Clear Sky Science · en
Efficacy and safety of cryoablation procedures using mouse models of bladder cancer
Freezing Tumors Instead of Removing the Bladder
For many people with advanced bladder cancer, the standard cure involves removing the entire bladder, a life‑altering operation that permanently changes how they urinate. This study explores a very different idea: using extreme cold to freeze and kill tumors while leaving the bladder in place. By testing this approach in mice, the researchers asked a question that matters to patients and families alike: could a simpler, organ‑sparing procedure control cancer, keep urination relatively normal, and even help the body’s own immune system fight off disease?
Why Bladder Removal Is So Hard on Patients
Bladder cancer is often divided into two main types: tumors that stay on the surface and those that invade the muscle wall. When cancer digs into the muscle, the usual treatment is to remove the whole bladder, a major operation called radical cystectomy. Although lifesaving for many, this surgery is highly invasive and forces doctors to reroute urine through a bag on the skin or a surgically created pouch. These changes can dramatically reduce quality of life, especially in older or medically frail patients who may not even be able to undergo such an operation. Because the survival benefit is far from perfect, doctors and patients have long searched for alternatives that are both effective and easier on the body.
How the Freezing Treatment Was Tested
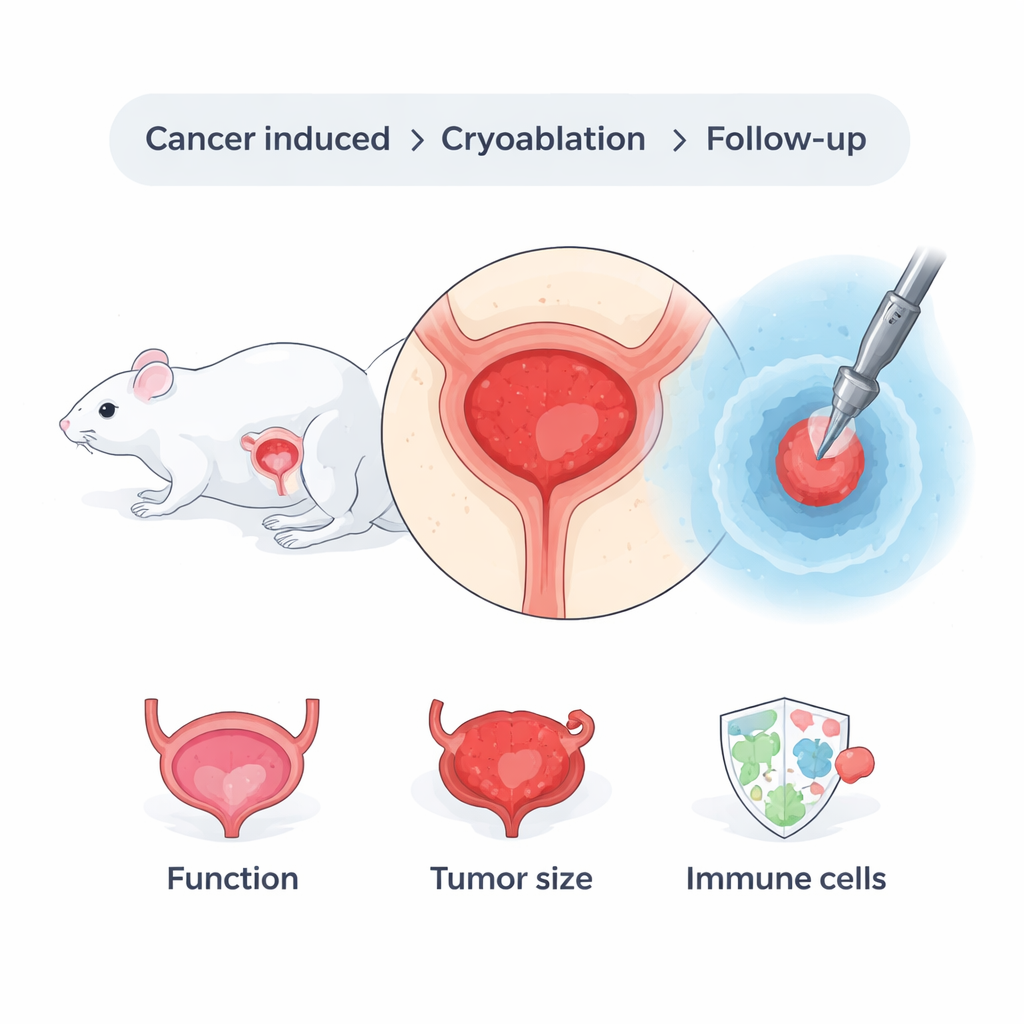
To explore one such alternative, the team used a well‑established method to induce bladder cancer in mice by adding a chemical, BBN, to their drinking water. Over several months, most of these mice developed tumors that grew into the muscle layer, mimicking the dangerous form of the disease seen in people. Once cancers were present, the researchers surgically exposed the bladder through a small abdominal incision and pressed a metal rod, pre‑cooled in liquid nitrogen, against the outer surface of the tumor‑bearing bladder. This created a tightly controlled “ice ball” that froze the tissue in two short freeze‑thaw cycles. Other mice underwent anesthesia and abdominal opening without freezing, serving as comparison animals to show what happens without the treatment.
Does Freezing Really Kill the Cancer and Spare Function?
