Clear Sky Science · en
SHP2 promotes osteosarcoma via regulating STAT3/TET3/HOXB2 signaling
Why this bone cancer study matters
Osteosarcoma is the most common primary bone cancer in children and teenagers, and despite modern chemotherapy and surgery, survival for patients with advanced or relapsed disease has barely improved in decades. This study digs into the inner wiring of osteosarcoma cells to find out which molecules act like "master switches" that help tumors grow, spread, and resist treatment. By uncovering a key control pathway centered on a protein called SHP2, the work points to new drug targets that could one day make osteosarcoma therapies more effective and more precise.
Following the clues in tumor gene activity
The researchers began by treating osteosarcoma as a data problem. They mined public gene expression datasets comparing tumor samples with normal bone tissue, then used statistical tools to spot genes that were consistently turned up or down in cancer. Hundreds of genes differed, and many of them clustered in well-known cancer pathways that drive cell growth, survival, and movement. Among these, one protein, SHP2, stood out as a hub strongly linked to several others: the signaling factor STAT3, the DNA-modifying enzyme TET3, and the developmental regulator HOXB2. This suggested that these molecules might form a connected chain that helps osteosarcoma cells behave aggressively. 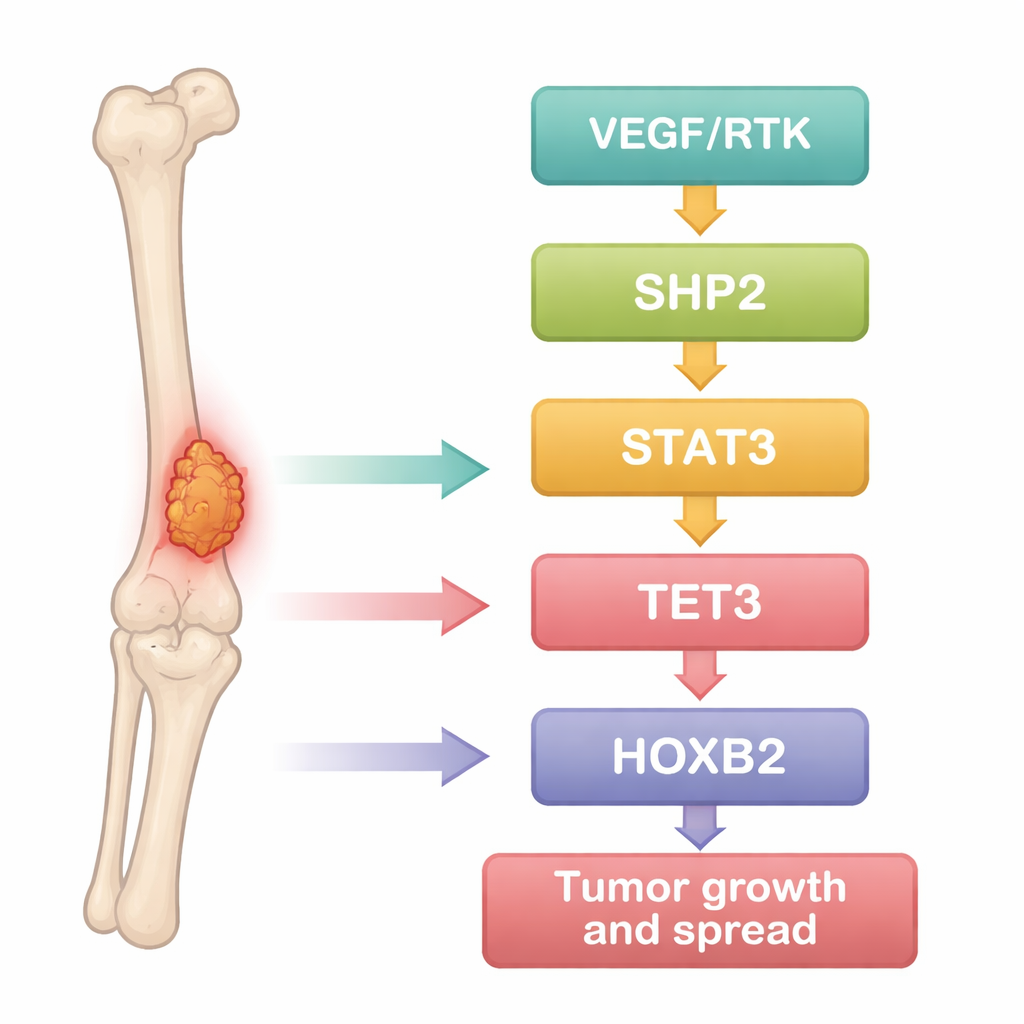
Testing SHP2’s role in cancer cells
To move beyond correlations, the team engineered human osteosarcoma cell lines (143B and MG63) to either reduce or boost SHP2. When they knocked SHP2 down, the cells’ ability to multiply, migrate across a surface, and invade through a membrane all dropped noticeably. The levels of STAT3, TET3, and HOXB2 proteins, as well as cancer-linked markers such as c-Myc, NANOG, and NUSAP1, also fell. When they reactivated STAT3 or overexpressed HOXB2, many of these effects were reversed, indicating that SHP2 sits upstream of a chain that ultimately feeds into genes controlling cell division, stem cell–like traits, and the machinery of cell division.
