Clear Sky Science · en
A functional and robust cellular model for high-throughput screening of piezo1 modulators
Why tiny pressure sensors in our cells matter
Every time blood rushes through our vessels, or we simply walk across a room, our cells feel tiny mechanical forces. A key "pressure sensor" protein called Piezo1 helps cells translate these forces into electrical and chemical signals that keep tissues healthy. Because Piezo1 is involved in blood vessel growth, bone strength, immunity, and rare genetic diseases, drug developers are eager to find molecules that can tune its activity. The paper described here presents a new, faster way to search for such molecules using engineered cells and light-based readouts instead of slow, labor‑intensive techniques.
Turning touch into a light signal
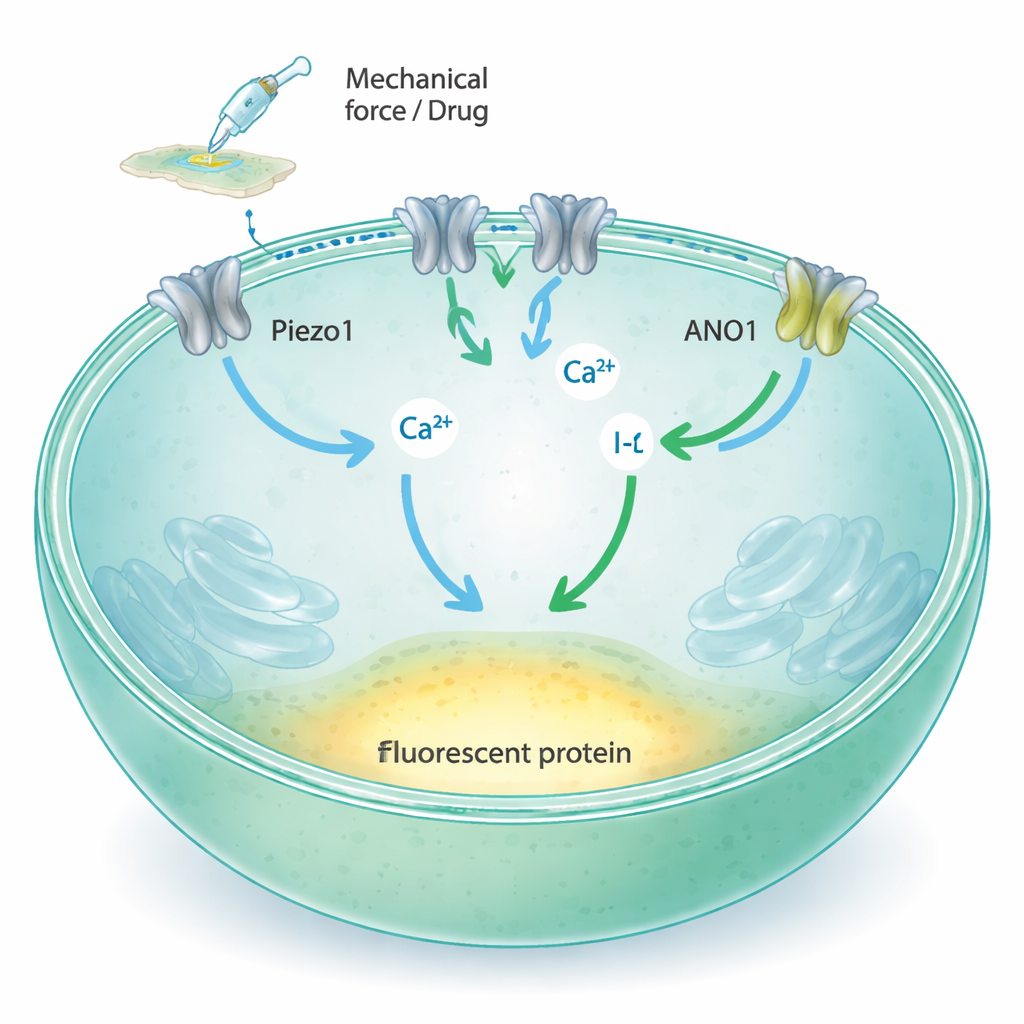
Piezo1 is a channel in the cell membrane that opens when the membrane is stretched or pushed, allowing calcium and other charged atoms to flow into the cell. Traditional methods to study such channels rely on fine glass electrodes or specialized fluorescent reporters, which are powerful but difficult to scale to the tens of thousands of tests needed for modern drug discovery. The authors set out to convert Piezo1 activity into a simple change in light that a plate reader can measure quickly across many samples at once. Their idea was to link Piezo1 to two additional components: ANO1, another ion channel that responds to calcium, and a modified yellow fluorescent protein that loses brightness when exposed to iodide ions.
Building a responsive test cell
The researchers started with Fischer Rat Thyroid (FRT) cells, which naturally produce Piezo1 and stick well to standard plastic plates. They confirmed that these cells express Piezo1 but not its close relative Piezo2, and showed that Piezo1 in these cells responds to fluid flow and mechanical probing by letting in calcium. Next, they introduced the ANO1 channel and the iodide‑sensitive fluorescent protein (YFP‑H148Q/I152L) into the cells. When calcium rises inside the cell, ANO1 opens and allows iodide to enter; the fluorescent protein then becomes dimmer as iodide builds up around it. Microscopy, flow cytometry, and electrical recordings showed that both ANO1 and the fluorescent sensor were present at high levels and working as intended.
