Clear Sky Science · en
Engrailed 1 promotes immune evasion and chemoresistance in glioma through single cell and CeRNA network analyses
Why this brain tumor study matters
Gliomas, including the deadly glioblastoma, are among the hardest cancers to treat. Even with surgery, radiation, and chemotherapy, most tumors come back, in part because they can hide from the immune system and resist drugs. This study uncovers the role of a little-known gene called Engrailed‑1 (EN1), originally important in brain development, showing how it helps glioma cells dodge immune attack and withstand chemotherapy. Understanding EN1 could open new paths to better diagnose, predict, and treat these stubborn brain tumors.
A developmental gene gone rogue
EN1 is a genetic "switch" that helps shape the developing brain, especially regions that control movement and certain nerve cells. The researchers first asked whether this same gene becomes misused in cancers. By mining large public databases that hold tumor and normal tissue data from thousands of patients, they found that EN1 is abnormally high in many cancers and clearly elevated in gliomas compared with healthy brain tissue. In brain tumor patients, higher EN1 levels went hand in hand with shorter survival times, suggesting EN1 is not just present but linked to more aggressive disease.
Spotlighting EN1 across patients and single cells
To see how useful EN1 might be in the clinic, the team tested whether it could help distinguish tumors from normal tissue and predict patient outcomes. Using statistical models, they showed that EN1 levels help identify several cancer types and, in glioma, add strong predictive power when combined with standard features such as patient age, tumor grade, and common genetic markers. They even built a “risk score” chart, or nomogram, that estimates the chances of surviving one, three, or five years. In a separate group of glioma patients, this tool still worked well, suggesting EN1-based predictions could generalize beyond a single dataset. At the same time, single-cell RNA sequencing—reading out gene activity in thousands of individual tumor and immune cells—revealed that EN1 is especially active in glioma cells with astrocyte-like features and becomes stronger as cells shift toward more advanced, malignant states.
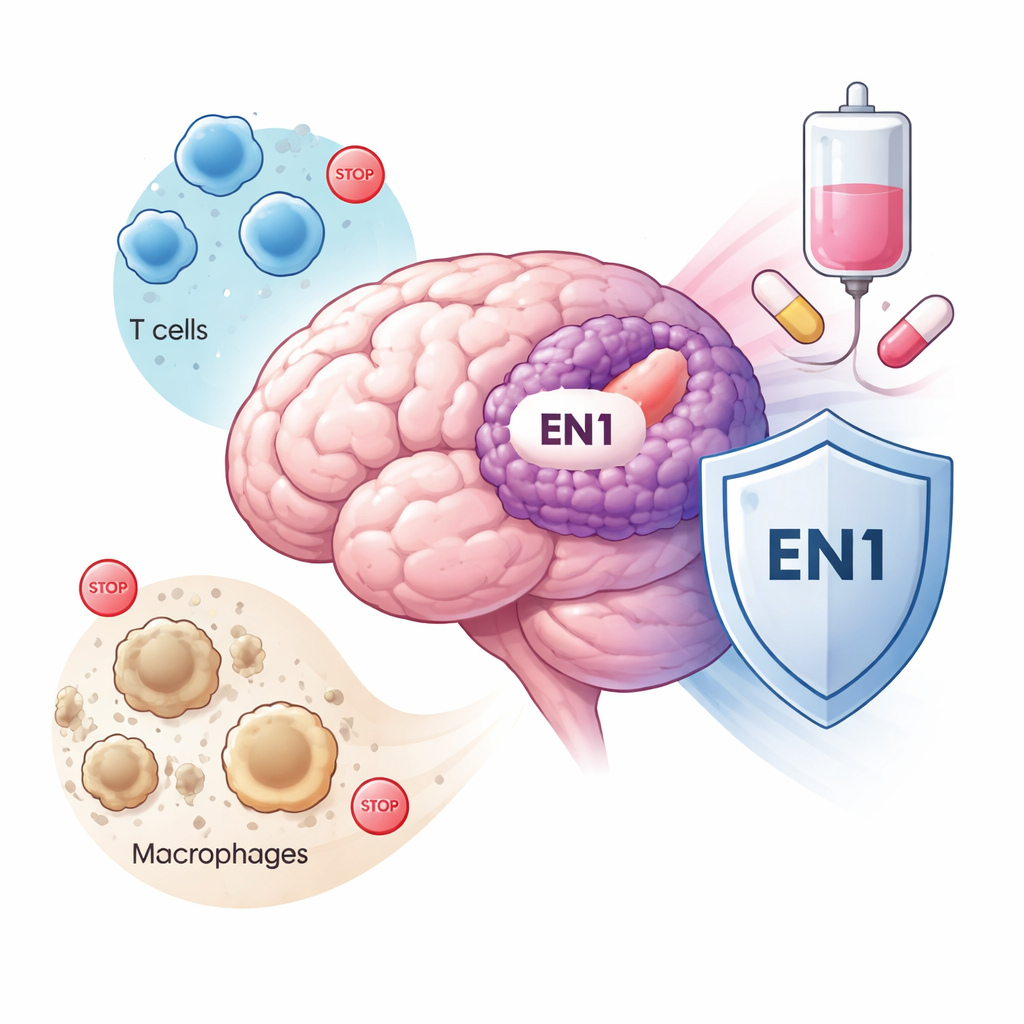
How EN1 shapes the tumor’s neighborhood
