Clear Sky Science · en
KidneyTox_v1.0 enables explainable artificial intelligence prediction of nephrotoxicity in small molecules
Why protecting kidneys from medicines matters
Many life‑saving medicines can quietly damage the kidneys, sometimes leading to serious illness that only becomes visible when it is too late. Doctors and drug developers need ways to spot this risk early, before a new pill ever reaches patients. This article describes KidneyTox_v1.0, a free online tool that uses explainable artificial intelligence to predict whether a small‑molecule drug is likely to harm the kidneys—and, crucially, shows users why it reaches that conclusion.
From scattered data to a big‑picture view
The researchers began by assembling a carefully curated collection of 565 approved or experimental drugs. Roughly half of these have been reported to injure the kidneys in people, while the rest have no known kidney toxicity. Instead of treating these compounds as a simple list, the team first mapped out their “chemical neighborhood”—basic properties such as size, weight, fat‑loving versus water‑loving tendencies, number of ring structures, and how many bonds in the molecule can twist and turn. They found that the set spans a very wide range: from tiny, highly water‑soluble molecules to large, flexible structures with many rings. This diversity is important; it means the tool is not limited to one narrow type of drug chemistry.
Teaching a computer to flag risky molecules
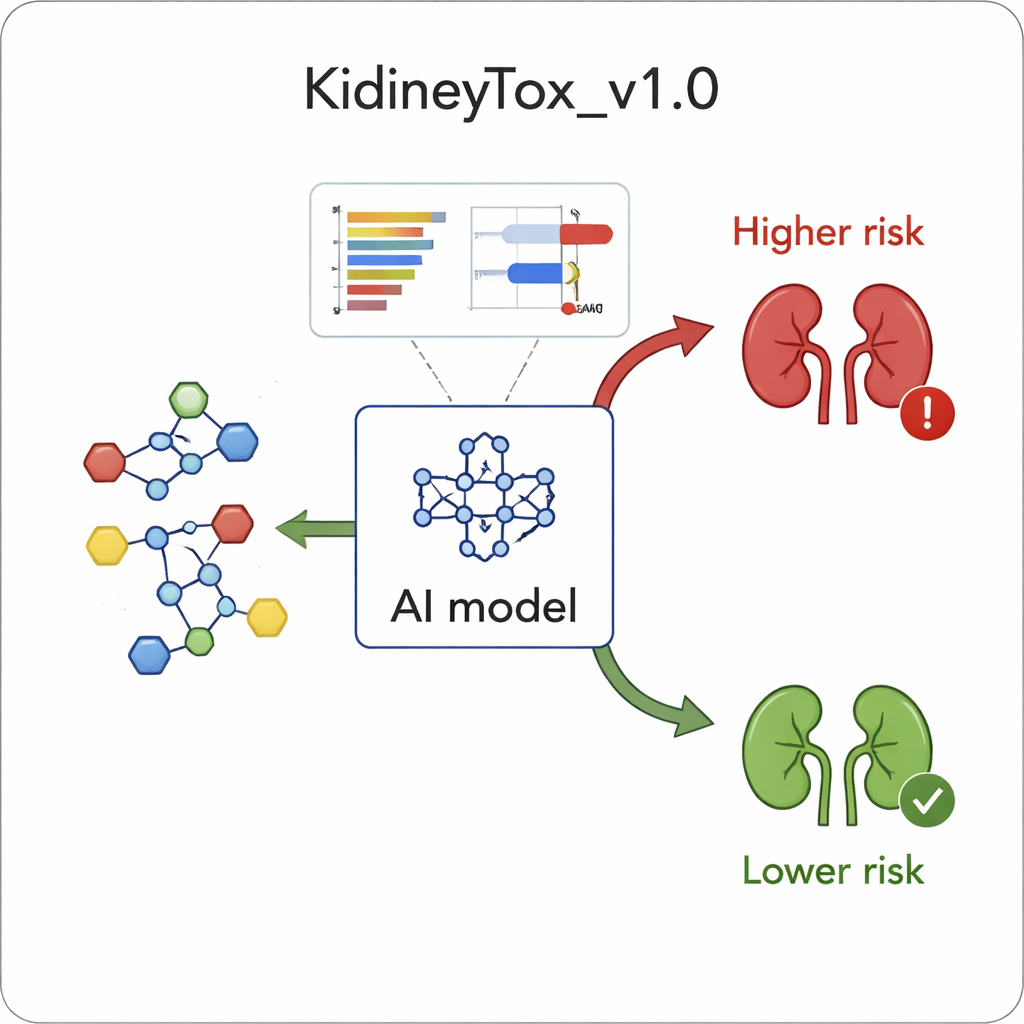
Using this diverse dataset, the team trained a machine‑learning model—a computer program that learns patterns from examples—to distinguish kidney‑toxic drugs from safer ones. The model, based on a method called a random forest, examines many numerical descriptors that capture a molecule’s shape, charge distribution, and other features. After carefully tuning the model and selecting the most informative descriptors, the system correctly classified about 84% of the unseen test compounds. To ensure this was not a fluke, the authors tried multiple different train–test splits, finding that their chosen model consistently performed among the best, suggesting that it had learned general rules rather than memorizing the data.
