Clear Sky Science · en
Rapid synergistic cloud point extraction of copper in environmental samples with greenness and toxicity evaluation using a triazole based Schiff base
Why cleaning copper from water matters
Copper is an essential nutrient, but like many good things, too much of it can be harmful. Even tiny excess amounts in rivers, drinking water, or supplements can threaten fish, wildlife, and human health. Detecting these low levels is surprisingly difficult, often requiring complex equipment and energy‑hungry preparation steps. This study introduces a fast, gentle way to “fish out” copper from water using a smart organic helper molecule and a mild soap‑like substance, making testing both more sensitive and more environmentally friendly.
A helper molecule that grabs copper
The researchers designed a new organic compound, called HIT, that acts like a tiny claw specifically shaped to grab copper ions. When HIT meets copper in water, they form a compact nanometer‑sized complex. Electron microscope images show that this copper–HIT complex forms nearly spherical particles only 13–25 billionths of a meter across. These nanoparticles are stable and easier to collect than free copper ions drifting in solution, which is crucial for detecting very low concentrations.
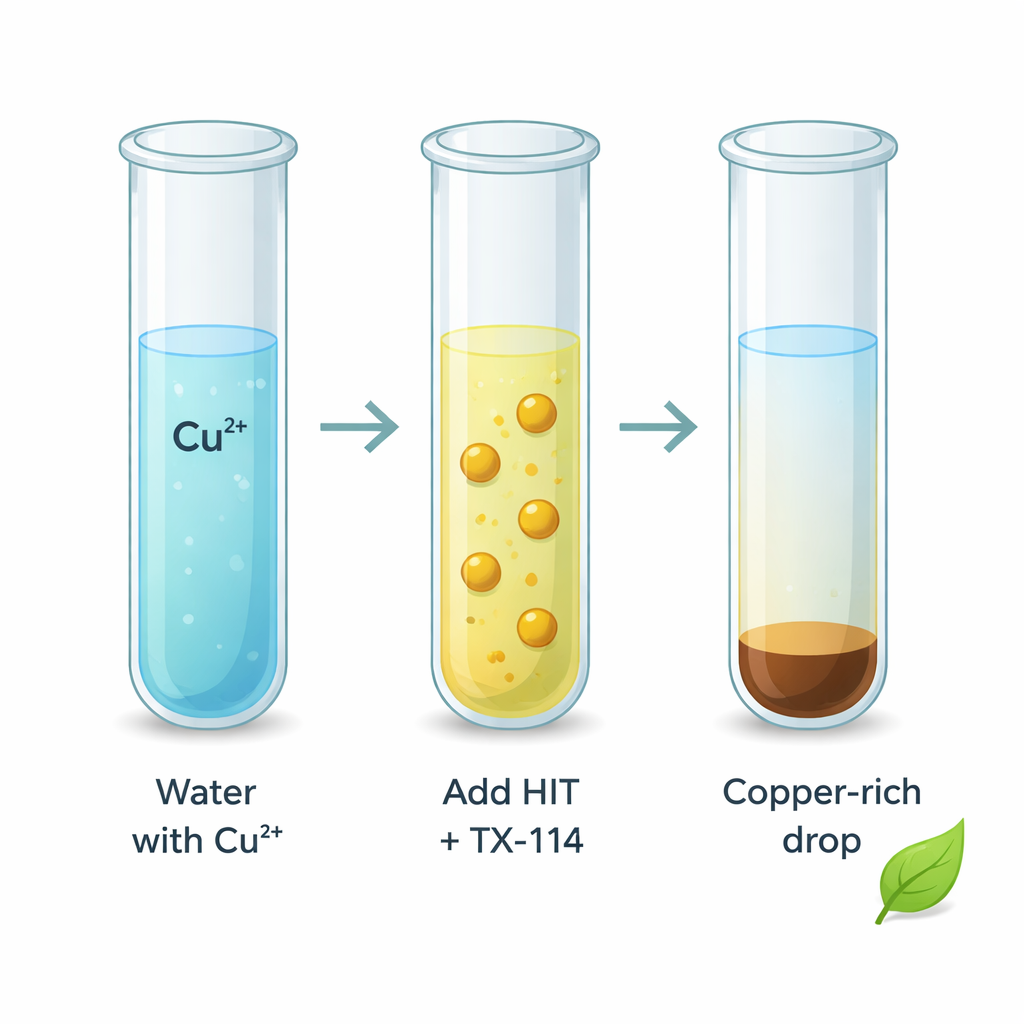
Using gentle soap to separate the metal
To pull the copper–HIT particles out of water, the team used a widely available, non‑ionic surfactant called Triton X‑114. This surfactant behaves like a gentle liquid soap: above a certain condition it gathers into microscopic droplets, or micelles, that are slightly oily compared with water. The copper–HIT nanoparticles prefer this micelle‑rich region. In a conventional “cloud point extraction,” the solution is heated until it turns cloudy, then spun so the surfactant‑rich phase containing the metal can be separated from the clear water. While effective, this traditional approach needs heating, waiting for the phases to separate, and cooling again, taking close to half an hour and using extra energy.
