Clear Sky Science · en
Theoretical insights of 2D carbon nitride (C3N) as a highly selective sensor for volatile analytes
Why watching invisible gases matters
Many of the most dangerous air pollutants are gases we can’t see or smell until it is too late. Some are used in industry, some appear in warfare, and others come from everyday combustion. This paper explores how an ultrathin sheet of carbon and nitrogen, known as a C3N monolayer, could act as a tiny, highly selective electronic “nose” to detect several of these volatile and toxic molecules quickly and safely.
A flat sheet built for sensing
C3N is a two-dimensional material: a single-atom-thick layer of carbon and nitrogen arranged in a honeycomb pattern, similar to graphene. Because it is so thin, almost all of its atoms are exposed at the surface, giving incoming gas molecules plenty of room to land. The nitrogen atoms enrich the surface with electrons and tune its electrical behavior, turning C3N into a semiconductor rather than a simple conductor. That combination of high surface area, reactive nitrogen sites, and a useful electrical band gap makes C3N an appealing candidate for gas sensors that convert a chemical encounter directly into an electronic signal.
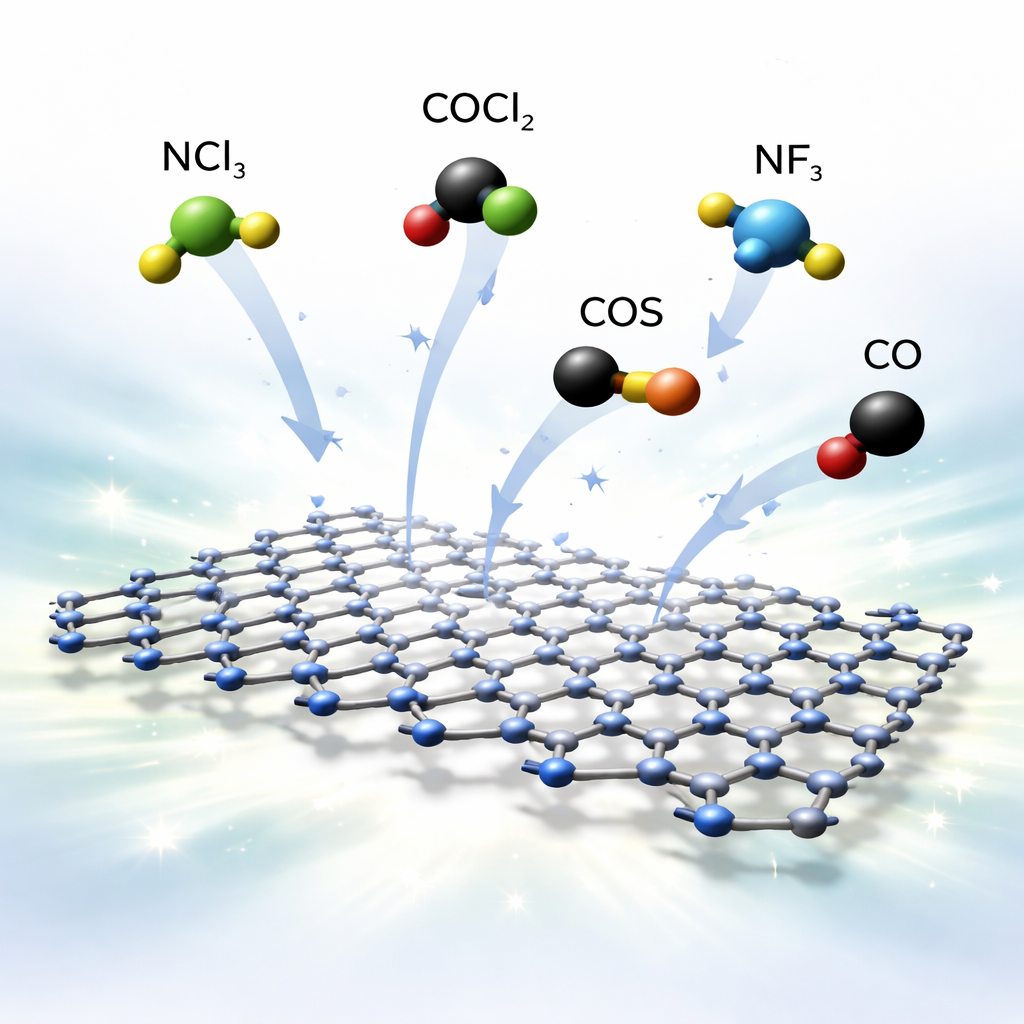
Targeting a family of toxic gases
The authors focus on five hazardous gases: nitrogen trichloride (NCl3), phosgene (COCl2), nitrogen trifluoride (NF3), carbonyl sulfide (COS), and carbon monoxide (CO). Each has a troubling safety profile, from corrosive lung damage to potent greenhouse effects and interference with oxygen transport in blood. Today, detecting such gases typically requires bulky, expensive instruments or complex sample preparation. A practical surface-based sensor has to strike a delicate balance: it must interact strongly enough with the gas to be detectable, but not so strongly that the gas sticks permanently and ruins the sensor. Theory suggests that C3N might offer just this compromise for these pollutants.
