations, molecular docking, and ADMET studies
Why a pill for a rare heart disease is so hard to make
Transthyretin amyloidosis (ATTR) is a serious disease in which a blood protein piles up in the heart and nerves, slowly damaging them. A new drug, Acoramidis, can help stop this buildup, but there is a practical problem: the drug does not dissolve well in water, which makes it hard for the body to absorb when taken as a pill. This study explores a clever way to redesign Acoramidis into new salt-like forms, called ionic liquids, that could make the medicine easier to swallow and more effective.
Turning a promising drug into a drinkable form
For a pill to work, it must first dissolve in the watery fluids of the stomach and intestines, then pass through the gut wall into the bloodstream. Acoramidis is powerful at stabilizing the transthyretin (TTR) protein, which helps prevent harmful protein clumps linked to ATTR, but it is poorly soluble and has weak oral bioavailability. The researchers set out to tackle this by chemically pairing Acoramidis with different charged partner molecules, creating three new ionic liquid versions (IL1, IL2, and IL3). These versions are designed to behave more like flowing salts than rigid crystals, potentially boosting how well the drug dissolves and spreads through the body. 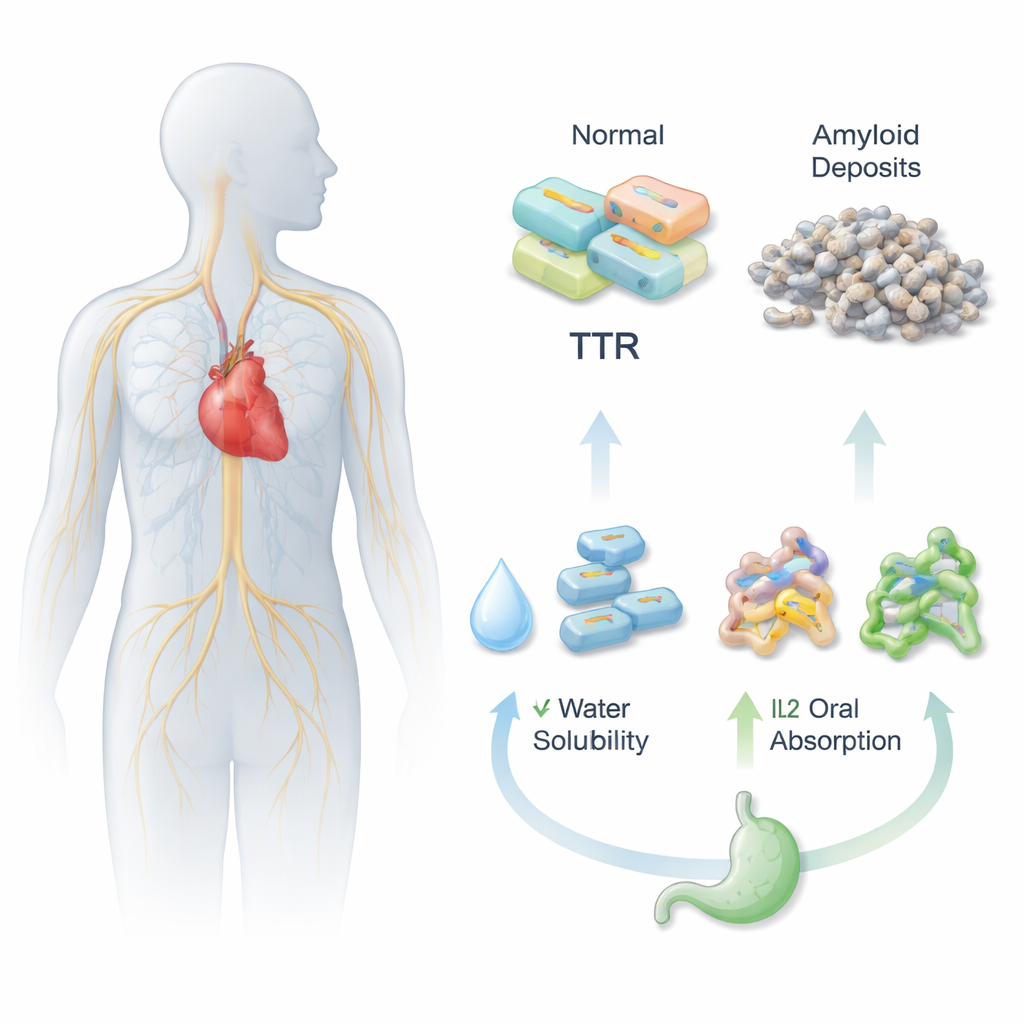
Probing the new molecules on a computer
Instead of going straight to animal or human tests, the team used advanced computer methods to predict how these new forms would behave. Quantum chemistry calculations estimated how polar each molecule is—a key predictor of water solubility—and how easily its electrons shift, which relates to how it might interact with proteins. IL1 stood out with the highest dipole moment and strongest electronic responsiveness, indicating it should dissolve better in water and form stronger, more flexible contacts with its targets. The energy calculations also suggested that IL1 is more thermally stable than the original drug and the other two ionic liquids, meaning it should be less likely to fall apart before reaching its destination.
