Clear Sky Science · en
Chlorinated bis-4-hydroxycoumarins suppress flavivirus replication by inhibiting dengue virus type 2 translation and replication
New Hopes Against Mosquito-Borne Viruses
Dengue and Zika viruses infect hundreds of millions of people each year, often in regions with limited medical resources. Vaccines offer only partial protection, and there is still no widely used antiviral drug that doctors can prescribe when patients first fall ill. This study explores a family of lab‑made chemicals derived from plant molecules, asking a simple but urgent question: can any of them reliably slow down these viruses inside human cells without harming the cells themselves?
Plant-Inspired Molecules as Virus Blockers
The researchers focused on coumarins, a class of natural compounds found in many plants and long known for their antimicrobial and antiviral potential. They tested twelve “biscoumarin” derivatives—two coumarin units linked together—against dengue virus type 2 and Zika virus in cultured cells. By measuring how many infectious virus particles were produced, and how healthy the cells remained, they discovered that two chlorinated variants, labeled compound 3 and compound 4, were especially promising. At low micromolar concentrations, these chlorinated bis-4-hydroxycoumarins cut virus production by more than 90% while keeping most cells alive and functioning.
Fine-Tuning Chemistry for Stronger Protection
Not all chemical tweaks worked equally well. By systematically changing the atoms around a ring-shaped part of the molecules, the team found that attaching chlorine at specific positions gave the best antiviral activity. Swapping chlorine for other halogens such as fluorine or bromine—or adding different chemical groups—usually weakened the effect on dengue and Zika. Using computer-based models, they linked antiviral strength to simple molecular features such as how charge is distributed across the molecule and how much surface area it exposes. Machine-learning methods captured these relationships far better than older statistical tools, suggesting that artificial intelligence can help guide the design of improved anti-dengue drug candidates.
How the Compounds Disrupt the Virus Life Cycle
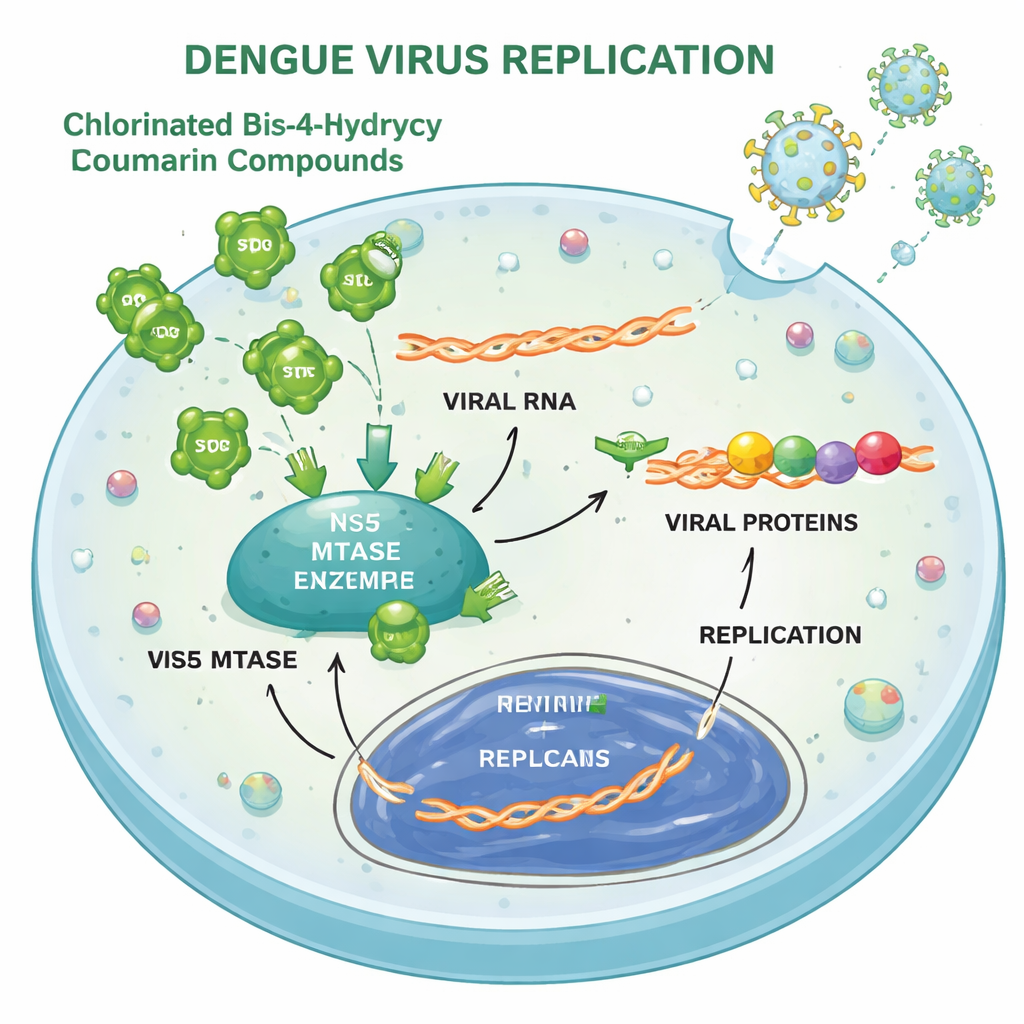
