Clear Sky Science · en
DFT calculation of Ac3+ and Bi3+ complexation with hybrid chelator 3p-C-DEPA for targeted alpha therapy
Why this research matters for cancer treatment
Modern cancer medicine increasingly relies on radioactive drugs that can hunt down tumors cell by cell. A powerful approach, called targeted alpha therapy, uses intensely energetic particles that can kill individual cancer cells with remarkable precision. But to deliver these particles safely, the radioactive metal must be locked into a tiny molecular “cage” so it does not wander into healthy tissues. This study explores a new cage molecule, 3p‑C‑DEPA, designed to hold especially challenging metals such as actinium‑225 and bismuth‑213, and asks: can it bind them more securely than today’s workhorse chelator, DOTA?
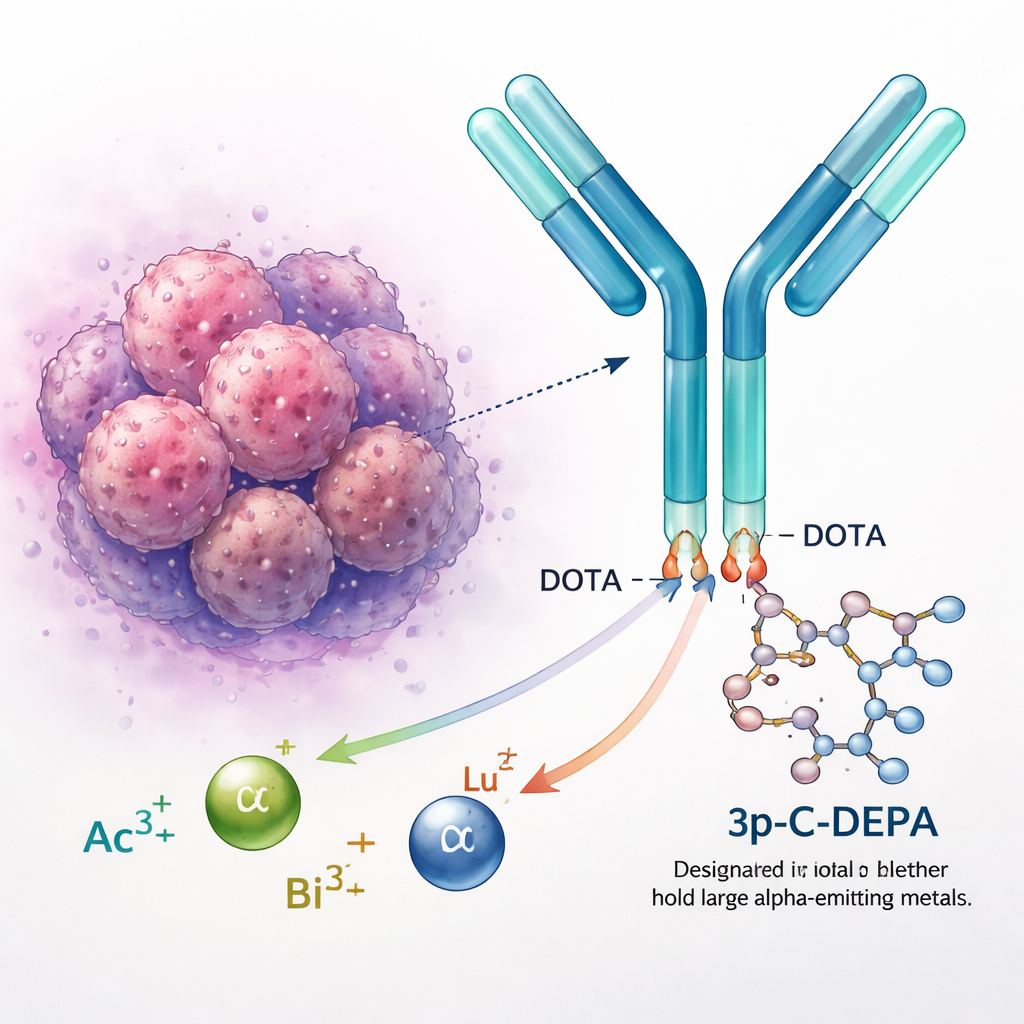
Locking down radioactive metals safely
Radioactive metals like actinium, bismuth, and lutetium are used to diagnose and treat cancer. On their own, however, these positively charged metal ions would interact freely with the body, potentially harming healthy organs. Chemists therefore attach them to “chelators,” ring‑shaped molecules that wrap around the metal and hold it in place. The gold‑standard chelator DOTA is used in several approved drugs, but it struggles with larger, more diffuse ions such as actinium‑225. These metals can slip out over time, raising concerns about long‑term safety and limiting how widely targeted alpha therapy can be used.
A hybrid cage built for bigger atoms
The new chelator, 3p‑C‑DEPA, combines features of two established designs: the rigid, ring‑like framework of DOTA and the more flexible, open structure of another chelator called DTPA. This hybrid architecture gives 3p‑C‑DEPA ten strong binding “hands” (nitrogen and oxygen atoms) compared with DOTA’s eight, and a roomier cavity that can better accommodate large metal ions. Earlier laboratory work suggested that 3p‑C‑DEPA can label antibodies quickly at mild temperatures and hold bismuth‑based radionuclides stably in blood serum. The current study takes the next step, using quantum‑level calculations to systematically compare how well 3p‑C‑DEPA and DOTA bind lutetium‑177, bismuth‑213, and actinium‑225.
