Clear Sky Science · en
Dipolar modulation of surface states in GaN via molecular ionization energy
Why tuning a crystal’s skin matters
Electronics built from gallium nitride (GaN) power today’s fastest chargers, 5G base stations, and electric vehicles. Yet the very outer “skin” of GaN—the few atomic layers exposed to air—often behaves unpredictably, causing unwanted power loss and device drift over time. This paper shows that simple gas molecules from our everyday environment, like water, carbon monoxide, and nitrogen dioxide, can systematically tune the electrical behavior of that surface. By revealing a clear rule that links a molecule’s ionization energy to how it shifts GaN’s surface, the work points to new ways to design more stable, efficient devices and even next‑generation light‑driven electron sources.
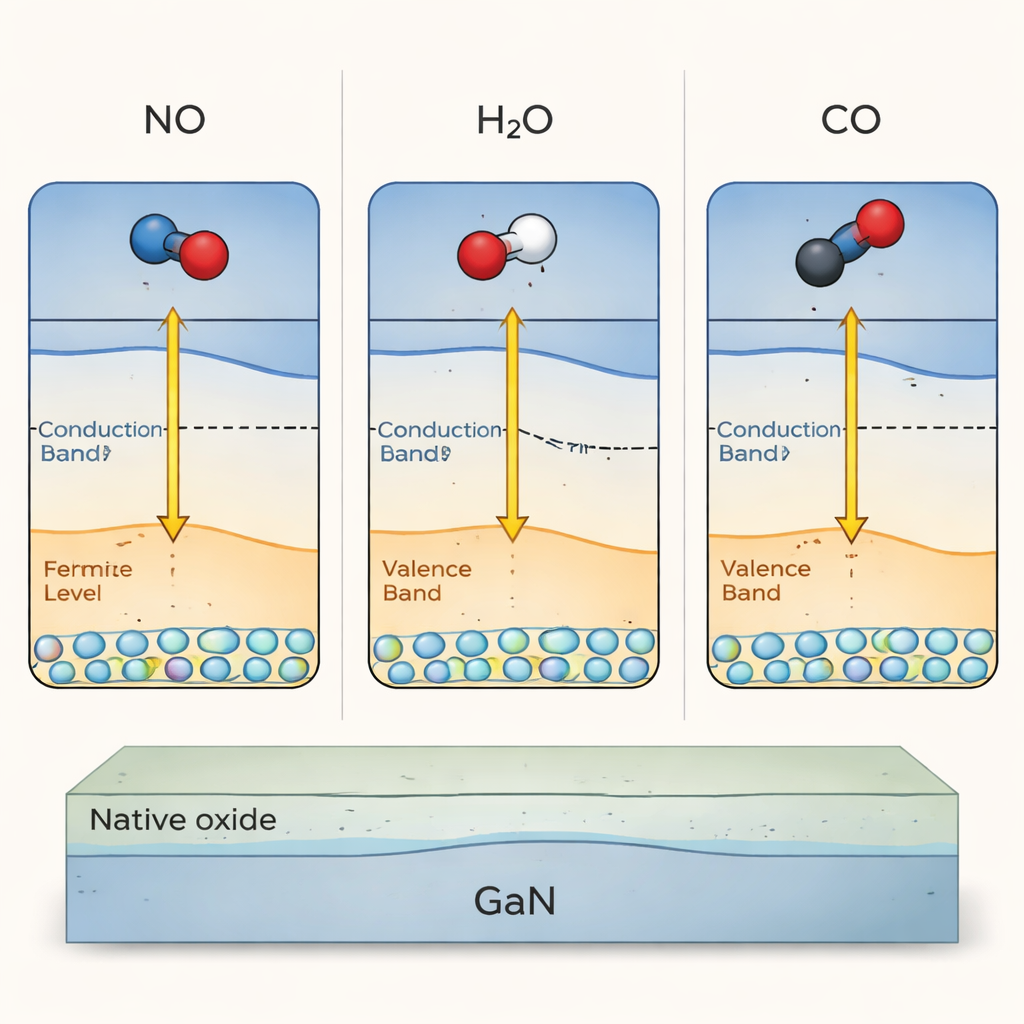
The fragile outer layer of powerful semiconductors
GaN is prized for handling high voltages and high frequencies, but its surface is a trouble spot. Unlike silicon, GaN does not form a smooth, well‑behaved native oxide. Instead, a thin, disordered gallium oxide film appears when the material is exposed to air. Electronic “surface states” at or near this Ga‑facing surface can trap charge, leading to problems such as sudden drops in current and unstable switching thresholds in transistors. Because the surface is so chemically reactive, everyday gases can change these states in ways that are hard to predict, making it difficult to design truly reliable GaN electronics.
Using light and electrons to watch charges move
To uncover what really controls these surface states, the researchers combined two sensitive tools. Surface photovoltage spectroscopy shines light on the sample and measures tiny voltage shifts as trapped charges are released, revealing how much charge is stored at different energies near the surface. X‑ray photoelectron spectroscopy, in turn, fires high‑energy X‑rays at the surface and records the energies of emitted electrons, which report on chemical bonds and the presence of the native oxide. They worked with carefully grown GaN layers, gently removed surface charge by mild heating in vacuum, and then exposed the same surface, in a controlled way, to three gases: nitrogen dioxide (NO₂), water (H₂O), and carbon monoxide (CO).
