Clear Sky Science · en
Structural and computational supported development of 2,5-disubstituted-1,3,4-oxadiazole analogues as active LOX, urease, and α-glucosidase inhibitors
New Chemical Tools to Tackle Common Diseases
Many everyday health problems—from ulcers and kidney stones to diabetes and chronic inflammation—are driven by overactive enzymes in our bodies. This study explores a family of newly designed small molecules that act like tiny brakes for three such enzymes. By tuning their structures and testing them in the lab and on computers, the researchers aim to lay groundwork for more effective and safer medicines in the future. 
Why These Enzymes Matter
The team focused on three enzyme targets that play very different but equally important roles in health. Urease helps break down urea; when it becomes overactive in certain bacteria or tissues, it can contribute to stomach and urinary tract ulcers, kidney stones, and even higher blood pressure. Alpha-glucosidase sits on the surface of intestinal cells and chops complex carbohydrates into glucose; blocking it is a well-established way to help control blood sugar in type 2 diabetes. Lipoxygenase (LOX) converts fats into signaling molecules that drive inflammation, asthma, and some cancer-related processes. Drugs that dial down these enzymes without harming other systems are therefore highly sought after.
Building a Library of Candidate Molecules
To search for such drugs, the researchers built a series of 15 related compounds based on a ring system called 1,3,4-oxadiazole linked to a piperidine unit. This chemical scaffold already appears in several modern medicines and is known to interact well with biological targets. The team varied the “decorations” attached to this core—small groups such as methyl, ethyl, methoxy, benzyl, and cyclohexyl—at different positions, creating a small library of candidates labeled 7a through 7o. They confirmed each structure using standard analytical tools like infrared and nuclear magnetic resonance spectroscopy, ensuring that what they had made matched their designs.
Testing How Well the Molecules Block Enzymes
Each compound was then tested against purified LOX, urease, and alpha-glucosidase to see how strongly it could slow the enzyme’s activity. Several molecules stood out. Against alpha-glucosidase, compounds 7a and 7n were particularly effective, outperforming the standard diabetes drug acarbose in the lab tests by achieving high percentages of inhibition at low micromolar concentrations. For LOX, compounds 7a, 7h, and 7n showed very strong activity, in some cases better than the natural reference inhibitor quercetin. In the urease assays, compounds 7a and especially 7l rivaled or slightly surpassed the standard inhibitor thiourea, suggesting they might one day serve as leads for anti-ulcer or anti-stone therapies. 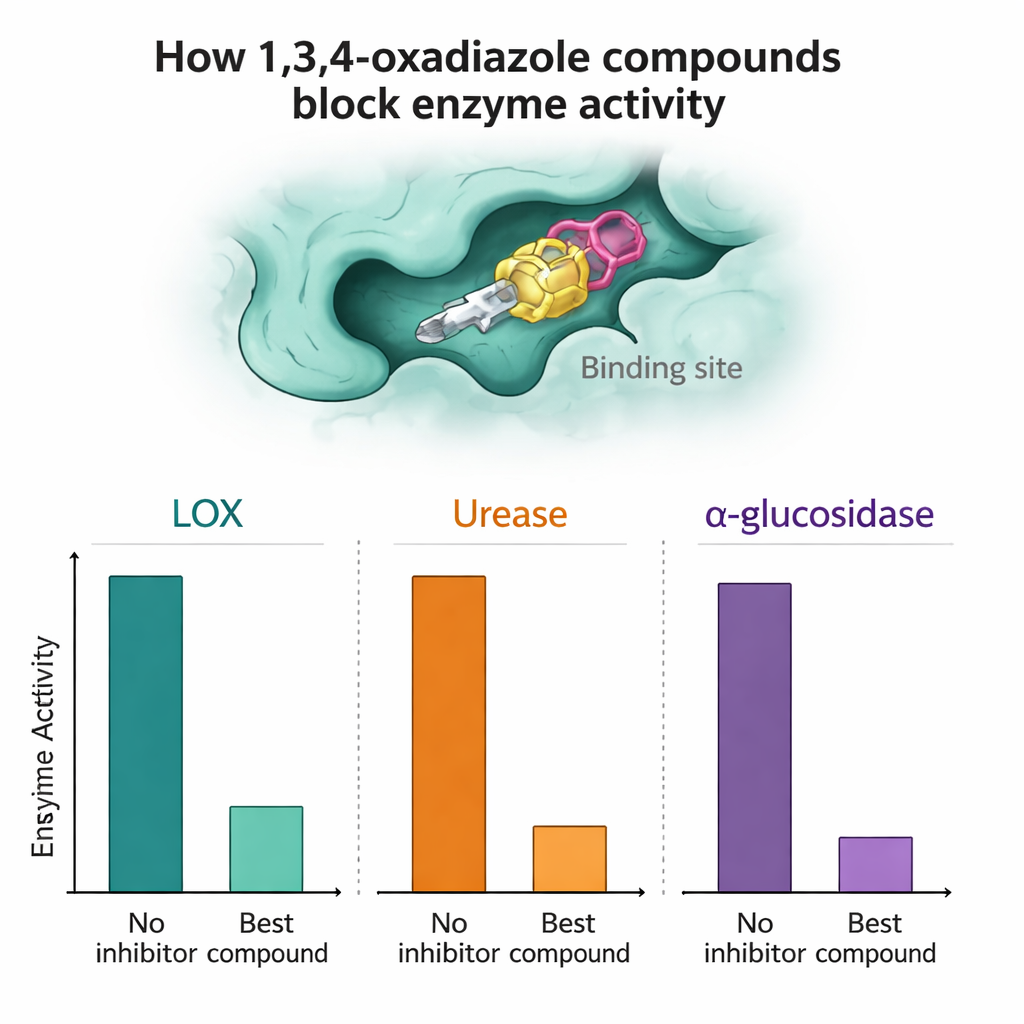
