Clear Sky Science · en
Co3O4 nanoneedles grown on graphene oxide as an efficient electrocatalyst for hybrid water electrolysis through alternative anodic oxidation reactions
Turning Fruit Waste into Future Fuel
Hydrogen is often hailed as a clean fuel of the future, but making it typically demands a lot of electricity. This study shows how something as ordinary as discarded orange peels can help change that. By transforming this waste into a special carbon material and combining it with a cobalt-based compound, the researchers created a low-cost catalyst that can produce hydrogen using much less energy than conventional water-splitting methods. Along the way, they also replace an inefficient step in water electrolysis with gentler reactions that turn problem chemicals into harmless gases.
Why Traditional Water Splitting Wastes Energy
To split water into hydrogen and oxygen, an electrolyzer pushes electrical current through water containing a dissolved salt or base. At one side, hydrogen gas is formed easily. At the other side, oxygen is produced in a reaction that is slow and demanding, because it must move four electrons in tightly choreographed steps. This oxygen-making step, called the anodic reaction, is what forces engineers to push the voltage higher, driving up energy costs. Worse, the oxygen itself is often just vented and not used, which means a big share of the electrical power fed into the system produces little practical value.
Swapping an Energy Hog for Gentler Reactions
The team tackled this bottleneck by redesigning what happens at the energy-hungry side of the cell. Instead of making oxygen from water, they asked: what if the system oxidized other, easier-to-handle chemicals while still making hydrogen at the opposite side? They chose two nitrogen-rich compounds, urea and hydrazine, which are common in wastewater streams and industrial processes. When these molecules are oxidized in alkaline solution, they break down into nitrogen gas, water, and in the case of urea, carbon dioxide. Crucially, these reactions start at much lower voltages than oxygen production does, meaning the same amount of hydrogen can be generated with far less electrical input.
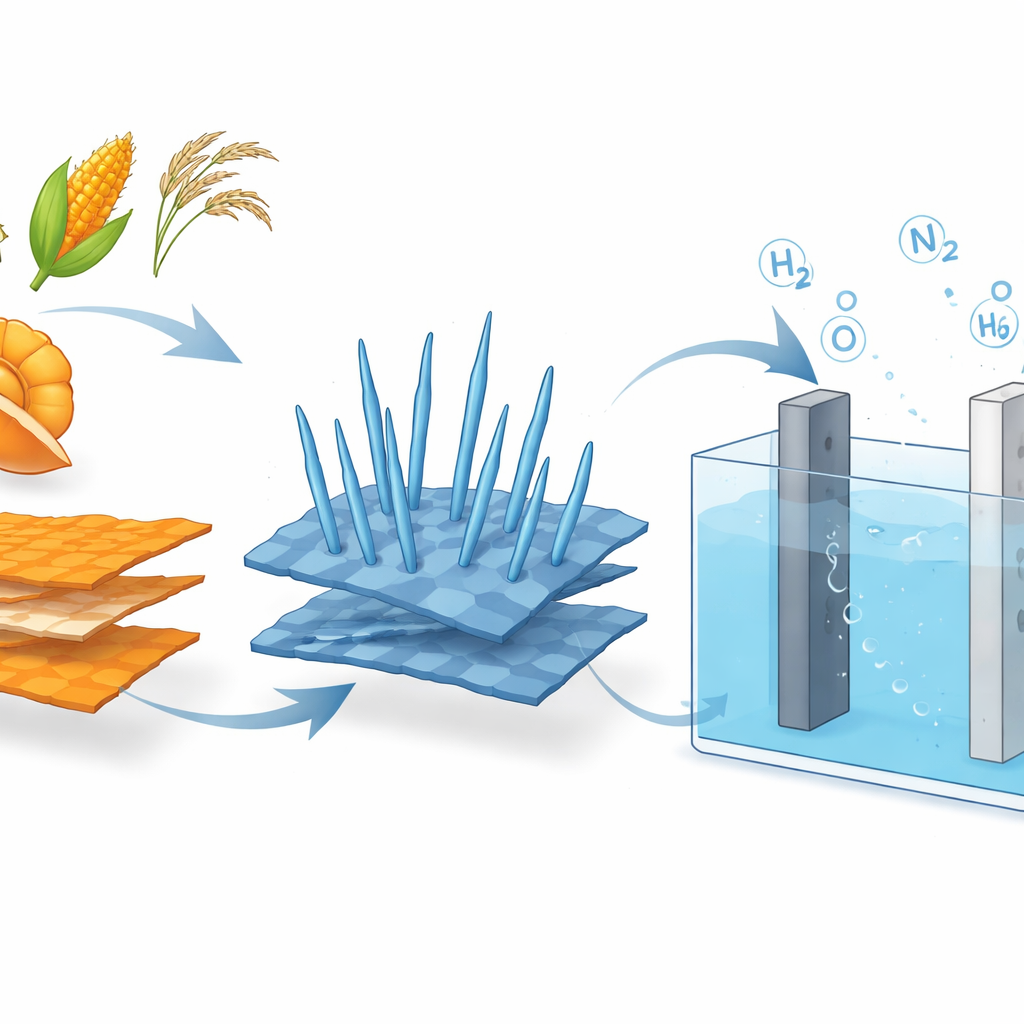
From Orange Peels to Smart Electrodes
