Clear Sky Science · en
Plasma exosomal hsa-miR-339-5p is associated with NOD-like receptor family CARD domain-containing 5 in heart failure with reduced ejection fraction
Why tiny molecules in blood may reveal a struggling heart
Heart failure with reduced ejection fraction (HFrEF) is a common condition in which the heart can no longer pump enough blood to meet the body’s needs. Doctors usually discover it only after damage is already substantial. This study explores whether microscopic messages circulating in the blood—small RNA molecules packed into tiny bubbles called exosomes—can flag trouble earlier and shed light on how the heart slowly stiffens and scars over time.
Hidden messages riding in the bloodstream
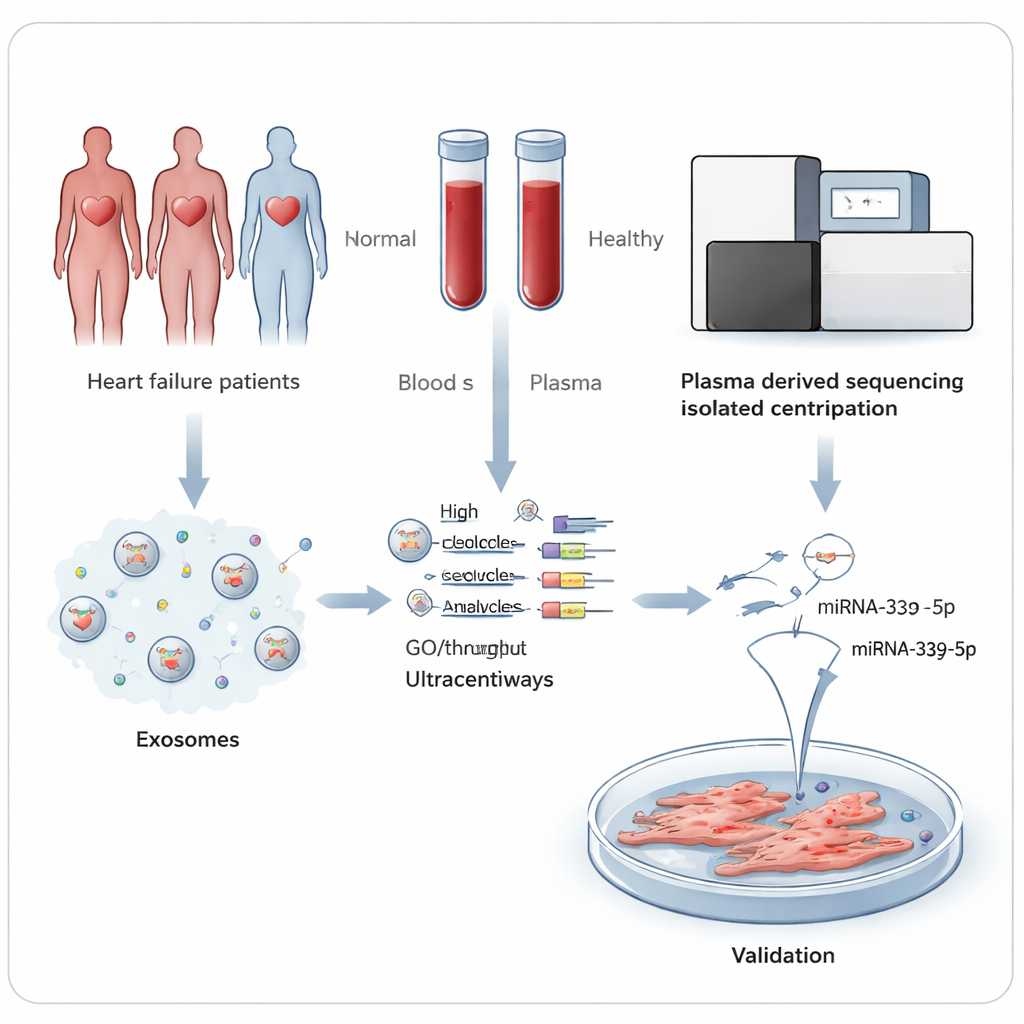
Our blood carries countless exosomes, nano‑sized sacs released by cells that ferry proteins and genetic material from one cell to another. Among their cargo are microRNAs, short strands that fine‑tune which genes are turned on or off. Because exosomes protect these microRNAs from breaking down, they can be measured reliably in a simple blood sample. The researchers reasoned that if the failing heart or related tissues release a different mix of microRNAs than a healthy heart, those patterns could serve as warning signs and also reveal processes driving the disease.
Comparing patients with weakened hearts to healthy volunteers
The team collected blood from 45 people with HFrEF and 45 similar but healthy volunteers at a hospital in Hefei, China. From the blood plasma, they carefully isolated exosomes and confirmed their size and shape using electron microscopy and particle‑tracking instruments. In a first step, they sequenced the small RNAs inside exosomes from a subset of five patients and five controls. This high‑throughput approach detected hundreds of known microRNAs and highlighted 27 that were present at clearly different levels between the two groups—10 higher and 17 lower in people with heart failure.
