Clear Sky Science · en
Experimental investigation and thermodynamic correlation of chlordiazepoxide solubility in supercritical CO₂
Why dissolving a familiar drug in a new way matters
Many modern medicines work well but are hard to formulate efficiently: they may not dissolve easily, may degrade during manufacturing, or require large amounts of organic solvents. This study looks at chlordiazepoxide, a long‑used anxiety medication, and asks a practical question with wide-reaching impact: how well does it dissolve in supercritical carbon dioxide, a cleaner, tunable solvent that could help make safer and more effective drug particles?
A green fluid that behaves like both gas and liquid
Supercritical carbon dioxide is carbon dioxide pushed above a certain pressure and temperature until it is neither a typical gas nor a normal liquid. In this state it flows like a gas but has a liquid‑like density that can dissolve many substances. Industry already uses it to decaffeinate coffee and extract flavors and oils. For pharmaceuticals, supercritical CO₂ is attractive because it is non‑toxic, approved by regulators, inexpensive, and easy to recycle. It can help create tiny, uniform drug particles and reduce the need for harsh organic solvents—if, and only if, the drug actually dissolves in it to a useful extent.
Measuring how an anxiety drug behaves under pressure
The authors set out to measure, for the first time, how much chlordiazepoxide dissolves in pure supercritical CO₂ over a practical processing window. They placed solid drug powder in a high‑pressure cell and flowed CO₂ through it at pressures between 12 and 30 megapascals and temperatures between 308 and 338 kelvin (about 35–65 °C). After allowing the system to reach equilibrium, they sampled the CO₂ phase, rapidly expanded it into a solvent, and used ultraviolet–visible light absorption to determine how much drug had been dissolved. Overall solubility ranged from about 20 to 576 parts per million, corresponding to mole fractions between 0.0198×10⁻³ and 0.576×10⁻³, values in line with many other moderately soluble drugs in supercritical CO₂.
How pressure and temperature shape solubility
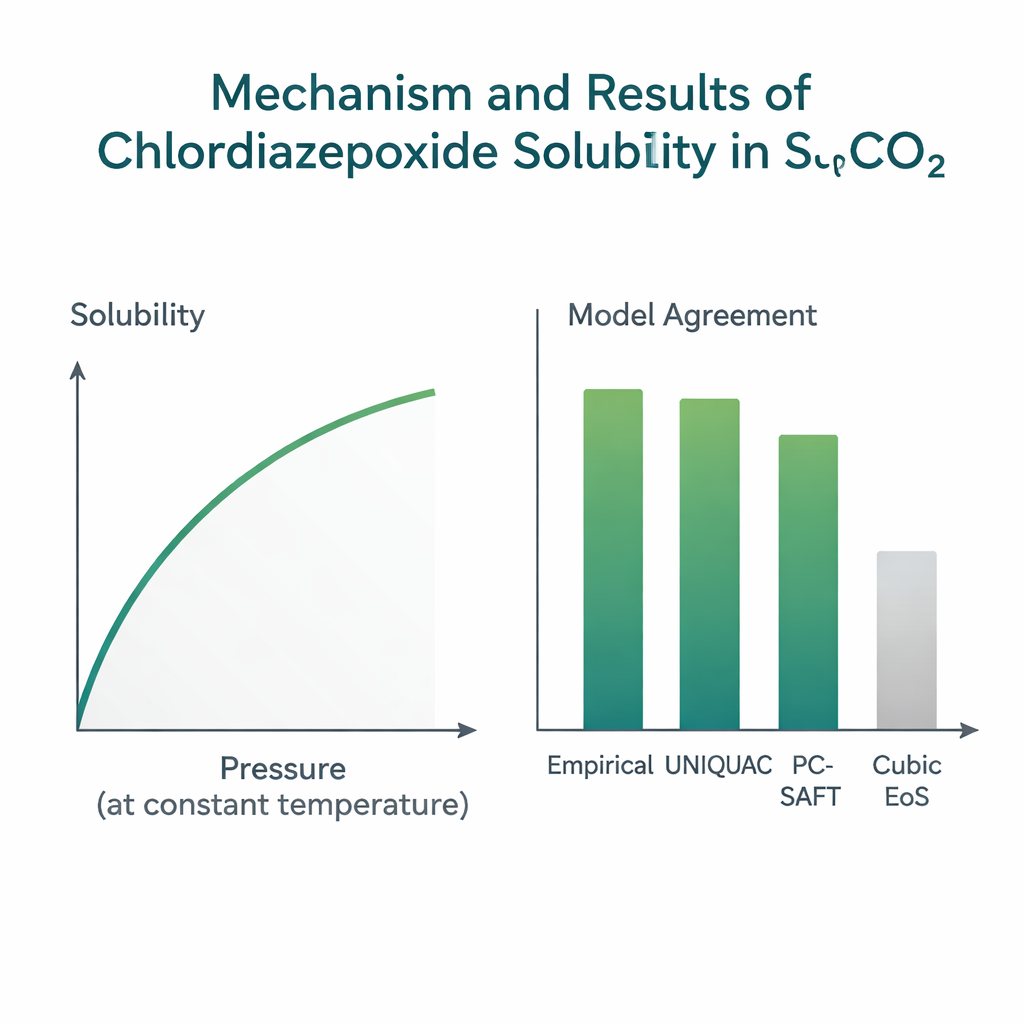
