Clear Sky Science · en
Sarilumab in the management of Graves orbitopathy with low clinical activity scores
Why this eye condition matters
Thyroid problems are common, and for many people they do not stop at the neck. A condition called Graves orbitopathy, or thyroid eye disease, can make the eyes red, bulging, and uncomfortable, and can even cause double vision. Even when doctors consider the eye disease “mild,” patients may feel disfigured, anxious, and limited in daily life. This study explores whether a modern anti‑inflammatory drug called sarilumab, already used for arthritis, can safely relieve eye symptoms and improve quality of life in people whose disease is usually just watched and not actively treated.
A closer look at thyroid eye disease
Graves orbitopathy happens when the body’s immune system mistakenly attacks the tissues around the eyes. Muscles and fat behind the eyeball swell and stiffen, pushing the eyes forward and pulling the eyelids back. Doctors often score how “active” the inflammation is with a Clinical Activity Score (CAS) from 0 to 10. Current European guidelines generally advise no strong medical treatment for people with low scores (2 or below), assuming their disease will settle on its own. The authors of this study argue that even low‑grade inflammation can seriously disturb vision, appearance, and confidence, and that these patients deserve more than simple observation.
The new drug on trial
Scientists know that a chemical messenger of the immune system called interleukin‑6 (IL‑6) is raised in active thyroid eye disease. Sarilumab is a laboratory‑made antibody that blocks the IL‑6 receptor, dampening this signal. The research team in Spain reviewed the records of 62 patients with Graves orbitopathy whose CAS was between 1 and 3 out of 10 but who reported a clearly impaired quality of life. All were treated at a single eye‑orbit center between 2019 and 2024. Depending on body weight, patients injected themselves with either 150 mg or 200 mg of sarilumab under the skin every 20 days, usually for about three to four sessions. No other eye‑specific immune drugs were given at the same time.
What happened to their eyes and blood tests
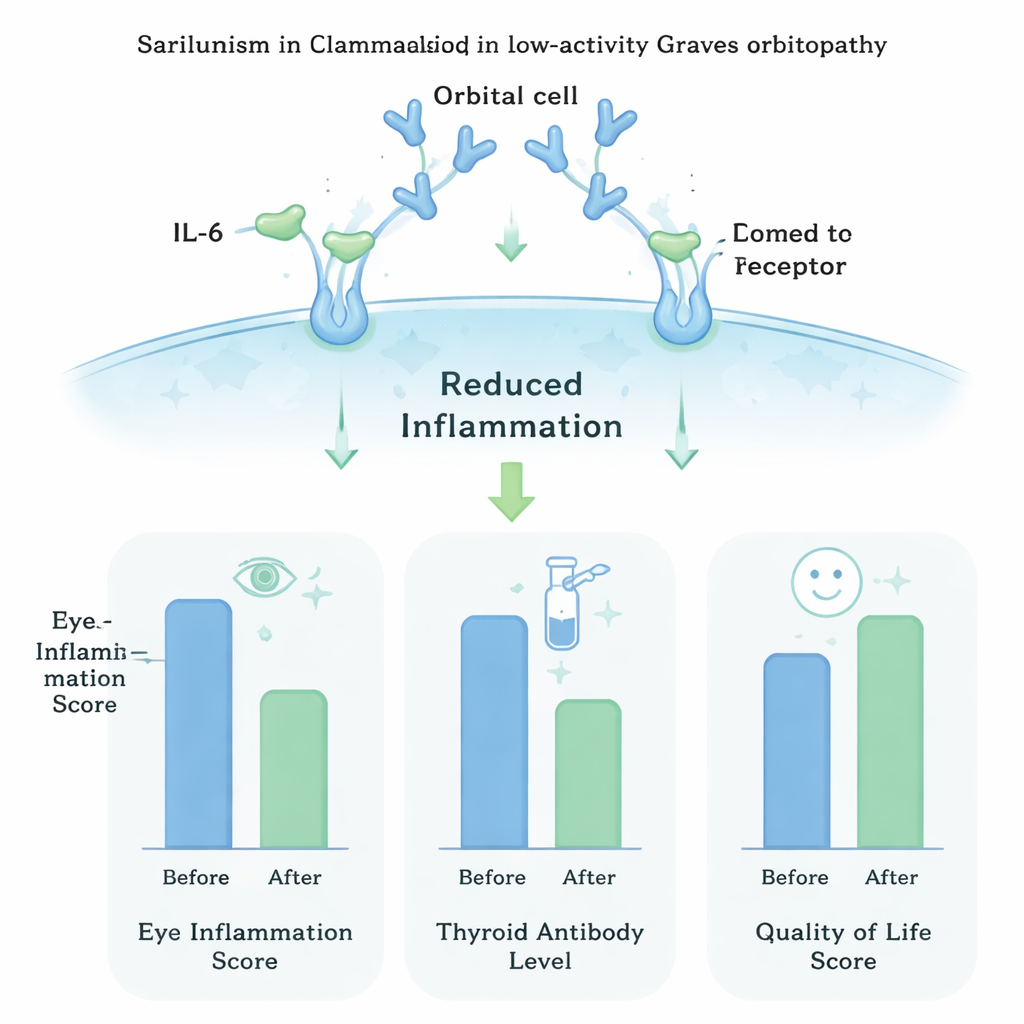
Before treatment, the average activity score was a little above 2, and blood tests showed high levels of thyroid‑stimulating immunoglobulins (TSI), antibodies that drive both thyroid overactivity and eye disease. After sarilumab treatment, the average CAS dropped almost to zero: 95% of patients had no measurable eye inflammation, and the rest had only minimal signs. TSI levels also fell sharply, and nearly two‑thirds of patients ended up with values in the normal range. Physical signs such as bulging of the eyes, eyelid redness and swelling, and eyelid retraction all eased. Fewer people had double vision, and imaging often showed less thickening of the eye muscles.
How patients felt about their daily lives
To capture the personal impact of the disease, the team used a questionnaire designed specifically for thyroid eye problems, called GO‑QOL. It asks about two main areas: how well people can function visually in everyday tasks, and how they feel about their appearance. At the start, the overall score averaged about 65 out of 100, reflecting noticeable limitations and emotional strain, especially about looks. After treatment, the average score rose to around 96, indicating that most patients felt almost fully restored in both day‑to‑day functioning and self‑image. Only three patients had a flare‑up of inflammation months after stopping sarilumab, and extra doses brought the eye activity back down.
Safety and what it means going forward
No life‑threatening side effects were seen, and no one had to stop sarilumab permanently, but many patients developed temporary drops in certain blood cells, mild changes in liver tests, or higher cholesterol; some required short delays between injections while their blood work normalized. Overall, the authors conclude that sarilumab appears to be an effective and reasonably safe option for people with low‑score thyroid eye disease whose lives are strongly affected. For lay readers, the key message is that even “mild” eye disease can be very serious for the person living with it, and that targeted immune treatments like sarilumab may prevent worsening and restore comfort and confidence—though larger, controlled trials are still needed before this approach becomes routine.
Citation: Pérez-Moreiras, J., Abelenda, D., Providência, J. et al. Sarilumab in the management of Graves orbitopathy with low clinical activity scores. Sci Rep 16, 5225 (2026). https://doi.org/10.1038/s41598-026-35682-4
Keywords: thyroid eye disease, Graves orbitopathy, sarilumab, interleukin-6, quality of life