Clear Sky Science · en
Enhancement of electroless copper coatings by triazole dithiocarbamate and green additives
Shinier Copper for Everyday Technology
Copper sits at the heart of modern life, carrying signals in our phones, powering circuit boards in cars and planes, and protecting parts from wear and corrosion. But the chemical baths used to coat surfaces with copper can be rough—both on the metal and on the environment. This study shows how a set of “green” ingredients, including a plant-derived sugar and a biopolymer from shellfish waste, can produce smoother, more durable copper films while cutting down on harsh chemicals.

From Electric Plating to Self-Driven Coatings
Most people imagine metal coating as something that requires wires and electricity. Electroless plating works differently: once the surface is prepared, copper atoms build up on their own through a chemical reaction, no power supply needed. This makes it ideal for coating complex shapes and tiny features on circuit boards. However, traditional electroless copper baths often rely on toxic ingredients and can leave behind rough, uneven layers that corrode too easily. The authors set out to redesign this process using more environmentally friendly components without sacrificing performance.
A Sugar Alcohol Sets the Stage
At the heart of the new bath is xylitol, a sugar alcohol better known as a low-calorie sweetener. Here, it acts as a “complexing agent,” gently holding copper ions in solution so they are released in a controlled way. Glyoxylic acid, a small organic molecule, serves as the reducing agent that turns dissolved copper ions into solid metal. Potassium hydroxide keeps the bath strongly alkaline, a condition needed for the reaction to proceed. Working at a modest 45 °C, this base recipe already deposits copper, but on its own it produces relatively thick, fast-growing coatings with a rough landscape of peaks and valleys.
Fine-Tuning with Smart Additives
To tame and refine the growing copper layer, the team added four ingredients step by step, creating five different baths. First came 1,2,4-triazole, a small ring-shaped molecule that stabilizes the bath and slows down uncontrolled growth. Next was methanesulfonic acid, a cleaner, less hazardous acid that improves the availability of copper in solution and nudges the growing film toward a more orderly crystal arrangement. A specialized compound called triazole dithiocarbamate further reshaped the way copper atoms packed together, boosting the coating’s electrochemical stability. Finally, the researchers introduced chitosan, a biodegradable polymer derived from crustacean shells, which acted like a microscopic polisher and “brightener,” leveling out the surface and giving the copper a shiny, mirrorlike finish.