The measurements revealed clear, intuitive patterns. At a fixed temperature, raising the pressure always increased solubility. Higher pressure squeezes CO₂ molecules closer together, making the supercritical phase denser and better at surrounding and carrying away drug molecules. Temperature had a more subtle effect. At lower pressures, warming the system tended to reduce solubility because it thinned out the CO₂ and lowered its dissolving power. Above a crossover pressure of roughly 19 megapascals, the trend reversed: higher temperature increased solubility because it boosted the drug’s tendency to escape from the solid state into the fluid. This balance between fluid density and the drug’s own volatility is a hallmark of supercritical systems and is important for choosing practical operating conditions.
Teaching models to predict what the lab sees
Because running high‑pressure experiments is slow and costly, engineers rely on mathematical models to predict solubility under new conditions or for related drugs. The team tested several families of models using their new data set. Simple “density‑based” empirical formulas, which link solubility directly to CO₂ density and temperature using just a few fitting constants, performed the best. In particular, the long‑established Chrastil correlation matched the data with an average deviation of about 5%, and other similar formulas also did well. A more physically detailed “expanded‑liquid” approach called UNIQUAC, which accounts for molecular size, shape, and interaction energies, achieved comparable accuracy, around 6% deviation. By contrast, commonly used cubic equations of state—general-purpose formulas for fluid behavior—showed deviations near 20%, missing finer details of how this specific drug interacts with CO₂.
What this means for future drug manufacturing
In plain terms, the study shows that chlordiazepoxide does dissolve in supercritical CO₂ at technologically relevant conditions, and that its behavior can be captured very well with relatively simple, well‑chosen models. This provides a reliable map for designing greener processes to make new solid forms or nanoparticles of the drug, which can improve stability or absorption in the body. More broadly, the work supplies a rare, carefully measured data set for a widely used medicine and demonstrates which modeling tools are most trustworthy when planning supercritical CO₂ processes—information that can guide cleaner, more efficient manufacturing of many other drugs.
Citation: Saadati Ardestani, N., Noubigh, A., Esfandiari, N. et al. Experimental investigation and thermodynamic correlation of chlordiazepoxide solubility in supercritical CO₂. Sci Rep 16, 6552 (2026). https://doi.org/10.1038/s41598-026-35623-1
Keywords: supercritical carbon dioxide, drug solubility, chlordiazepoxide, green pharmaceutical processing, thermodynamic modeling