How tightly the drug can grip its target
Next, the researchers used molecular docking, a virtual “lock-and-key” test, to see how well Acoramidis and its three ionic forms fit into the binding channel of the TTR protein. All four compounds docked into the correct region of TTR, but IL1 formed the tightest grip, with the most favorable binding energy and several stabilizing contacts—particularly hydrogen bonds—with key amino acids in the protein. This stronger binding suggests IL1 may be even better than the parent drug at holding TTR in its safe, four-part structure, helping to prevent the dangerous breakdown and misfolding that leads to amyloid deposits.
Will the body absorb and clear these new forms?
Finally, the team used online pharmacology tools to probe how the body might handle these compounds—how well they dissolve, cross the gut wall, circulate in blood, and are eventually cleared. All three ionic liquids were predicted to be more soluble in water than Acoramidis, with IL3 showing the highest raw solubility. Measures of intestinal permeability and overall oral absorption improved for all three ionic liquids, with IL1 and IL2 giving the best balance of gut penetration and predicted bioavailability. Protein binding in blood, clearance rates, and half-lives also shifted in ways that suggest the ionic liquid forms could offer more reliable exposure after oral dosing, with IL1 again offering a particularly favorable profile. 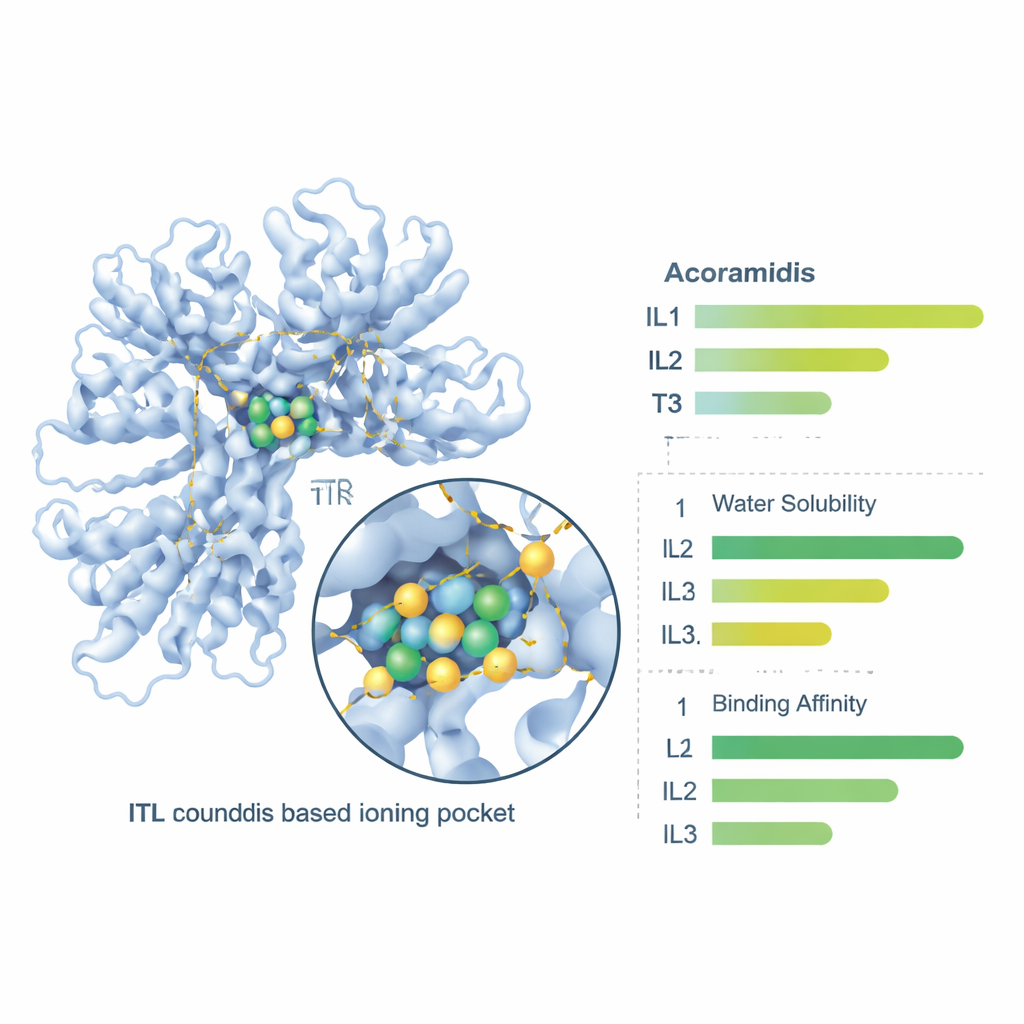
What this could mean for future ATTR treatments
To a non-specialist, the main message is that the drug Acoramidis already looks promising for a rare but serious heart and nerve disease, yet its poor solubility limits how well it can work by mouth. By turning Acoramidis into specially designed ionic liquids, this study shows—entirely through computer-based tests—that it may be possible to create versions that dissolve better, bind more strongly to their protein target, and are absorbed more efficiently. Among the three candidates, IL1 appears to strike the best balance of stability, binding power, solubility, and predicted oral uptake, making it a strong lead for future laboratory and clinical development aimed at delivering more effective, easy-to-take treatments for ATTR.
Citation: Mostaghni, F., Mahani, N.M. Investigation of Acoramidis-based ionic liquids as potential stabilizers of transthyretin using DFT calculations, molecular docking, and ADMET studies. Sci Rep 16, 6540 (2026). https://doi.org/10.1038/s41598-026-35684-2
Keywords: transthyretin amyloidosis, Acoramidis, ionic liquids, protein misfolding, oral drug delivery