How theory tests a tiny sensor
Instead of building the sensor in the lab, the researchers use high-level quantum chemistry calculations to simulate how each gas approaches and binds to the C3N sheet. They map out the preferred landing spots, the distances between atoms, and the energies involved when a molecule rests on the surface. Multiple complementary tools probe what holds each gas in place: overall interaction energies, the way electron density shifts, and fine-grained analyses of non‑covalent forces such as van der Waals attraction and “halogen bonding” between chlorine or fluorine atoms and nitrogen sites on the sheet. They also compute how long each gas would tend to remain attached at realistic temperatures, a key factor for whether the sensor can reset itself between measurements.
Weak grip, strong signal
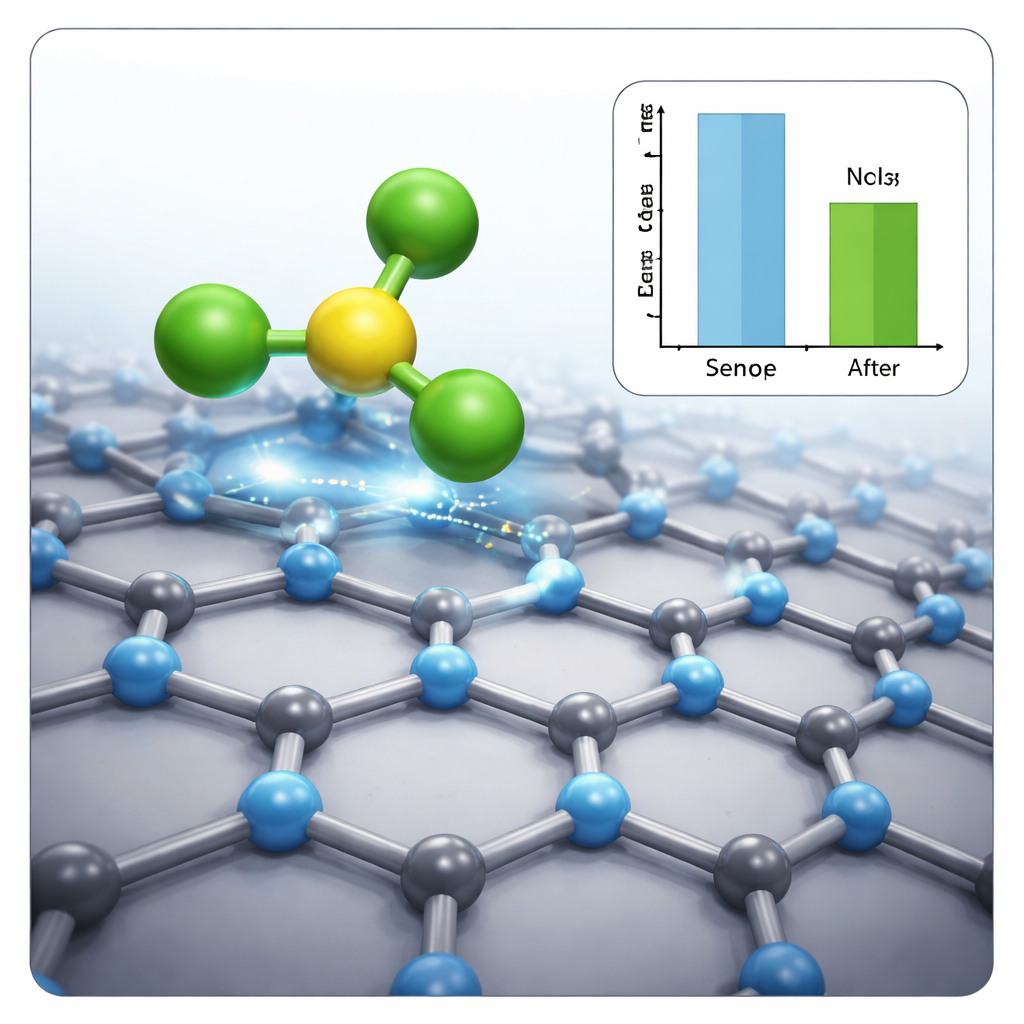
The simulations show that all five gases attach to C3N through relatively weak physical adsorption, with interaction energies well below the level associated with true chemical bonding. This means the gases can be captured and then released again, supporting fast sensor recovery rather than permanent fouling. Yet the electronic response is far from weak. For nitrogen trichloride in particular, the energy gap that governs how easily electrons move in the C3N layer shrinks markedly when the gas is present. Charge flows from the surface toward the NCl3 molecule via halogen bonds, producing a clear change in conductivity. Other gases, such as CO and COS, interact mainly through gentler dispersion forces and cause smaller shifts in the electronic structure, implying that C3N would be especially sensitive and selective toward NCl3 compared with the rest of the group.
From theory to future devices
By combining structural, energetic, and electronic analyses, the study paints a consistent picture: C3N can host several toxic gases via non‑covalent forces strong enough to be detected but weak enough to allow rapid desorption, with calculated recovery times ranging from fractions of a microsecond to well under a millisecond at room temperature. Among the tested pollutants, nitrogen trichloride stands out as the one that most strongly perturbs the electrical properties of the sheet. To a lay reader, the bottom line is that this ultrathin carbon–nitrogen scaffold looks like a promising building block for compact, low‑power, and reusable sensors that could one day help watch over industrial sites, battlefields, and city air for some of the most troublesome invisible gases.
Citation: Azam, T., Ahmad, Z., Sarfaraz, S. et al. Theoretical insights of 2D carbon nitride (C3N) as a highly selective sensor for volatile analytes. Sci Rep 16, 5780 (2026). https://doi.org/10.1038/s41598-026-35679-z
Keywords: gas sensing, 2D materials, carbon nitride, toxic gases, electrochemical sensors