Building a step‑by‑step cancer pathway
Piecing the experiments together, the authors outline a stepwise model. In the tumor environment, high levels of vascular endothelial growth factor (VEGF) stimulate receptor tyrosine kinases (RTKs) on the cell surface. These, in turn, activate SHP2 inside the cell. Active SHP2 switches on STAT3, a transcription factor that moves to the nucleus and helps increase production of TET3. TET3 then edits the chemical tags on DNA, specifically removing methyl groups from the HOXB2 gene region. This demethylation acts like lifting a brake, allowing HOXB2 to be more strongly expressed. HOXB2, a gene that normally guides development, now boosts the activity of c-Myc, NANOG, and NUSAP1, which together fuel relentless tumor cell growth, enhance invasive behavior, and support features associated with metastasis.
From Petri dish to living animals
To see whether this pathway mattered in whole tumors, the scientists implanted modified osteosarcoma cells under the skin of mice. Tumors formed from cells overproducing SHP2 grew larger and heavier than control tumors. When TET3 was silenced in these SHP2‑rich cells, tumor growth slowed and the levels of HOXB2 dropped, even though SHP2 and STAT3 remained active. Reintroducing HOXB2 restored the tumor’s vigor. Throughout, measurements of protein levels in tumor tissue mirrored the earlier cell-culture findings, strengthening the idea that SHP2, STAT3, TET3, and HOXB2 act as a connected axis that pushes osteosarcoma toward more malignant behavior. 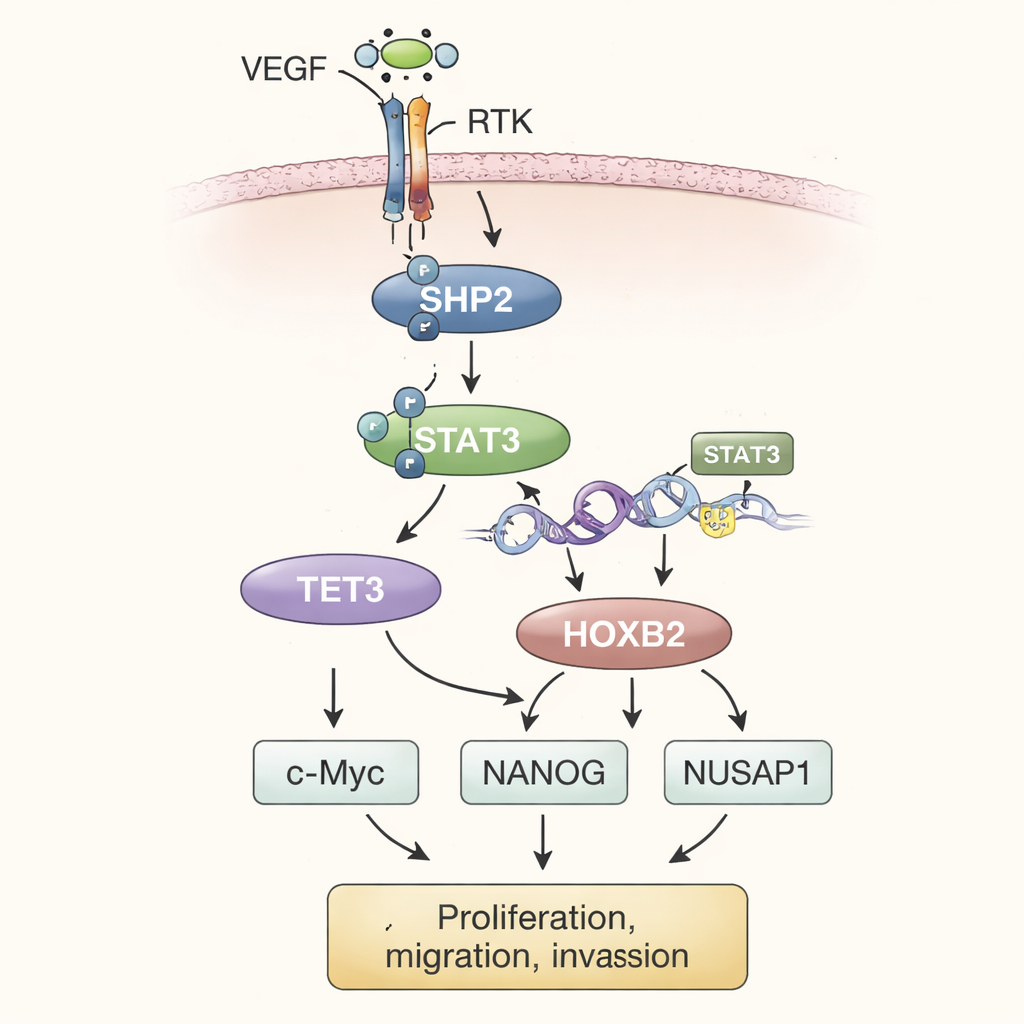
What this means for future treatments
For a lay reader, the take‑home message is that the researchers have traced a clear signal chain—from VEGF and its surface receptors, through SHP2 and STAT3, down to TET3, HOXB2, and several powerful growth genes—that helps explain why osteosarcoma can grow fast and spread early. Because SHP2 and STAT3 are enzymes that can, in principle, be blocked by small‑molecule drugs, and TET3 and HOXB2 mark later steps in the same route, this pathway offers multiple points where future therapies could intervene. While this study was done in cell lines and mice, it provides a detailed map that drug developers can use to design combination treatments aimed at shutting off the tumor’s central control switches rather than just attacking its end results.
Citation: Yang, H., Ji, J. SHP2 promotes osteosarcoma via regulating STAT3/TET3/HOXB2 signaling. Sci Rep 16, 6158 (2026). https://doi.org/10.1038/s41598-026-35493-7
Keywords: osteosarcoma, SHP2, STAT3 pathway, tumor signaling, targeted therapy