Clear Sky Science · en
Noninvasive PET imaging of LPS-induced oxidative stress in skeletal muscle using a ROS-targeting radiotracer
Why stressed muscles matter
As we age, fight infections, or spend time immobilized in a hospital bed, our muscles can shrink and weaken. Much of this hidden damage starts long before muscle size visibly changes, driven by unstable molecules called reactive oxygen species, or ROS. This study explores a new way to "see" that invisible chemical stress inside muscles using a medical scan called PET, potentially allowing doctors to catch trouble early—before muscle loss becomes hard to reverse. 
A closer look at invisible muscle stress
Skeletal muscle atrophy is the gradual loss of muscle mass and strength that can follow aging, severe illness, inactivity, or treatments like chemotherapy. Traditionally, doctors have relied on tools such as biopsies, MRI, or CT scans to evaluate muscle health. These approaches reveal changes in size and structure but cannot show the early chemical signals that set damage in motion. One of the earliest and most important of these signals is oxidative stress—an overload of ROS that upsets the cell’s normal chemical balance and activates pathways that break down muscle proteins. Being able to measure this stress directly inside living muscle could help identify people at risk and track whether treatments aimed at protecting muscle are working.
A tracer that lights up harmful molecules
The researchers focused on a specialized PET imaging tracer called [18F]ROStrace, designed to home in on superoxide, a major type of ROS. After confirming that the tracer could be produced reliably and remained stable for several hours, they tested whether it truly responded to oxidative stress in muscle cells grown in the lab. Mouse muscle cells (C2C12 myotubes) exposed to bacterial toxin LPS, which triggers inflammation and ROS, accumulated more [18F]ROStrace over time than untreated cells. When the cells were protected with an antioxidant drug, tracer uptake dropped; when ROS were boosted with a separate compound, uptake rose. These patterns showed that the tracer’s signal increased and decreased in step with ROS levels rather than with general changes in cell shape or metabolism. 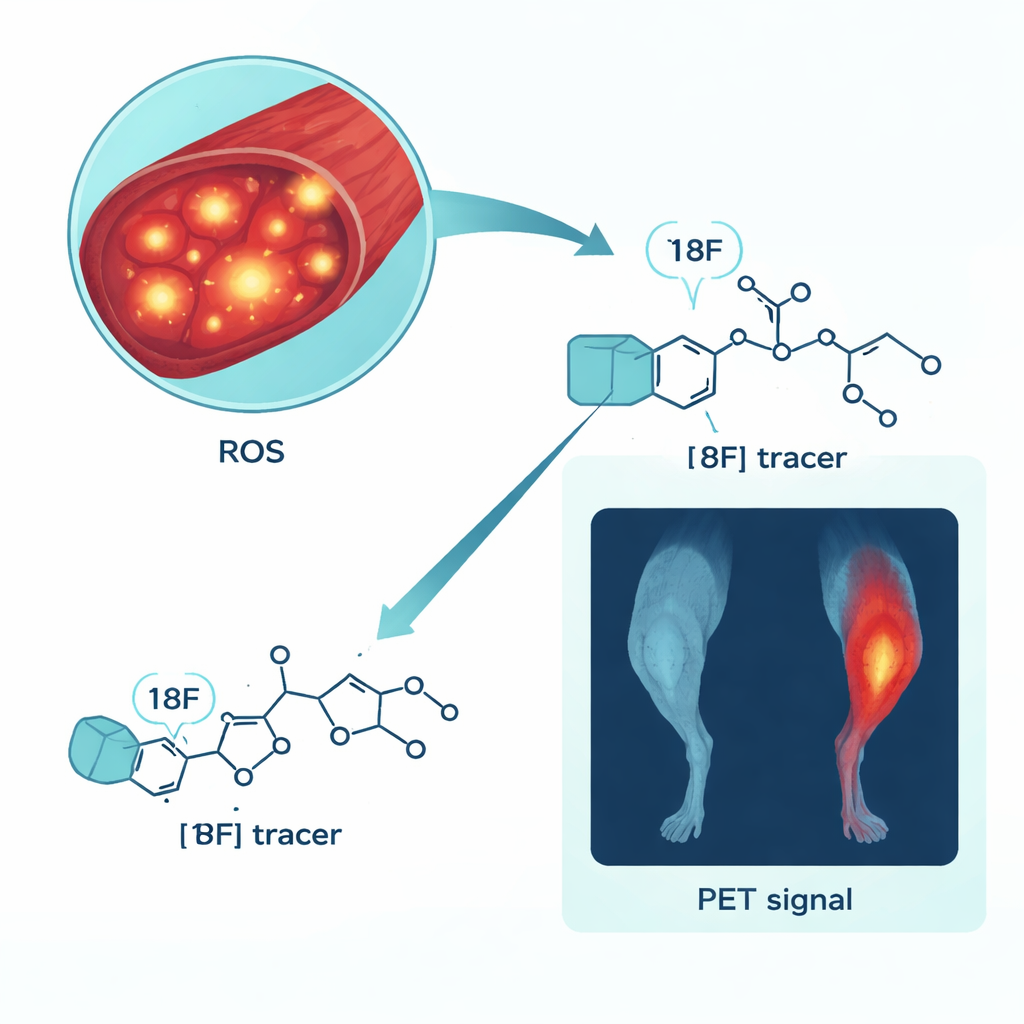
From dishes to living muscles