Peering into the molecular handshake
Because working directly with short‑lived alpha emitters is difficult, the researchers turned to density functional theory (DFT), a powerful computational method that estimates how electrons are arranged in molecules and how strongly atoms attract each other. They modeled each metal ion in water, then its complex with either DOTA or 3p‑C‑DEPA, and calculated the change in free energy when the metal moves from water into the chelator’s cage. This energy change is translated into a “stability constant”: the higher the value, the more tightly the chelator holds the metal. Two different DFT approaches and two solvation models were used to mimic realistic solution conditions and to check that the trends were robust rather than artifacts of a single computational setup.
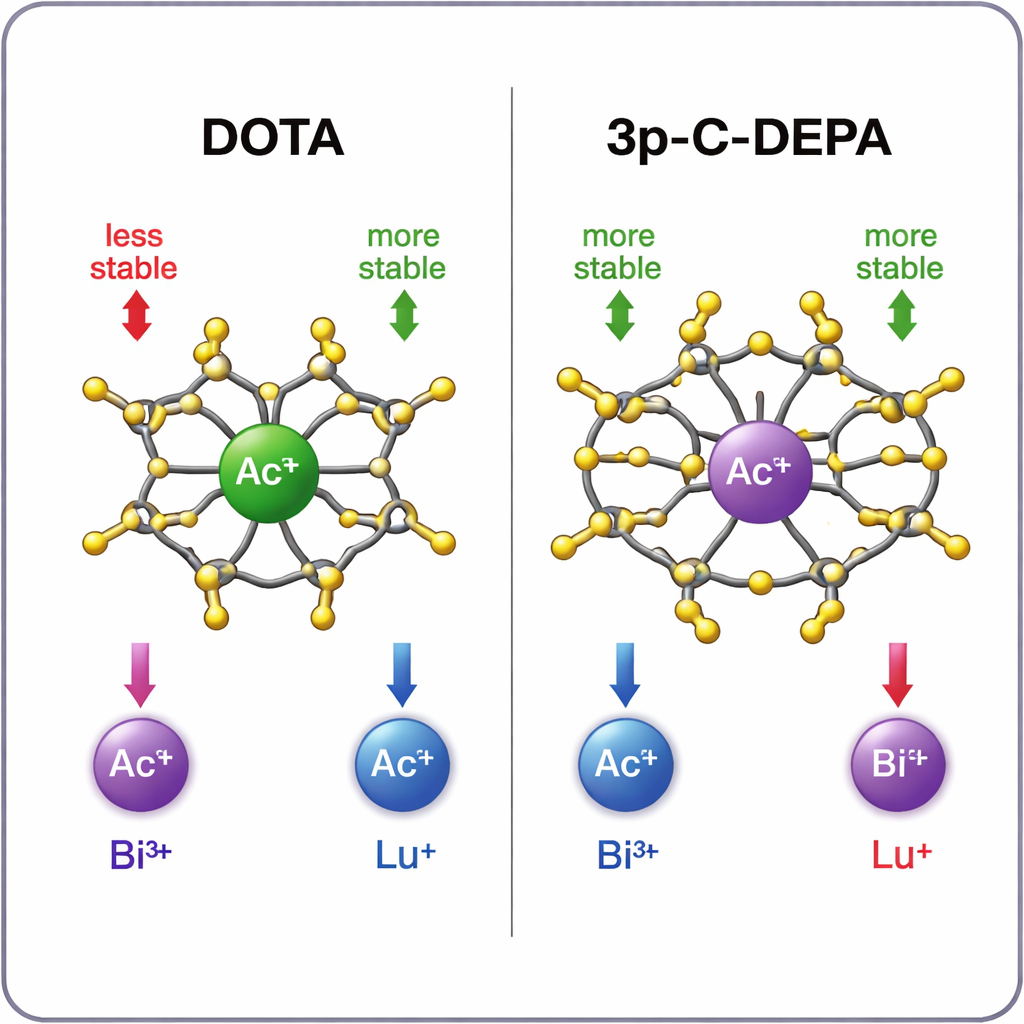
Which cage holds which metal best?
The simulations show a clear pattern. For the large actinium ion, 3p‑C‑DEPA forms a markedly more stable complex than DOTA, thanks to its larger cavity and greater number of oxygen donors that can grip the metal. Bismuth‑213 is also well accommodated by 3p‑C‑DEPA, benefiting from both its size and its electron‑accepting character, which matches the chelator’s electron‑donating groups. In contrast, the smaller lutetium‑177 fits more comfortably into DOTA’s tighter, eight‑armed environment. In 3p‑C‑DEPA, excess donor groups crowd around the small ion, generating repulsive interactions that appear to slow labeling and slightly weaken the final complex. Calculated stability trends agree well with available experimental data and radiolabeling yields, lending credibility to the theoretical predictions.
What this means for future cancer therapies
In everyday terms, the study suggests that DOTA is not a one‑size‑fits‑all cage: it works very well for smaller metals like lutetium, but is suboptimal for bulkier alpha emitters such as actinium‑225. 3p‑C‑DEPA, by contrast, behaves like a custom‑built holder for these larger ions, gripping them more tightly and under gentler conditions that suit delicate targeting molecules like antibodies. While further experimental and clinical work is needed, the calculations point to 3p‑C‑DEPA as a promising backbone for safer, more effective targeted alpha therapies—potentially expanding access to highly potent cancer treatments that can eradicate tumors while sparing surrounding healthy tissue.
Citation: Ramdhani, D., Watabe, H., Ahenkorah, S. et al. DFT calculation of Ac3+ and Bi3+ complexation with hybrid chelator 3p-C-DEPA for targeted alpha therapy. Sci Rep 16, 6587 (2026). https://doi.org/10.1038/s41598-026-35633-z
Keywords: targeted alpha therapy, radiopharmaceuticals, chelator design, actinium-225, density functional theory