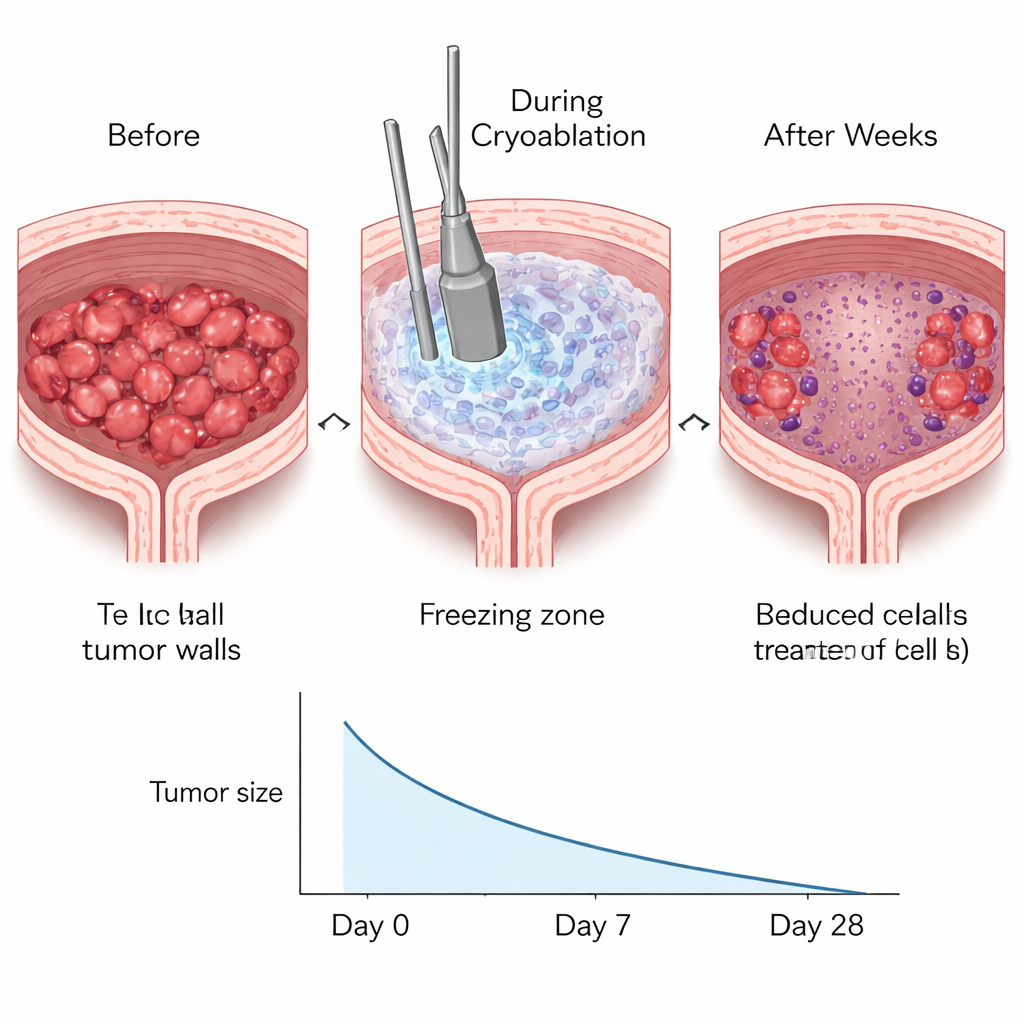
Careful microscopic examination of bladder tissue showed that the freezing procedure effectively destroyed cancer cells in the treated areas. Within hours, the frozen region appeared damaged, with the loss of smooth muscle cell nuclei and the formation of empty spaces where cells had died. Over the following days, these damaged zones shrank, and by about two weeks they were largely replaced by neighboring tissue. Tumor size decreased significantly in the weeks after the procedure. At the same time, the mice continued to drink, urinate, and maintain body weight at levels similar to untreated animals. A simple test that counted urine spots on paper showed that the frequency of urination did not worsen, suggesting that basic bladder function was preserved despite the localized injury caused by freezing.
Awakening the Body’s Own Defenses
The researchers were also interested in whether freezing the tumor might stir up the immune system. When cells die from extreme cold, they can release cancer‑related proteins in a way that the immune system can recognize. In this study, tissue samples taken from the treated bladders showed a marked rise, several weeks after cryoablation, in two key types of T cells: CD4 and CD8 cells. These immune cells help coordinate and carry out targeted attacks on abnormal cells. The increase was especially strong in mice that actually had bladder cancer, hinting that the immune system was responding not just to generic damage but to tumor‑specific signals at the treatment site. Interestingly, similar changes were not seen in the spleen, a major immune organ, suggesting that this response was localized in the bladder rather than spread throughout the body.
What This Could Mean for Future Cancer Care
Taken together, the findings suggest that carefully controlled freezing can shrink invasive bladder tumors in mice, avoid added deaths around the time of surgery, preserve basic urination, and draw helpful immune cells into the treated area. While this work was done with an external probe in small animals, the same principles could guide the design of devices that freeze tumors from inside the bladder in people. If such approaches prove safe and effective in further studies, they might one day offer patients a middle path between major organ‑removing surgery and less effective treatments—using cold not only to destroy visible cancer, but also to help the body keep watch for its return.
Citation: Inui, S., Ito, S., Ueda, T. et al. Efficacy and safety of cryoablation procedures using mouse models of bladder cancer. Sci Rep 16, 5157 (2026). https://doi.org/10.1038/s41598-026-35571-w
Keywords: bladder cancer, cryoablation, organ-sparing treatment, cancer immunology, mouse model