Brain tumors do not grow in isolation; they live in a complex neighborhood of immune cells, blood vessels, and support cells. The study found that tumors with high EN1 expression were packed with immune cells that tend to suppress the body’s defenses, particularly a type known as M2 macrophages and regulatory T cells. These cell types usually calm immune reactions and can create a “safe zone” for the tumor. High‑EN1 tumors also showed stronger signals for molecules called immune checkpoints—proteins like PD‑1 and PD‑L1 that act as brakes on immune cells. Together, these patterns paint EN1‑rich tumors as skilled at turning down immune attack while surrounding themselves with supportive, tumor-friendly cells.
Clues to drug resistance and a hidden RNA loop
Standard drugs for glioma, such as temozolomide, often fail over time. Using drug sensitivity prediction tools, the authors found that tumors with high EN1 levels were likely less responsive to several common chemotherapy agents. To understand how EN1 itself is controlled, they probed a regulatory “conversation” among different RNA molecules. They uncovered a loop involving a long RNA called NEAT1 and two small RNAs, miR‑9‑5p and miR‑128‑3p, which normally act as brakes on EN1. NEAT1 appears to sponge up these small RNAs, weakening their ability to hold EN1 in check, while EN1 may in turn boost NEAT1, forming a self-reinforcing circuit that keeps EN1 high and the tumor aggressive.
Putting EN1 to the test in the lab
Bioinformatic results can be suggestive, but the team also tested EN1 directly in glioblastoma cell lines grown in the lab. They showed that tumor cells carry much more EN1 than normal brain support cells. When they used genetic tools to reduce EN1 levels, glioblastoma cells slowed their growth and became less able to invade through a barrier that mimics tissue. These changes support the idea that EN1 actively drives tumor expansion and spread, rather than merely being a bystander marker.
What this means for patients
In plain terms, this work positions EN1 as both a warning light and a possible weak point in glioma. High EN1 levels signal more aggressive tumors that better evade immune defenses and are harder to treat with current drugs. At the same time, because EN1 sits at the center of a regulatory loop and influences the tumor’s immune surroundings, it offers several new angles for therapy—from directly blocking EN1, to disrupting its RNA network, to pairing such strategies with immune checkpoint drugs. While more detailed experiments and clinical studies are needed, EN1 now stands out as a promising target in the ongoing effort to make brain tumors more treatable.
Citation: Jia, Z., Wang, Y., Yao, J. et al. Engrailed 1 promotes immune evasion and chemoresistance in glioma through single cell and CeRNA network analyses. Sci Rep 16, 4913 (2026). https://doi.org/10.1038/s41598-026-35553-y
Keywords: glioma, immune evasion, chemoresistance, Engrailed-1, brain tumor