Clear Sky Science · en
Global RNA expression analysis of patient samples identified potential diagnostic biomarkers specific for peritoneal, ovarian and deep endometriosis
Why this condition matters to everyday life
Endometriosis affects an estimated 190 million women worldwide, often causing severe pelvic pain, fatigue, and fertility problems. Yet many people wait nearly a decade for a clear diagnosis, usually confirmed only through surgery. This study asks a simple but powerful question: can we instead read the body’s molecular “fingerprints” to detect endometriosis earlier, and even tell its different forms apart, using a simple test in the future?
Different faces of the same disease
Endometriosis occurs when tissue similar to the uterine lining grows where it does not belong, such as on the ovary, the thin lining of the abdomen (peritoneum), or deep in the pelvic organs. These locations define three main subtypes: ovarian, peritoneal, and deep endometriosis. Each can cause different patterns of pain, fertility issues, and organ damage. Today, doctors rely on symptoms, imaging, and often laparoscopy—an operation under anesthesia—to confirm the diagnosis and see which subtype is present. A non-invasive way to distinguish these forms would spare many women years of uncertainty and surgery.
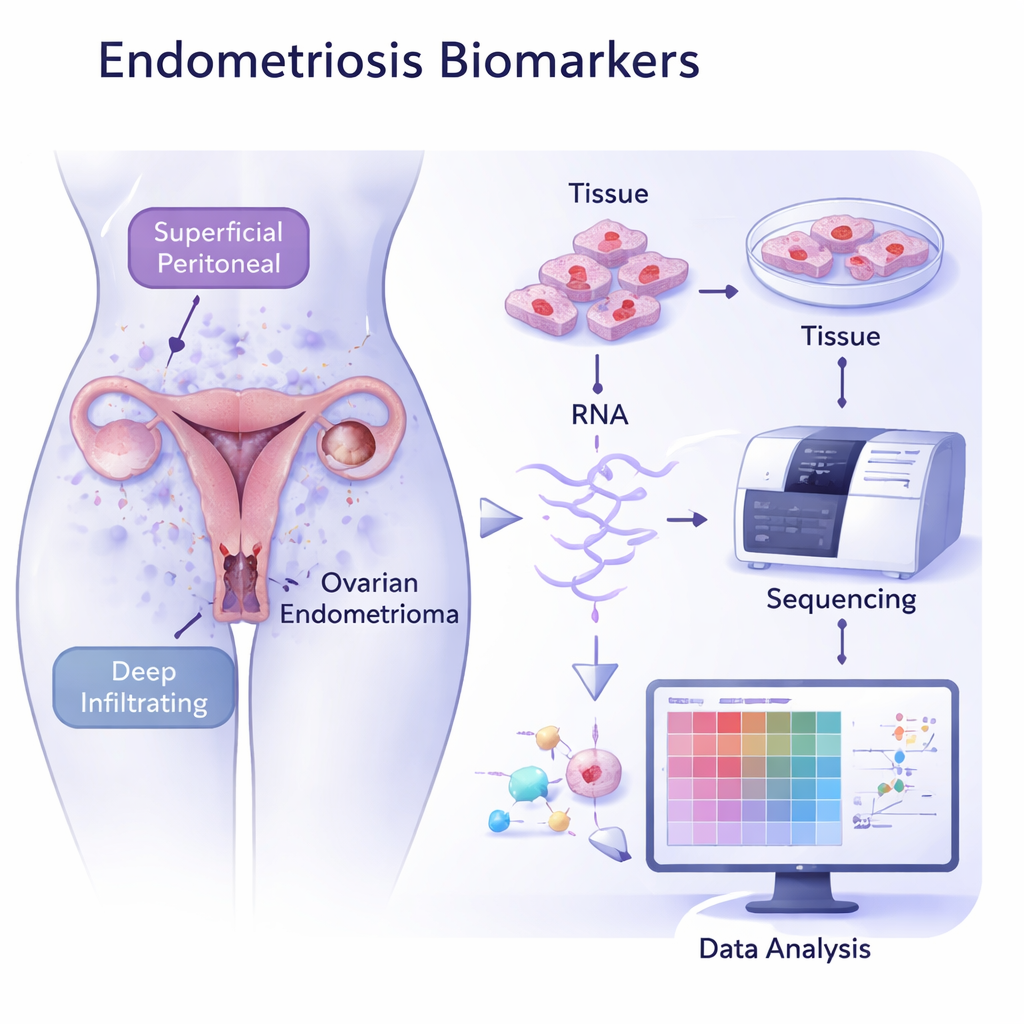
Reading the RNA “barcode” of diseased tissue
The researchers collected tissue samples from 26 women with endometriosis—across all three subtypes—and from 15 women without the disease. They then used RNA sequencing, a method that measures which genes are turned on or off, to build a global picture of the activity inside these tissues. When they compared diseased tissue to healthy uterine lining, the samples fell into distinct clusters, revealing clearly different gene activity patterns. Ovarian lesions formed one tight group, while peritoneal and deep lesions grouped together, supporting the idea that these subtypes follow different biological routes even though they share some common features.
Shared pathways of scarring and inflammation
Across all three subtypes, the team found higher activity in genes linked to building and remodeling the tissue scaffold (the extracellular matrix), increased cell contractility typical of scarring, and strong inflammatory signaling. These changes fit with what doctors see clinically: stiff, fibrotic lesions that cause pain and adhesions. The study also detected an altered immune landscape within lesions. So-called M2 macrophages, a type of immune cell associated with wound healing and fibrosis, were especially abundant, while natural killer (NK) cells—important for clearing abnormal cells—were markedly reduced. This imbalance may help endometriotic tissue survive and spread despite being out of place.