Next, the team turned to mice to see whether [18F]ROStrace could reveal oxidative stress in real muscle tissue. Mice were injected with LPS to create a whole-body inflammatory state that causes early signs of muscle wasting. Using PET/CT, the scientists scanned the animals about a day later and measured tracer uptake in the hindlimb muscles. Compared with healthy control mice, the LPS-treated group showed roughly double the PET signal in these muscles, indicating a substantially higher oxidative burden. Time-course scans revealed that tracer levels in muscle reached a steady plateau around one hour after injection, defining a practical imaging window for future studies and possible clinical translation.
Biological clues behind the bright signal
To confirm that the PET images truly reflected harmful chemistry rather than blood flow or other nonspecific effects, the researchers analyzed the same muscles using several standard laboratory methods. Under the microscope, LPS-treated muscles and cultured myotubes appeared thinner, and individual fibers had a smaller cross-sectional area—features consistent with the early stages of atrophy. Mitochondria, the cell’s power plants, showed reduced membrane potential, a sign of disturbed internal balance under oxidative stress. Key genes that drive muscle breakdown, MuRF-1 and Atrogin-1, were sharply elevated in LPS-treated cells and tissues. Finally, staining with a fluorescent dye closely related to the [18F]ROStrace chemistry confirmed that ROS levels were indeed higher in muscles showing stronger PET signals, both in cell cultures and in living mice.
What this could mean for patients
Taken together, the findings show that [18F]ROStrace PET can noninvasively highlight pockets of oxidative stress in skeletal muscle and that the brightness of the signal tracks with other hallmarks of early muscle damage. For everyday patients, this approach could one day offer a way to detect muscle stress before substantial wasting occurs, to monitor how well new drugs or exercise programs are calming harmful chemistry, and to better understand conditions ranging from sepsis-related weakness to age-related frailty. While more work is needed in long-term and human studies, this ROS-targeted imaging tool opens a new window on how and when muscles begin to fail—and how we might intervene sooner.
Citation: Park, J.Y., Park, S.M., Lee, T.S. et al. Noninvasive PET imaging of LPS-induced oxidative stress in skeletal muscle using a ROS-targeting radiotracer. Sci Rep 16, 4917 (2026). https://doi.org/10.1038/s41598-026-35489-3
Keywords: muscle atrophy, oxidative stress, PET imaging, reactive oxygen species, [18F]ROStrace