Once inside a cell, dengue virus uses its RNA genome as a blueprint to make a long polyprotein that is cut into working parts, then copies its RNA to spawn new viruses. The study shows that compounds 3 and 4 interfere mainly with these translation and replication steps. In infected cells, levels of a key viral coat protein dropped dramatically when the compounds were present, and a reporter system that tracks viral RNA replication also dimmed in a dose‑dependent way. Biochemical tests and computer docking pointed to one viral enzyme, called the NS5 methyltransferase, as a direct but relatively weak target: the compounds could inhibit its RNA “capping” function, though not nearly as strongly as a well‑known reference inhibitor. They also modestly slowed the virus’s protease, another enzyme used to cut the viral polyprotein.
Viruses Struggle to Adapt to the Attack
Viruses often escape drugs by mutating. To see whether this could happen here, the team repeatedly grew dengue virus in cells bathed in the new compounds over many infection cycles. Several changes did arise in a different viral protein, NS4B, which helps bend cell membranes into tiny pockets where replication occurs. Surprisingly, these mutated viruses were no less sensitive to the compounds than the original strain. Computer-predicted structures suggested that the overall shape of NS4B stayed much the same. This pattern hints that the mutations were general adjustments to life under stress, not true resistance. It also supports the idea that the compounds act on multiple parts of the replication machinery or on host cell factors that the virus cannot easily rewire.
Why These Findings Matter for Future Treatments
For non‑specialists, the main message is that the researchers have uncovered a new chemical “scaffold” that can slow dengue and Zika viruses at several points in their life cycle, particularly when the viruses are trying to translate their genes and copy their RNA inside cells. The most promising molecule, compound 3, works at low doses, harms cells relatively little, hits all four major dengue types as well as Zika, and does not quickly drive the virus to evolve resistance in the lab. Although these chlorinated bis-4-hydroxycoumarins are still far from being medicines, they provide a solid starting point for chemists and virologists to refine, test in animals, and eventually combine with other drugs—steps that could bring us closer to a much‑needed antiviral pill for mosquito‑borne infections.
Citation: Loeanurit, N., Phan, THT., Hengphasatporn, K. et al. Chlorinated bis-4-hydroxycoumarins suppress flavivirus replication by inhibiting dengue virus type 2 translation and replication. Sci Rep 16, 5300 (2026). https://doi.org/10.1038/s41598-026-35654-8
Keywords: dengue virus, Zika virus, antiviral compounds, coumarin derivatives, viral replication