From drug action to a measurable glow
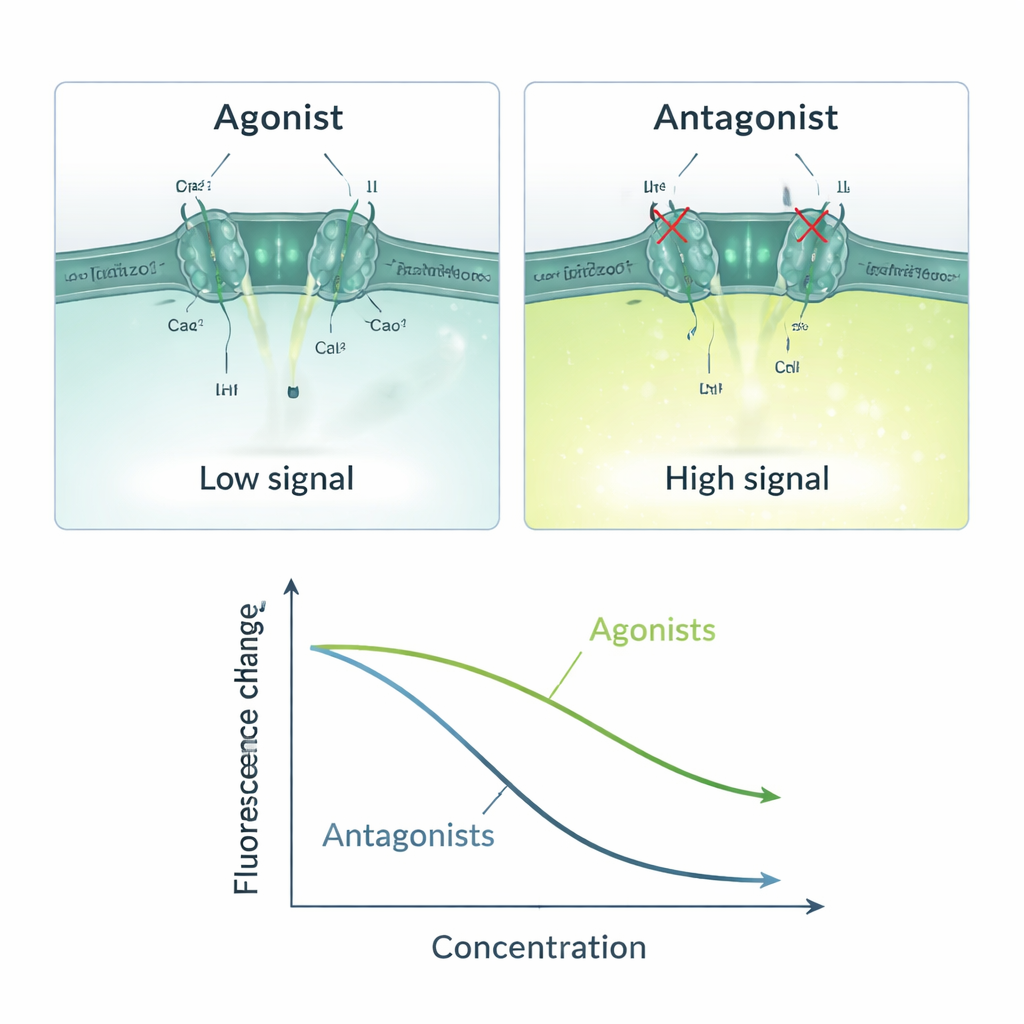
With this three‑part system in place, the team created a simple cause‑and‑effect chain: if a test molecule activates Piezo1, calcium flows in, ANO1 opens, iodide rushes in, and the fluorescence signal drops; if a molecule blocks Piezo1, the signal stays bright. They validated this logic using well‑known Piezo1 activators (Yoda1, Jedi1, Jedi2) and blockers (Ruthenium Red, GsMTx4). Activators produced dose‑dependent decreases in fluorescence, with sensitivity values matching previously reported data. Blockers caused the opposite pattern, reducing the response as their concentration increased. The assay worked only when both calcium and iodide were present, underscoring that the signal truly depended on Piezo1‑driven calcium entry and subsequent ANO1‑mediated iodide influx. Statistical measures such as the Z‑factor and signal‑to‑noise ratio indicated that the method is stable and reliable enough for industrial‑scale screening.
Fast, scalable testing with some caveats
The new cell model can complete a single well measurement in about 14 seconds and scan an entire 96‑well plate in roughly 22 minutes, and it can be adapted to instruments that read hundreds of wells simultaneously. The engineered cells remain stable for many generations under antibiotic selection, allowing repeated experiments over time. Because the readout is optical and uses standard microplate equipment, the approach is relatively low‑cost and accessible. However, the authors note that their test is indirect: any compound that acts on steps before or after Piezo1 in the signaling chain—such as ANO1 itself or other calcium‑handling proteins—may produce a false positive. For that reason, hits from this screen still need confirmation with more direct methods like electrophysiology.
What this means for future treatments
In everyday terms, the researchers have built a lab "smoke alarm" that flashes dimmer whenever a compound nudges Piezo1 open, and stays bright when Piezo1 is blocked. This alarm is sensitive, quick, and easy to run in large numbers, making it well suited for the first pass in the search for drugs that tune the body’s mechanical sensors. While it will not replace more detailed follow‑up tests, this cellular model provides a powerful starting point for discovering molecules that could one day help treat blood vessel disorders, bone diseases, immune problems, and other conditions linked to how our cells feel and respond to physical force.
Citation: Liu, X., Zheng, K., Wang, Y. et al. A functional and robust cellular model for high-throughput screening of piezo1 modulators. Sci Rep 16, 6048 (2026). https://doi.org/10.1038/s41598-026-35673-5
Keywords: Piezo1 channel, mechanosensitive ion channels, high-throughput screening, cell-based assay, drug discovery