A faster, room‑temperature route
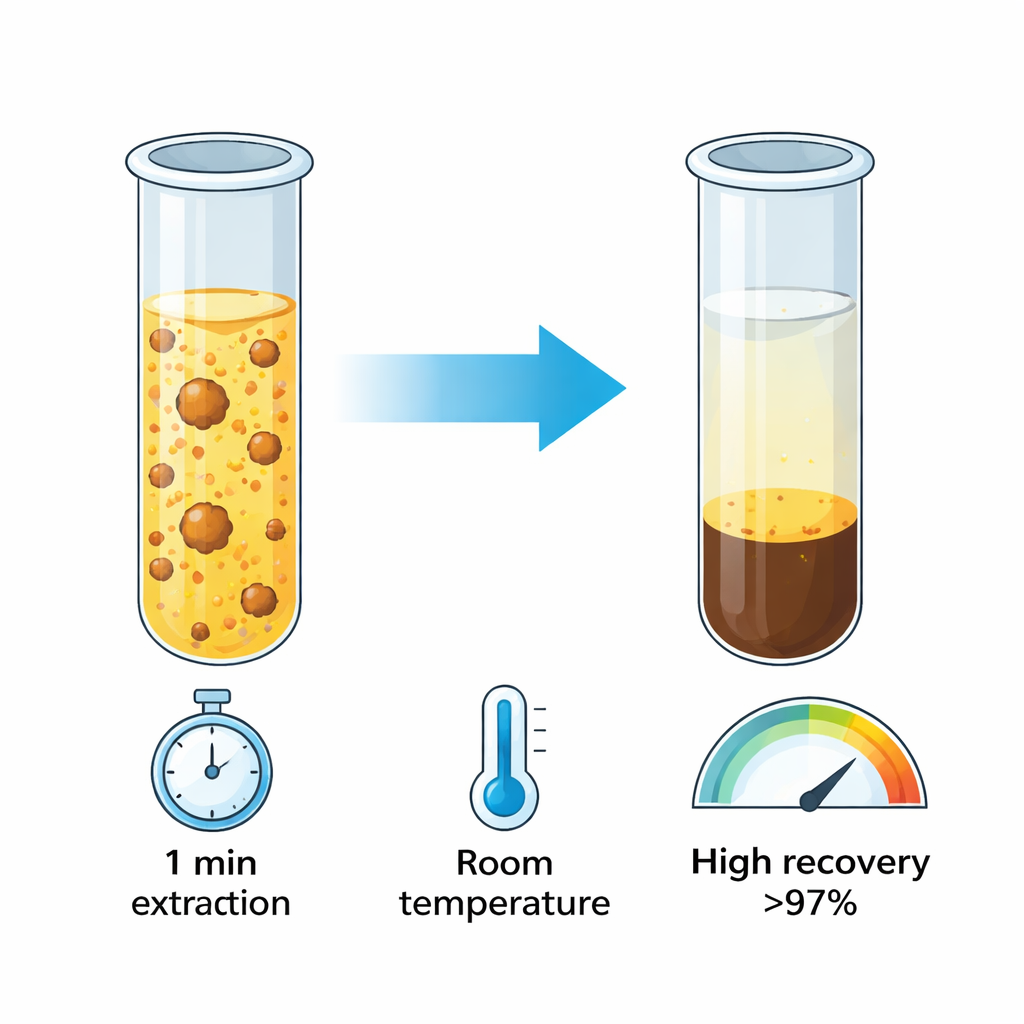
The authors developed a variation they call rapidly synergistic cloud point extraction, or RS‑CPE. Here, they add a small amount of decanol, an alcohol that works together with Triton X‑114 to trigger micelle formation immediately at room temperature. The mixture turns cloudy as micelles form and instantly collect the copper–HIT complex. A brief shake and a short spin in a centrifuge are enough to send the copper‑rich surfactant layer to the bottom of the tube. The entire extraction process now takes about one minute instead of dozens, and it works at ordinary room temperature without any heating step, cutting energy use and making the procedure far more convenient for routine testing.
Sharper measurements with a greener footprint
After concentrating the copper into a tiny volume, the team measured it using a sensitive technique called optical emission spectroscopy, which detects the light given off when atoms are excited in a hot plasma. Because RS‑CPE boosts the copper concentration by a factor of 125, the method can reliably detect levels down to tens of parts per trillion. Tests on tap water, Nile River water, seawater, and copper‑containing vitamin tablets showed that more than 97% of added copper was recovered, with excellent repeatability. The scientists also assessed how “green” their method is using established environmental scoring tools. RS‑CPE earned higher scores than the traditional approach, thanks to lower energy use, smaller reagent amounts, and reduced waste, while toxicity predictions for the new HIT compound and its copper complex indicated low to moderate hazard.
What this means for everyday water and health
In practical terms, this work offers a quick, low‑waste way to concentrate and measure tiny amounts of copper in real‑world samples, from drinking water to supplements. By combining a tailor‑made copper‑binding molecule with a mild surfactant and a clever room‑temperature extraction step, the researchers showed that we can improve both sensitivity and environmental safety at once. Such methods help laboratories and regulators monitor copper more easily and sustainably, supporting cleaner water and safer products without relying on harsh chemicals or energy‑intensive procedures.
Citation: Akl, M.A., Ghaith, E.A. & Mostafa, A.G. Rapid synergistic cloud point extraction of copper in environmental samples with greenness and toxicity evaluation using a triazole based Schiff base. Sci Rep 16, 4719 (2026). https://doi.org/10.1038/s41598-026-35659-3
Keywords: copper in water, green analytical chemistry, cloud point extraction, nanocomplex, environmental monitoring