Clear Sky Science · en
Radiosensitizing effects of silver nanoparticles targeting angiogenesis and matrix metalloproteinase signaling in triple negative breast cancer cell lines
Making Tough Breast Cancers More Vulnerable
Triple-negative breast cancer is one of the hardest forms of breast cancer to treat. It tends to grow quickly, spread early, and lacks the usual molecular "handles" that many modern drugs grab onto. Radiation is often part of treatment, but these tumors can be stubbornly resistant. This study asks a simple, intriguing question: can tiny particles of silver make radiation work better, while also slowing the tumor’s ability to grow new blood vessels and spread?
Tiny Silver Helpers for Radiation
The researchers focused on silver nanoparticles—ultra-small spheres of silver measured in billionths of a meter. Because silver is dense and interacts strongly with X-rays, these particles can concentrate the effects of radiation right where they accumulate. The team worked with two widely used laboratory models of triple-negative breast cancer, comparing them with a noncancerous breast cell line. They first tested how toxic silver nanoparticles were on their own and found that cancer cells were killed at much lower doses than normal cells, suggesting a useful treatment window. Using mathematical methods to analyze how the particles and radiation interact, they showed that combining the two produced a clearly stronger cancer-killing effect than either treatment alone.
Pushing Cancer Cells into Self-Destruct
Next, the scientists examined how the combined treatment killed cells. They measured apoptosis, a form of programmed cell suicide, and tracked the buildup of highly reactive molecules known as reactive oxygen species, or ROS. Radiation is already known to work in part by generating ROS that damage DNA. In both triple-negative breast cancer cell lines, silver nanoparticles alone raised apoptosis and ROS levels, and radiation alone did as well—but using them together pushed both measures dramatically higher. This means more cancer cells were nudged into an orderly self-destruct mode, rather than lingering in a damaged state that can sometimes encourage relapse or resistance.
Cutting Off Blood Supply and Slowing Spread
Tumors rely on blood vessels to bring oxygen and nutrients, and aggressive cancers can even mimic blood vessels themselves. The team looked at two key molecular switches involved in building and stabilizing tumor vessels, called VEGFR2 and Tie2. In both cell lines, silver nanoparticles reduced the activity of these genes, and the combination with radiation shut them down even further. They also examined two enzymes, MMP-2 and MMP-9, that help cancer cells chew through surrounding tissue and migrate. In one of the cell models, both radiation and silver nanoparticles lowered these enzymes, while the combined treatment had the strongest effect. When the scientists watched cancer cells move into an artificial “scratch” in a dish, and when they tested how well blood-vessel-like tubes formed in a co-culture system, the combination treatment most clearly slowed migration and tube building.
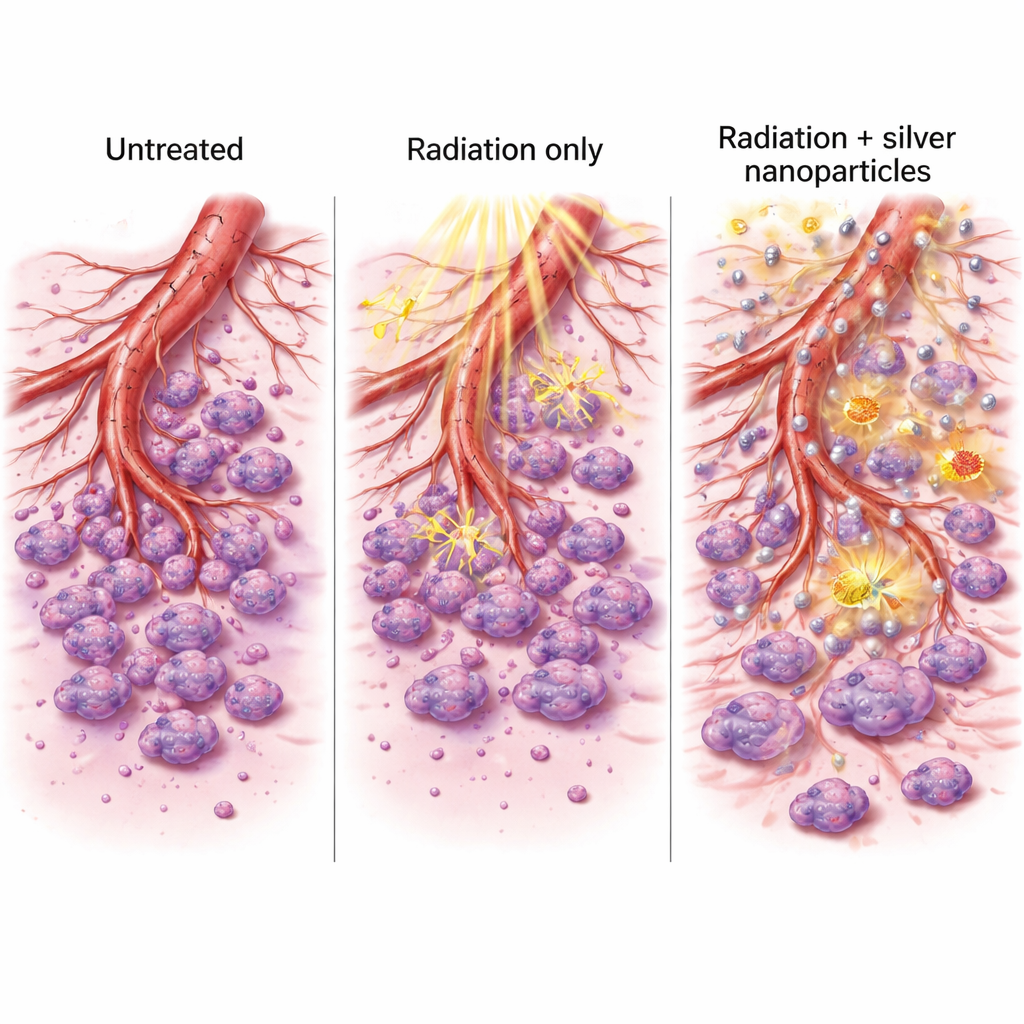
Why These Changes Matter
By dialing down genes that promote new blood vessel growth and enzymes that help cells invade, the silver-plus-radiation pairing did more than just kill cancer cells. It also disrupted the support systems tumors use to expand and spread. The treated cells were less able to close wounds in laboratory dishes and less able to organize into branch-like networks that resemble early blood vessels. Together with the strong boost in cell death and ROS production, these effects suggest a two-pronged benefit: stronger direct attack on the tumor and weaker tools for future growth and metastasis.
What This Could Mean for Patients
For now, these results come from cancer cells grown in the lab, not from patients. Still, they paint a hopeful picture. Silver nanoparticles acted as radiosensitizers, making standard radiation therapy more potent against triple-negative breast cancer cells. At the same time, they appeared to choke off the tumor’s ability to build blood supply lines and invade nearby tissue. If future animal and clinical studies confirm that these particles can be delivered safely and selectively to tumors, they could help doctors use lower radiation doses while getting better results, offering a new way to tackle one of the most challenging forms of breast cancer.
Citation: Montazersaheb, S., Farahzadi, R., Mansouri, E. et al. Radiosensitizing effects of silver nanoparticles targeting angiogenesis and matrix metalloproteinase signaling in triple negative breast cancer cell lines. Sci Rep 16, 6820 (2026). https://doi.org/10.1038/s41598-026-35662-8
Keywords: triple-negative breast cancer, silver nanoparticles, radiation therapy, angiogenesis, metastasis