Opening the “black box” with visual explanations
A common criticism of AI in medicine is that it often acts like a black box: it may predict that a drug is dangerous, but cannot say why. To counter this, the authors built explainability directly into KidneyTox_v1.0. They used a technique called SHAP, which assigns each descriptor a positive or negative contribution to the final prediction for any given molecule. In practical terms, users see a waterfall plot where red bars push the prediction toward “toxic” and blue bars push toward “non‑toxic.” For example, higher values of certain electronegativity‑related features tended to nudge predictions toward kidney harm, while other features linked to overall molecular flexibility or polarizability often supported a safer profile. Case studies with well‑known drugs like Lansoprazole and Ciprofloxacin, both associated with kidney problems, showed how specific structural traits drive the model’s warning signal, whereas relatively safer drugs show the opposite pattern.
Blending similarity reasoning with AI insight
Beyond the main model, the study also developed so‑called qRASAR models, which combine machine‑learning descriptors with “read‑across” ideas long used in toxicology. Here, a drug’s risk is inferred partly from how similar it is to known toxic or non‑toxic neighbors, and how consistent the surrounding data are. Remarkably, one simplified model based on just three such similarity‑and‑error features still performed well, striking a balance between accuracy and transparency. This means regulators and medicinal chemists can see not only that a compound resembles known kidney‑damaging drugs, but also how reliable that analogy is, given the data in its neighborhood.
A practical tool for designing safer medicines
All of these elements come together in KidneyTox_v1.0, a browser‑based platform built with a user‑friendly interface. A chemist can draw a new molecule or paste its standard text code (a SMILES string) into the tool, and within moments receive a “toxic” or “non‑toxic” prediction, a confidence assessment based on how similar the molecule is to the training set, and side‑by‑side plots comparing it with its closest known neighbor. Because the underlying data and code are openly shared, the platform can be improved and expanded as new kidney toxicity information emerges, and companies can test proprietary compounds without sending structures to a remote server for storage.
What this means for patients and future drugs
In plain terms, this work shows that we can now use explainable AI to flag drug candidates with a higher chance of injuring the kidneys, long before they reach clinical trials or the pharmacy shelf. By revealing which molecular features are most closely linked to kidney damage, KidneyTox_v1.0 can guide chemists toward safer design choices—tweaking polarity, ring systems, or charge distribution to lower risk while preserving benefit. Although the current model is built on a few hundred compounds and will improve with more data, it already represents a practical step toward faster, cheaper, and more humane safety testing, with the ultimate aim of protecting patients from avoidable kidney harm.
Citation: Amin, S.A., Kar, S. & Piotto, S. KidneyTox_v1.0 enables explainable artificial intelligence prediction of nephrotoxicity in small molecules. Sci Rep 16, 5099 (2026). https://doi.org/10.1038/s41598-026-35496-4
Keywords: kidney toxicity, drug safety, artificial intelligence, machine learning, cheminformatics