Linking Structure to Performance
Because the only differences between compounds 7a–7o are small changes around the core ring system, the researchers could begin to map out simple structure–activity relationships. For instance, adding small carbon-rich groups at certain “ortho” positions on an attached ring often boosted the ability to block alpha-glucosidase or LOX. Other substitutions, such as certain methoxy or ester groups placed less favorably, tended to weaken activity. To understand why, the team used computer simulations. Quantum-chemical calculations showed that all molecules were thermodynamically stable and flexible enough to adapt to enzyme pockets. Docking studies—virtual fitting of each molecule into three-dimensional models of the enzymes—revealed that the most active compounds made tighter contact through a mix of hydrogen bonds and hydrophobic (oil-like) interactions at key spots in the active sites, whereas weaker candidates sat less snugly or missed critical contact points.
What This Means for Future Medicines
In everyday terms, the study identifies a handful of promising “keys” that fit well into three disease-related “locks” and begins to explain, at the atomic level, why some keys turn better than others. None of these molecules is ready to be a drug yet—they still need safety testing, optimization for behavior in the body, and trials in animals and humans. But the work shows that the 1,3,4-oxadiazole–piperidine framework is a fertile starting point for future treatments targeting diabetes, inflammatory conditions, and urease-related disorders. The combination of careful lab measurements with detailed computer modeling provides a roadmap for sharpening these candidates into more precise and effective medicines.
Citation: Javid, J., Aziz-ur-Rehman, Iqbal, J. et al. Structural and computational supported development of 2,5-disubstituted-1,3,4-oxadiazole analogues as active LOX, urease, and α-glucosidase inhibitors. Sci Rep 16, 5866 (2026). https://doi.org/10.1038/s41598-026-35499-1
Keywords: enzyme inhibitors, drug discovery, oxadiazole compounds, diabetes and inflammation, urease and lipoxygenase