Subtype-specific molecular clues
Beyond these shared patterns, each subtype carried its own molecular signature. In deep and peritoneal endometriosis, genes involved in phosphodiesterase signaling—pathways already linked to other fibrotic diseases—were strongly increased. In ovarian endometriosis, genes related to immune presentation and hormone production stood out, suggesting that the ovary provides a distinct biochemical environment. The researchers also explored non-coding RNAs, molecules that do not make proteins but can regulate gene activity. They identified several that were greatly increased overall or specifically in certain subtypes, hinting at new regulatory players and possible biomarkers that might be detectable in blood or other body fluids.
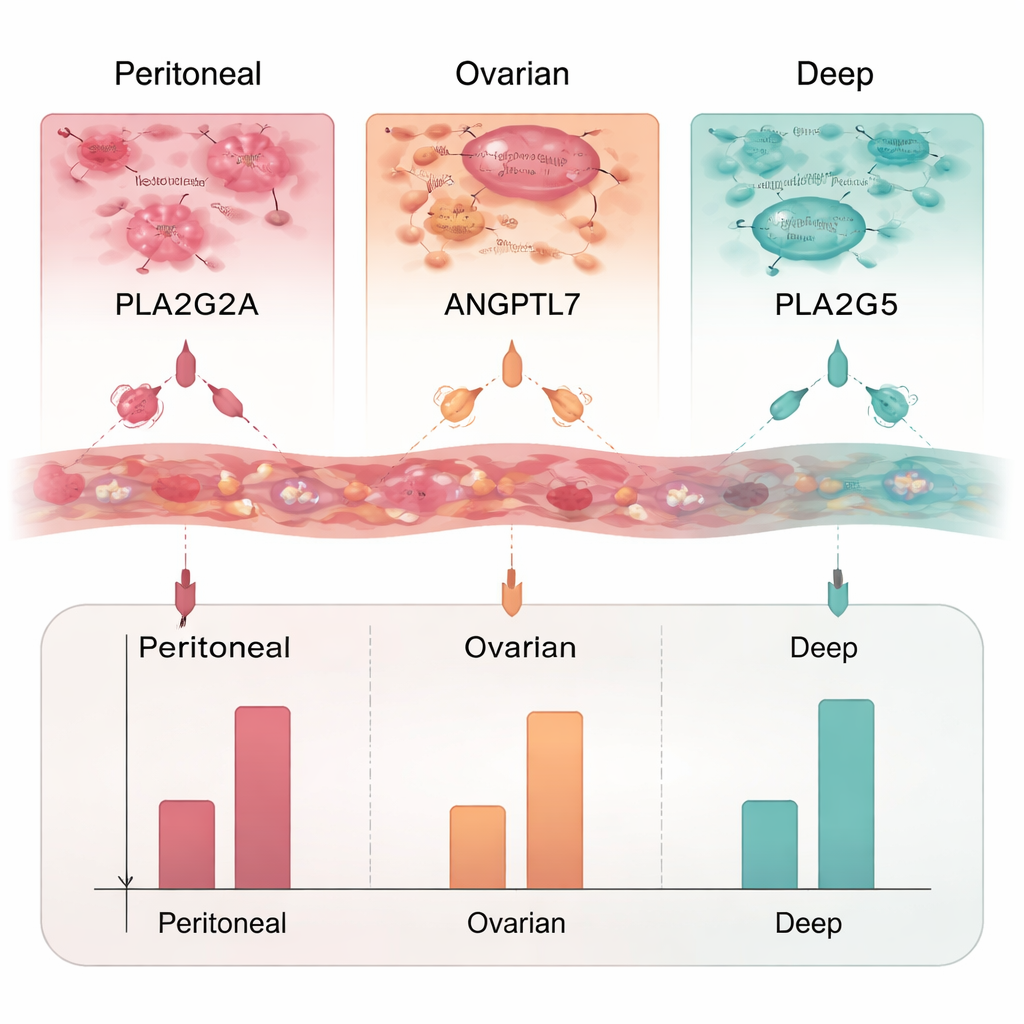
Promising protein markers for future tests
From the long list of altered genes, the team focused on those that produce secreted proteins—ideal candidates for a future blood test. They chose three proteins for closer study: PLA2G2A, ANGPTL7, and PLA2G5. Using ELISA, a laboratory method that measures protein levels, they found that PLA2G2A was elevated in all three endometriosis types, particularly in ovarian lesions. ANGPTL7 was increased mainly in deep and peritoneal lesions, while PLA2G5 was raised only in deep endometriosis. Together, these proteins form a potential panel that could not only flag the presence of endometriosis, but also hint at where the lesions are located.
What this means for patients
This research does not yet deliver a ready-made blood test, but it lays important groundwork. By mapping how thousands of genes and immune cells behave in different endometriosis subtypes, the study pinpoints specific proteins— PLA2G2A, ANGPTL7, and PLA2G5—that could be measured non-invasively in the future. If validated in larger, independent groups of patients, combinations of such markers could shorten the long road to diagnosis, reduce the need for surgery just to confirm the disease, and allow more tailored treatment based on the exact subtype a woman has.
Citation: Lisá, Z., Fanta, M., Kokavec, J. et al. Global RNA expression analysis of patient samples identified potential diagnostic biomarkers specific for peritoneal, ovarian and deep endometriosis. Sci Rep 16, 5070 (2026). https://doi.org/10.1038/s41598-026-35467-9
Keywords: endometriosis, biomarkers, RNA sequencing, immune cells, women’s health