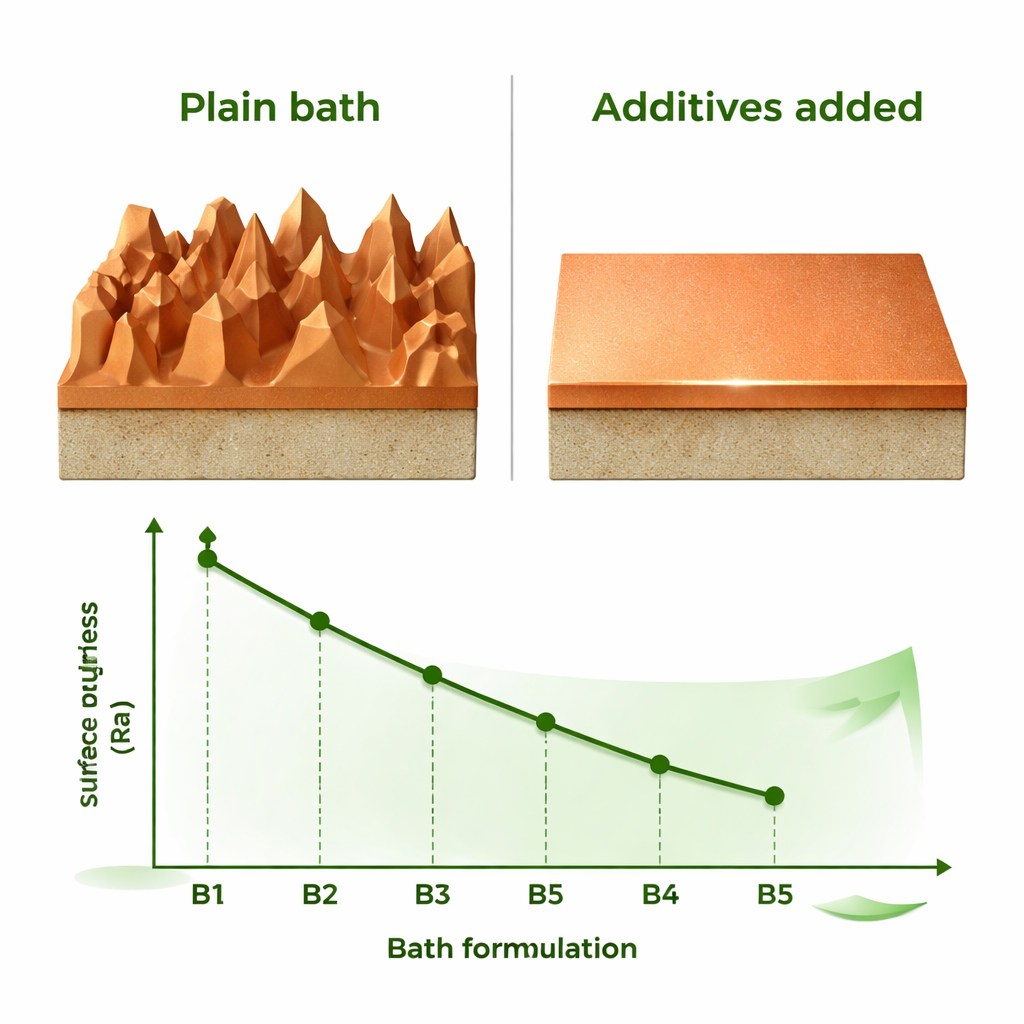
Measuring Smoothness, Structure, and Protection
The team carefully weighed and measured each coated sample to track how quickly copper was being deposited and how thick the films became. As more additives were added, the deposition rate dropped from about 3.46 to 2.68 micrometers per hour, and the layer thickness declined as well. This slower, more controlled growth proved beneficial. Atomic force microscopy, which scans the surface with a tiny probe, showed that the average roughness fell dramatically—from about 156 nanometers in the plain bath to just 19 nanometers in the fully modified “brightener” bath, a level of smoothness prized in high-performance electronics.
Inside the Copper and Against Corrosion
X-ray diffraction measurements revealed how the copper crystals were oriented and how large they were. As additives were introduced, the crystallite size shrank slightly and the arrangement of crystal planes shifted, changes that increase the effective surface area and support more uniform growth. Electrochemical tests, including cyclic voltammetry and Tafel polarization, probed how the coatings behaved in corrosive conditions. The optimized baths showed lower corrosion current densities—an indicator that the films resist attack more effectively—while the electrical response suggested a better balance between slowing down harmful reactions and supporting controlled copper deposition.
What It Means for Greener Electronics
By combining a sugar alcohol, a mild acid, advanced organic molecules, and a biopolymer, this work demonstrates a practical way to produce smoother, more corrosion-resistant copper coatings without relying on many of the harsh chemicals traditionally used in plating. For manufacturers of circuit boards, sensors, and precision components, such baths could mean longer-lasting products and cleaner production lines. For the rest of us, it shows how thoughtful chemistry can quietly improve the reliability of the electronic devices and infrastructure we depend on every day, while taking a gentler approach to the environment.
Citation: Balaramesh, P., Venkatesan, R., Jayalakshmi, S. et al. Enhancement of electroless copper coatings by triazole dithiocarbamate and green additives. Sci Rep 16, 6074 (2026). https://doi.org/10.1038/s41598-026-35680-6
Keywords: electroless copper, green additives, xylitol, corrosion resistance, chitosan