Clear Sky Science · en
Production of biopolymer and polymer from carbon dioxide employing ionic liquid supported on dendritic fibrous nanosilica
Turning a Climate Problem into Everyday Materials
Carbon dioxide (CO2) is usually seen as a climate villain, but it is also a rich, untapped raw material. This study explores how to turn CO2 into useful plastics and bioplastics under relatively gentle conditions, using a smart, recyclable solid catalyst. The work points toward cleaner ways to make materials for packaging, coatings, and foams while reusing a waste gas that is warming the planet.

A New Way to Build Plastics from Waste Gas
Most plastics today come from fossil fuels and often require harsh chemicals and energy-intensive steps. Chemists have long known that CO2 can, in principle, be stitched into polymer chains, but existing methods usually demand high pressures, high temperatures, and catalysts that are hard to recover and reuse. In this study, the authors developed a solid catalyst that can efficiently couple CO2 with small reactive molecules called oxetane, epoxides, and limonene epoxide (derived from citrus peel oil). The result is a family of polymers and biopolymers, including poly(trimethylene carbonate), made under comparatively mild conditions with impressive yields of up to 98%.
A Fibrous Sponge as a Smart Catalyst Support
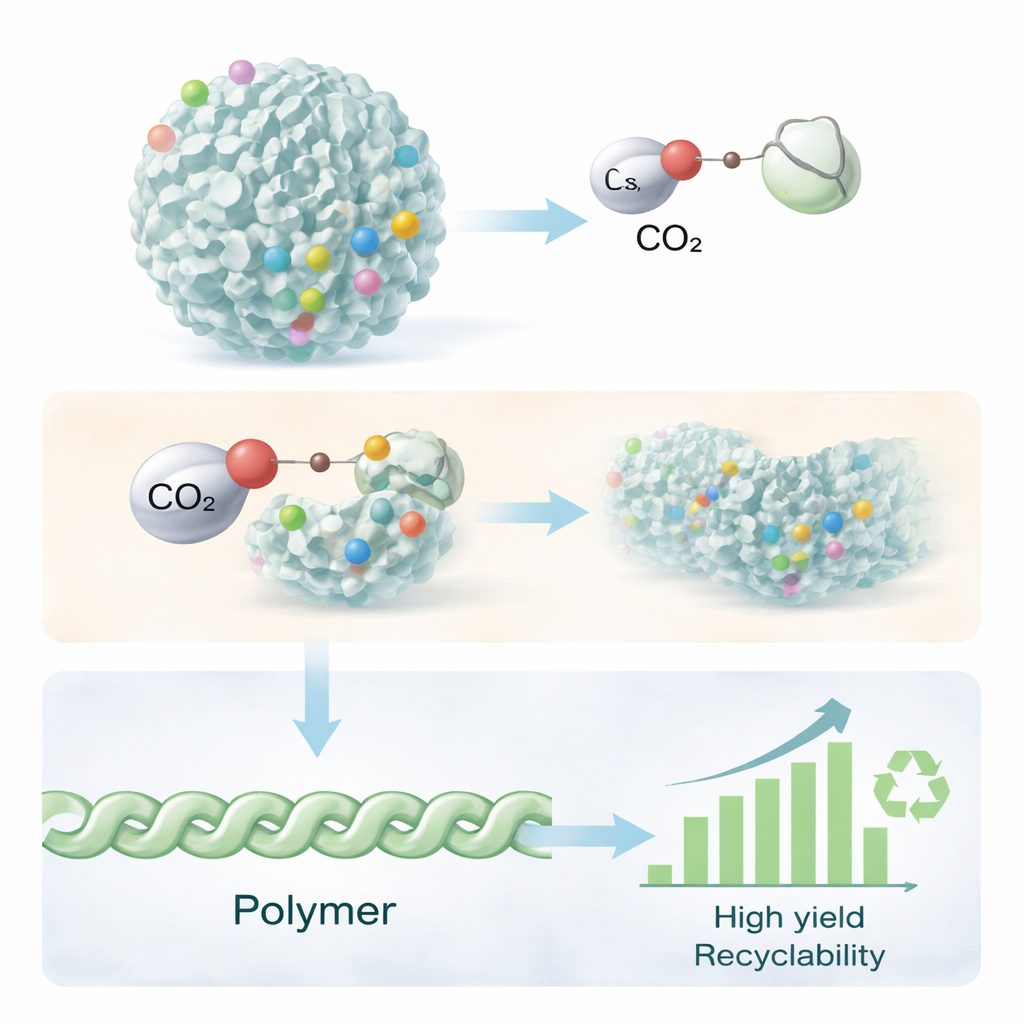
The heart of the system is a tiny, ball-shaped material called dendritic fibrous nanosilica (DFNS). Under the microscope, DFNS looks like a sea urchin or a pom-pom, with many thin silica fibers radiating outward. This unusual structure gives it an enormous surface area and easy access to internal spaces, making it an ideal scaffold for holding active catalytic sites. The researchers chemically attached special salts known as ionic liquids onto the surface of DFNS. These ionic liquids carry carbonate groups that can grab and activate CO2, while the surrounding silica framework keeps them well spaced, stable, and easy to handle as a solid powder.
How the Catalyst Works and Why It Matters
To test their design, the team carried out reactions in a small high-pressure vessel. They mixed one of the small ring-shaped molecules (such as an epoxide) with a tiny amount of the DFNS–ionic liquid catalyst, flushed the vessel with CO2, and heated it to about 100 °C under moderate pressure. Under these conditions, the activated CO2 and the ring molecule open up and join together repeatedly, forming long polymer chains. Careful measurements showed that the fibrous silica kept its structure even after being coated with ionic liquid, and that the active sites remained accessible. Compared with other supports such as plain silica or more conventional porous materials (SBA-15, MCM-41), the DFNS-based catalyst produced significantly higher polymer yields under the same conditions.
From Waste Oils to Greener Plastics
Beyond simple model molecules, the researchers pushed their system toward more practical, bio-based feedstocks. They converted waste vegetable oils, rich in fatty acids like oleic and linoleic acid, into epoxidized oils and then into “carbonated” oils using the same DFNS–ionic liquid catalyst and CO2. These carbonated oils can be further reacted with small amines to create non-isocyanate polyurethanes, a class of polymers that avoid the toxic isocyanates used in standard polyurethane production. The catalyst delivered high conversions and could be filtered off and reused for at least ten cycles with little loss of activity, underscoring its promise for real-world processes.
Cleaner Chemistry with Reusable Nanosponges
Overall, the study shows that a carefully engineered nanospongelike material can turn CO2 from a waste gas into a building block for useful polymers, using lower temperatures and pressures than many competing methods. By combining a high-surface-area fibrous support with tailor-made ionic liquids, the authors created a robust, recyclable catalyst that works for both simple epoxides and complex mixtures derived from used cooking oils. For non-specialists, the key takeaway is that smart material design can help close the carbon loop: instead of simply emitting CO2, we can increasingly lock it into everyday materials made through cleaner, more sustainable chemistry.
Citation: He, J., Gao, C., Feng, D. et al. Production of biopolymer and polymer from carbon dioxide employing ionic liquid supported on dendritic fibrous nanosilica. Sci Rep 16, 6313 (2026). https://doi.org/10.1038/s41598-026-35620-4
Keywords: carbon dioxide utilization, green polymers, nanocatalyst, ionic liquids, waste vegetable oil