Clear Sky Science · en
Antibody-functionalized gold nanospheres for multimodal imaging
Sharper Views of Hidden Tumors
Catching cancer early often depends on how clearly doctors can see what is happening inside the body. This study introduces tiny gold-based particles that light up tumors in several different ways at once, giving doctors both a detailed map of where a head and neck tumor sits and clues about the tissue’s behavior. The work points toward future scans and surgeries that are faster, more accurate, and less likely to miss dangerous cells.
Why Better Imaging Matters
Head and neck cancers develop in areas such as the mouth, throat, and voice box, where surgery must balance removing all cancer with preserving speech and swallowing. Surgeons increasingly rely on fluorescent dyes that cause tumors to glow under special cameras, helping them trace the true edges of a mass. Yet most near‑infrared dyes, which are preferred because they see deeper into tissue with less background fog, shine too dimly inside the body. Computed tomography (CT) scans, on the other hand, give crisp anatomical pictures but reveal little about the biology of the tumor itself. The authors set out to bridge these gaps with a single tool that can serve both CT and advanced light‑based imaging.
Designing a Tiny Gold Beacon
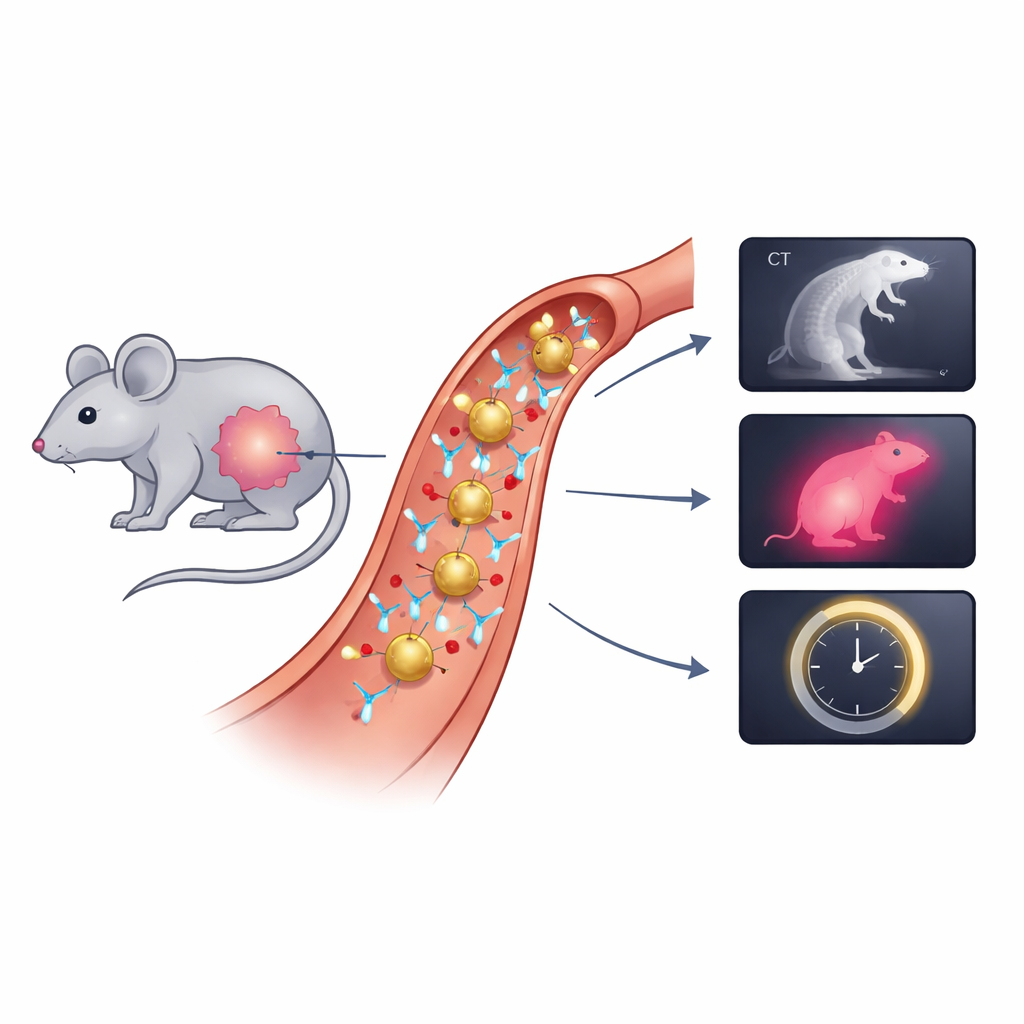
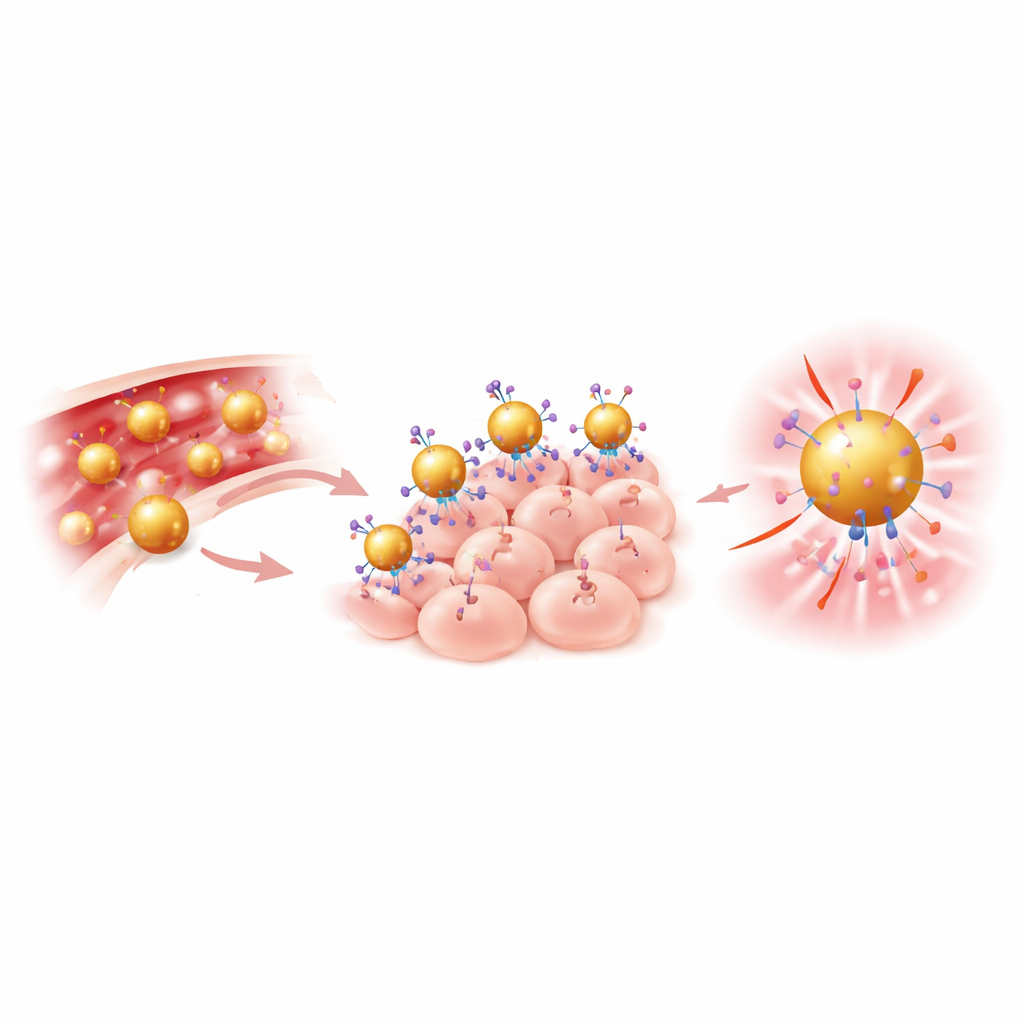
The team built a “gold nanodye” by starting with very small gold spheres about 25 nanometers wide—thousands of times thinner than a human hair. They coated the gold with flexible polymer chains to keep the particles stable in blood and then attached two key components: a widely used near‑infrared dye (IRDye 800) and antibodies that recognize a protein called EGFR, which is often found at high levels on head and neck cancer cells. This combined particle, called Anti‑AuND, is designed to home in on tumors via the antibodies, boost the dye’s brightness through interactions with the gold surface, and strongly block X‑rays so it also shows up on CT scans. Laboratory tests confirmed that the particles had the expected size, charge, and structure, and that cancer cells readily took them up.
How the Particles Perform in Mice