A simple rule linking molecules to surface energy
Each gas rebuilt the charge trapped in GaN’s so‑called “yellow band” of surface states, but with a twist: the peak in the charge spectrum shifted slightly for each molecule. By fitting those peaks with a standard Fermi function, the team extracted where the surface Fermi level—the energy that separates filled from empty electronic states—ended up after adsorption. When they plotted this Fermi level position against each molecule’s ionization energy (a basic property that measures how hard it is to remove an electron from the molecule), the points lined up almost perfectly along a straight line. This means the Ga‑face of GaN is not locked, or “pinned,” to one surface energy; instead, it can be predictably tuned by choosing molecules with different ionization energies that donate or withdraw different amounts of charge.
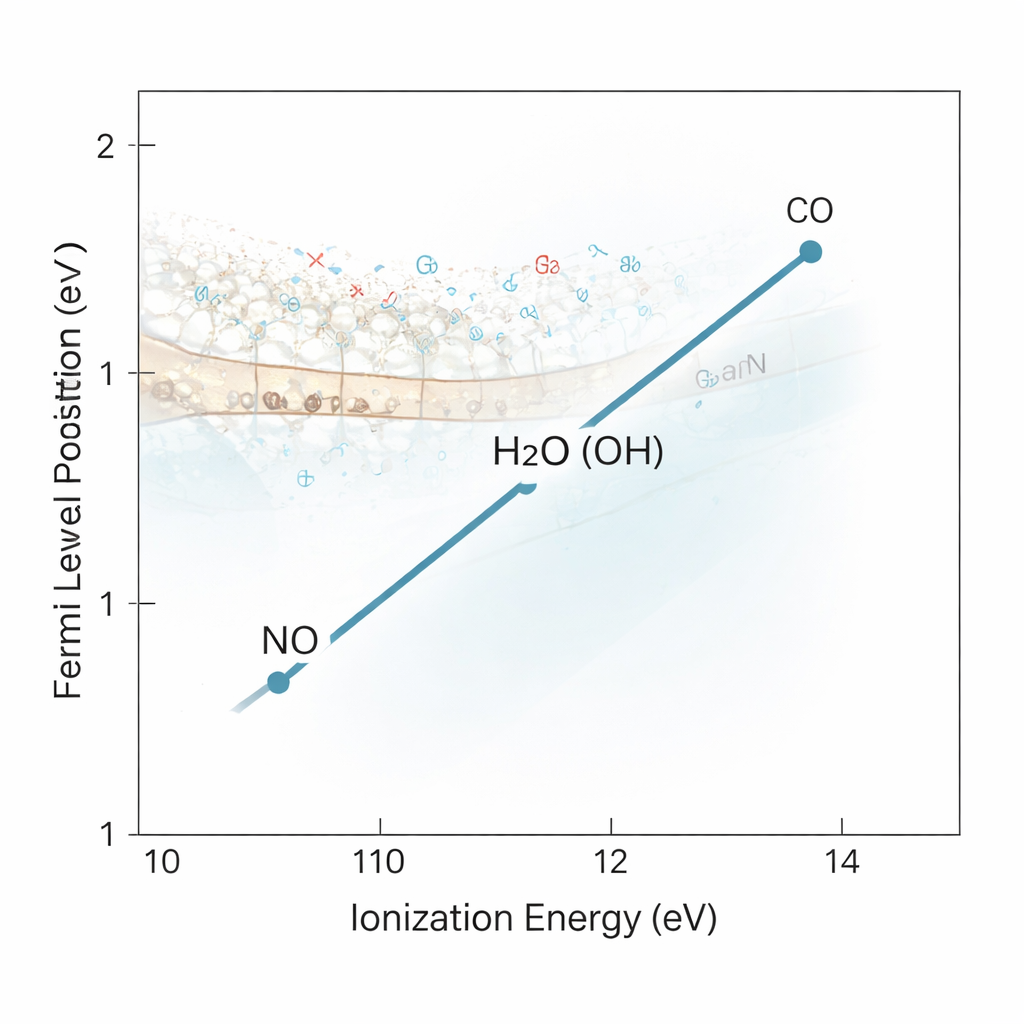
The hidden role of the native oxide interface
A surprising finding was that this tunability vanished when the native gallium oxide was stripped away with a hydrochloric acid etch. After removing the Ga–O–related signal in the X‑ray spectra, the distinctive charge peaks tied to the adsorbed molecules all but disappeared. That points to the key action happening not in the GaN crystal itself, but at the boundary where GaN meets its thin, amorphous oxide. In effect, the molecules form a dipolar layer on top of this oxide that acts like the “gate” in a transistor, shifting the bands inside the GaN by electrostatic means. Modeling this situation as a leaky metal–oxide–semiconductor stack, the authors showed that the amount of band bending—and thus the surface charge—matches what one would expect from such an interface dipole.
Toward robust, low‑barrier electron surfaces
When the team translated their measurements into work function values—the energy needed for an electron to escape from the surface—they found numbers near just 1 electron volt, strikingly low compared with the large ionization energies of the individual molecules. This is reminiscent of so‑called negative electron affinity surfaces, where electrons can be emitted with very little energy cost. Classic versions use fragile cesium–oxygen layers that only survive under ultra‑high vacuum. Here, however, common molecules like water and carbon monoxide appear to form chemically bonded, dipolar structures with the native oxide, promising far greater stability in air. While the exact microscopic bonding arrangements remain to be nailed down, the message is clear for non‑experts: by smartly choosing and attaching the right molecules to GaN’s native oxide, engineers can dial in its surface energy landscape—mitigating device instabilities today, and possibly enabling rugged, low‑barrier electron emitters tomorrow.
Citation: Chaulker, O.H., Turkulets, Y., Shapira, Y. et al. Dipolar modulation of surface states in GaN via molecular ionization energy. Sci Rep 16, 5224 (2026). https://doi.org/10.1038/s41598-026-35475-9
Keywords: gallium nitride surfaces, molecular adsorption, surface states, negative electron affinity, interface dipoles