Clear Sky Science · en
Revisiting type 1 diabetes progression in the non-obese diabetic mouse
Why tiny mouse pancreases matter to human health
Type 1 diabetes is often diagnosed only after most insulin-producing cells in the pancreas are already gone. To prevent or delay the disease, scientists need animal models that mirror the very earliest stages of this slow destruction. This study revisits a classic research model—the non-obese diabetic (NOD) mouse—to see whether standard ways of calling these mice “diabetic” are missing a crucial early window of damage that looks more like what happens in people. 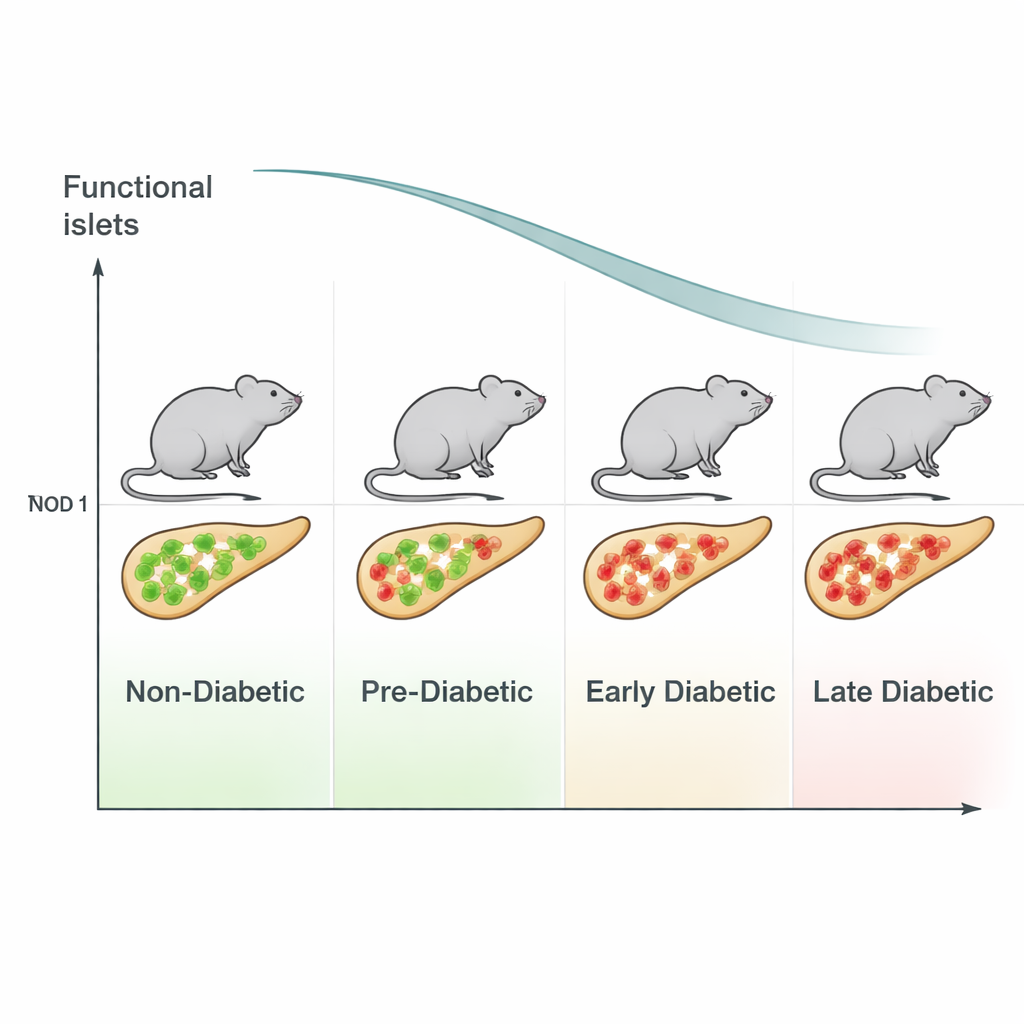
Looking inside the pancreas before diabetes is obvious
The researchers focused on NOD mice whose blood sugar levels were still below the usual mouse diabetes cutoff of 200 milligrams per deciliter (mg/dL). That range—80 to 200 mg/dL—is where subtle changes might be happening long before full-blown disease. They examined the pancreas of 38 female NOD mice of different ages, plus healthy control mice and mice with very advanced diabetes. Using advanced fluorescent staining and automated image analysis, they mapped thousands of “islets of Langerhans,” the small clusters of endocrine cells that include insulin-making beta cells. This allowed them to count which cells were present and how many immune cells had invaded each islet.