Narrowing in on one standout signal
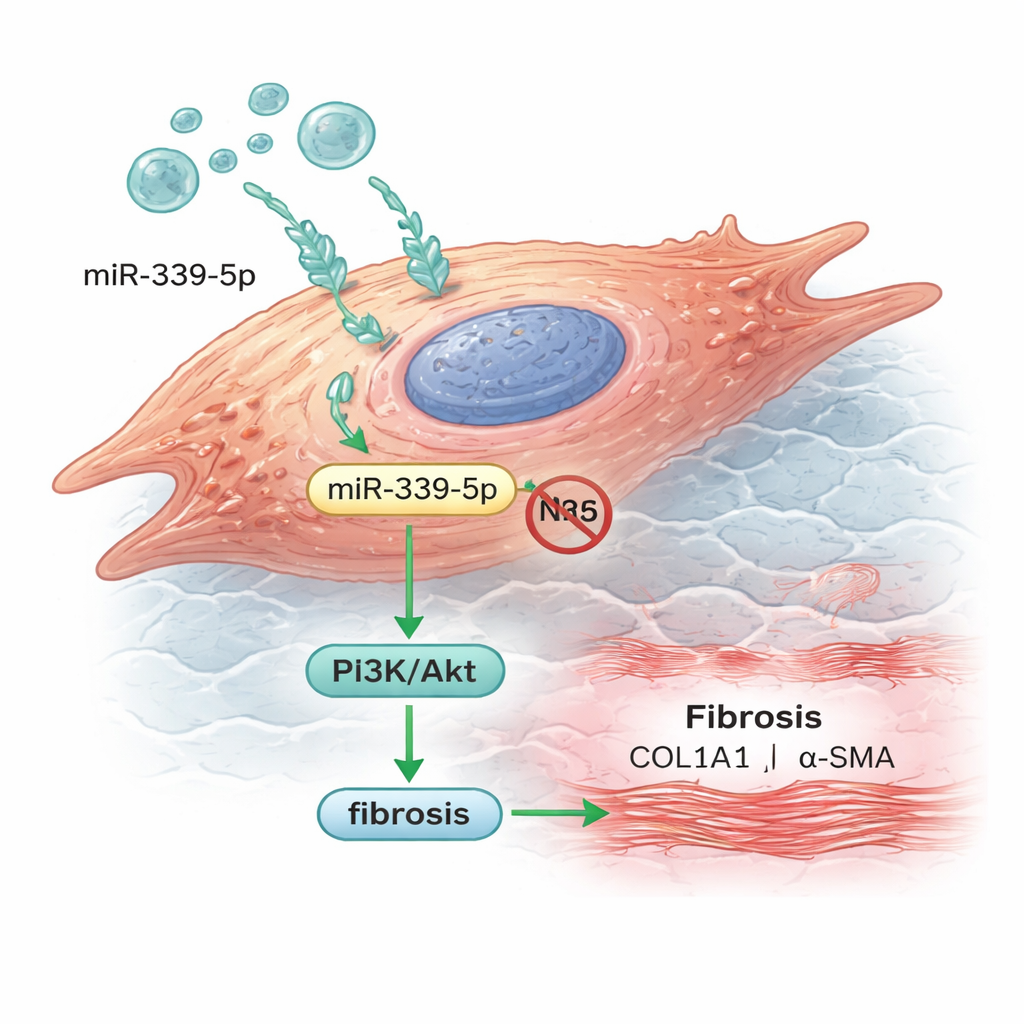
To test which of these changes were most reliable, the scientists next used a highly sensitive PCR method to measure six promising microRNAs in all 40 remaining patients and 40 controls. Several signals held up, but one in particular—called hsa‑miR‑339‑5p—stood out. It was consistently higher in patients with HFrEF and, when analyzed as a diagnostic marker, best distinguished them from healthy individuals. Computer‑based pathway analyses suggested that this microRNA might act on a gene named NLRC5, which in turn influences a central growth and survival route inside cells known as the PI3K/Akt pathway, previously linked to heart muscle thickening and scarring.
From blood marker to driver of scarring
The researchers then moved to heart‑like cells grown in the lab to see how this microRNA behaves inside tissue. They showed that exosomes from patients were readily taken up by human cardiomyocytes, delivering miR‑339‑5p into the cells. When they artificially boosted miR‑339‑5p, levels of NLRC5 dropped and the PI3K/Akt pathway became more active. At the same time, the cells produced more collagen and a protein called alpha‑smooth muscle actin—both hallmarks of fibrotic, stiff tissue rather than flexible, healthy muscle. When miR‑339‑5p was blocked, these changes were reversed, suggesting that this single microRNA helps push heart cells toward a scar‑forming state.
What this means for patients and future care
By mapping this chain of events—from elevated miR‑339‑5p in blood exosomes to reduced NLRC5, activated PI3K/Akt signaling, and increased scarring proteins—the study links a measurable blood marker to a plausible mechanism of damage inside the heart. For patients, this raises the possibility that a routine blood test could one day help detect harmful remodeling earlier than current scans and lab markers allow. It also hints that drugs designed to dial down miR‑339‑5p, or restore NLRC5 activity, might slow the stiffening and scarring that make heart failure progressively worse. While larger and longer‑term studies are still needed, these tiny RNA messengers may offer both a warning light on the dashboard and a new target under the hood.
Citation: Cheng, D., Hu, J., Zhao, M. et al. Plasma exosomal hsa-miR-339-5p is associated with NOD-like receptor family CARD domain-containing 5 in heart failure with reduced ejection fraction. Sci Rep 16, 5690 (2026). https://doi.org/10.1038/s41598-026-35519-0
Keywords: heart failure, exosomes, microRNA, fibrosis, biomarkers