To test the new probes in living systems, the researchers implanted human‑derived head and neck cancer cells under the skin of mice. Once the tumors grew, the animals received Anti‑AuND through a vein. Using high‑resolution micro‑CT, the team saw much stronger signals in tumors of mice given gold particles with targeting antibodies than in untreated animals, showing that active targeting outperformed simple passive accumulation. Next, they turned to near‑infrared cameras to check how brightly the tumors glowed. Compared to the free dye alone, Anti‑AuND produced roughly ten times higher fluorescence, creating clear contrast between tumor and normal tissue and sharply outlining tumor boundaries on the skin surface.
Adding a New Dimension with Lifetime Signals
Beyond brightness, the researchers measured how long the dye stayed in its excited state after being illuminated, a property known as fluorescence lifetime. This lifetime is sensitive to the surrounding environment, such as local ion levels, proteins, and oxygen. The Anti‑AuND particles showed a modest lifetime change when bound to gold, but a much larger increase when located inside tumor tissue compared with normal mouse tissue. That shift suggests that lifetime imaging can reveal not only where the particles are but also that they reside in a tumor‑like environment, offering information that is less dependent on dye concentration and more tied to tumor chemistry.
What This Could Mean for Future Care
Together, the CT, fluorescence intensity, and lifetime measurements build a richer picture of head and neck tumors than any single imaging method alone. The gold nanodye acts as a dual‑purpose beacon, brightening near‑infrared signals while also serving as a strong CT contrast agent and a sensitive probe of the tumor microenvironment. Although further work is needed to track long‑term safety and clearance, this multimodal approach could eventually help clinicians detect smaller tumors earlier, guide surgeons to cleaner margins, and monitor how cancers respond to treatment using the very same injected agent.
Citation: Chacko, N., Motiei, M., Rotbaum, R. et al. Antibody-functionalized gold nanospheres for multimodal imaging. Sci Rep 16, 8608 (2026). https://doi.org/10.1038/s41598-026-35561-y
Keywords: gold nanoparticles, near-infrared imaging, head and neck cancer, fluorescence lifetime, multimodal imaging