Five stages of islet damage
By combining detailed images with computer-based rules, the team sorted 3,324 islets into five stages of damage, from stage 0 (largely healthy) to stage 4 (severely damaged). In early stages, islets still contained many insulin-producing beta cells, and only a few immune cells hovered nearby. As the stages advanced, more and more T cells surrounded and penetrated the islets, beta cells nearly vanished, and glucagon-producing alpha cells, along with other endocrine cells, took over. In the most damaged islets, almost no beta cells remained, and immune cells had largely disappeared, leaving behind islets dominated by other cell types. Healthy control mice looked almost entirely like stage 0, while late-diabetic mice showed mostly stages 3 and 4. 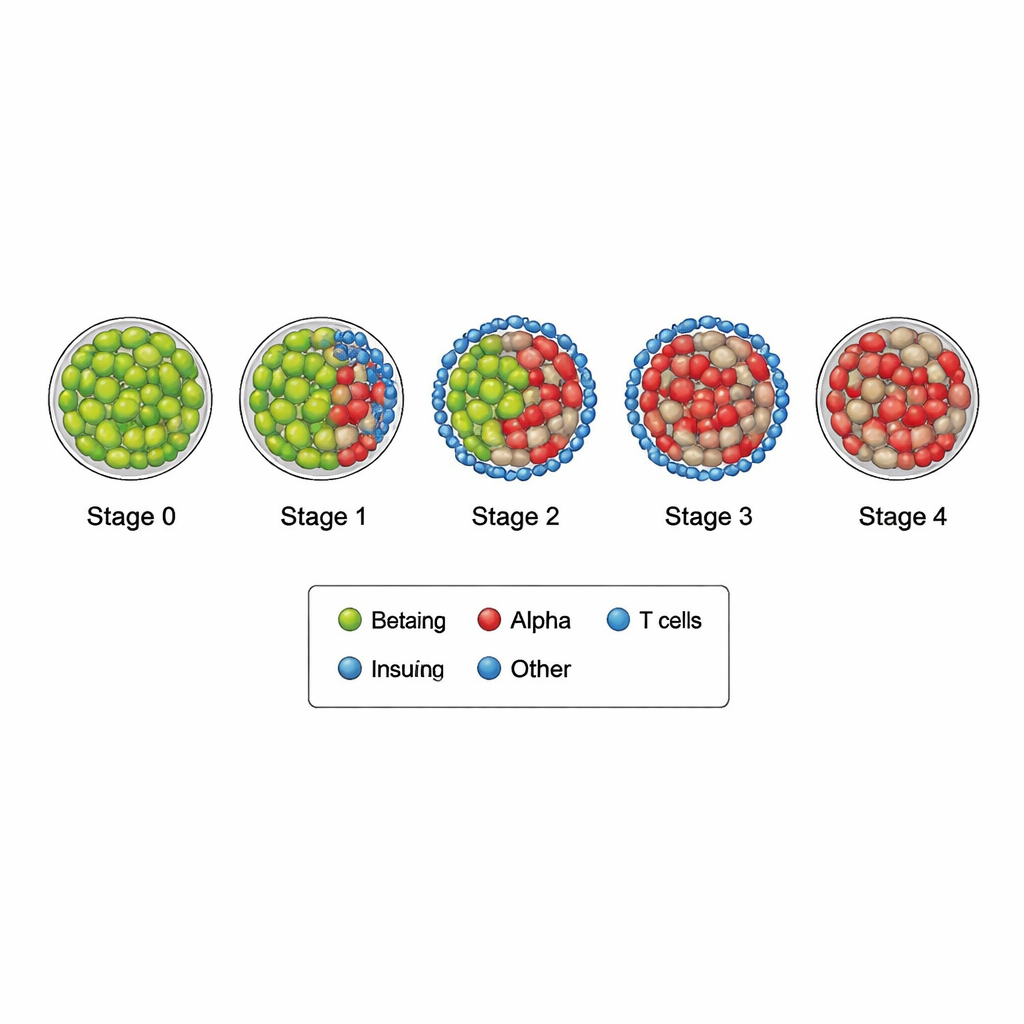
Blood sugar as an early warning sign
The scientists then linked these islet stages to each animal’s average blood sugar over the previous two weeks. They found that a major shift in islet health occurred around a blood sugar of 126 mg/dL—a familiar number, because it is also the standard fasting threshold used to diagnose diabetes in humans. Below about 115 mg/dL, most islets were in the earliest, healthier stages. Above about 135 mg/dL, most islets had progressed to the more damaged stages 3 and 4, with few beta cells left. Between these values lay a transition zone where some mice already had a large share of damaged islets despite blood sugar readings that would not yet qualify as diabetic in the usual mouse experiments.
Redefining “non-diabetic,” “pre-diabetic,” and “early diabetic” mice
To capture this hidden progression, the team created an “Islet Score,” which reflects the average stage of all islets in each mouse. Using this score together with blood sugar, they identified three meaningful subgroups in animals whose blood sugar was still under 200 mg/dL. Mice with low scores and blood sugar below 126 mg/dL were called non-diabetic; they had mostly intact islets. A small group with higher scores but still-normal blood sugar were labeled pre-diabetic: their islets were already heavily attacked even though blood sugar looked acceptable. A third, early-diabetic group had both higher scores and blood sugar over 126 mg/dL, marking a point where the pancreas was losing its grip on blood sugar control, but before the extreme levels traditionally used to define diabetes in NOD mice.
What this means for future diabetes research
This work suggests that the standard mouse threshold of 200 mg/dL misses a critical early phase of beta cell loss. By lowering the practical cutoff to around 126 mg/dL and using the new islet-based scoring system, scientists can align the NOD mouse model more closely with how type 1 diabetes develops in humans. For non-specialists, the key message is that damage to insulin-producing cells can be extensive long before blood sugar looks dramatically abnormal. Recognizing and studying this earlier stage in mice may help researchers design and test therapies aimed at protecting beta cells before they are largely destroyed.
Citation: Ehall, B., Herbsthofer, L., Obermüller, B. et al. Revisiting type 1 diabetes progression in the non-obese diabetic mouse. Sci Rep 16, 5768 (2026). https://doi.org/10.1038/s41598-026-35483-9
Keywords: type 1 diabetes, NOD mouse, beta cell loss, islet inflammation, early diagnosis