To make this approach practical, the researchers needed a catalyst that was cheap, robust, and active for three different tasks: conventional oxygen production, urea oxidation, and hydrazine oxidation. They began by converting dried orange peels into graphene oxide, a thin, conductive carbon material, using a simple heating process rather than harsh chemical treatments. Onto these sheets they grew tiny "nanoneedles" of cobalt oxide in a pressure vessel. The resulting hybrid—cobalt oxide nanoneedles on graphene oxide—forms a rough, sponge-like surface with many exposed reactive sites and an easy pathway for electrons to travel. Measurements showed that the graphene support prevents cobalt particles from clumping and dramatically boosts the effective surface area and electrical conductivity.
How the New Catalyst Cuts the Power Bill
When tested in alkaline solution, the new electrode reached a standard benchmark current at significantly lower voltages than bare cobalt oxide. For conventional oxygen production, it performed comparably to some commercial noble-metal catalysts. When urea was added, the required voltage dropped further, and with hydrazine, the improvement was striking: the electrode needed only a tiny extra push above the natural reference level to sustain the same current. In a full two-electrode cell paired with a standard platinum-based hydrogen-making electrode, hydrazine-assisted electrolysis produced hydrogen at just 0.33 volts—about 1.3 volts lower than traditional water splitting under the same conditions. The system also remained stable over many hours, with the structure and composition of the catalyst essentially unchanged.
What This Means for Clean Hydrogen
To a non-specialist, the takeaway is simple: by rethinking both the material of the electrode and the reaction that happens at it, the researchers showed that hydrogen can be produced using much less electricity and with inexpensive ingredients. Fruit waste becomes a high-performance carbon scaffold; cobalt oxide nanoneedles provide active sites; and replacing oxygen formation with the oxidation of urea or hydrazine slashes the voltage needed. In the hydrazine case, the by-products are mainly nitrogen and water, avoiding extra carbon emissions. While further work is needed to manage chemical supplies and safety at scale, this hybrid electrolysis strategy points toward cleaner, cheaper hydrogen production that also valorizes waste streams and renewable biomass.
Citation: Rahamathulla, N., Murthy, A.P. Co3O4 nanoneedles grown on graphene oxide as an efficient electrocatalyst for hybrid water electrolysis through alternative anodic oxidation reactions. Sci Rep 16, 8452 (2026). https://doi.org/10.1038/s41598-026-35522-5
Keywords: hydrogen production, water electrolysis, graphene oxide, hydrazine oxidation, biomass-derived catalysts