Clear Sky Science · en
Zebra bodies recognition by artificial intelligence (ZEBRA): a computational tool for Fabry nephropathy
Why tiny kidney changes matter
Fabry disease is a rare inherited condition that slowly damages many organs, especially the kidneys. Treating it early can prevent serious problems, but the first warning signs inside kidney tissue are often subtle and easy to miss, even for experts. This study presents a new artificial intelligence (AI) tool, called ZEBRA, that scans digital images of kidney biopsies to help doctors spot these early changes more reliably and quickly.
A rare disease with quiet beginnings
In Fabry disease, a missing or faulty enzyme causes fatty molecules to build up inside cells throughout the body, including the tiny filters in the kidneys. These filters contain specialized cells called podocytes, which help clean the blood. When they become overloaded, their insides look swollen and “foamy” under the microscope. This foamy appearance is one of the few early clues that the kidneys are affected, especially in women and in people with milder, late-onset forms of the disease. However, similar foamy changes can appear in other conditions, and the gold-standard confirmation method—electron microscopy—is not always available. As a result, Fabry kidney involvement can be overlooked, delaying diagnosis and treatment.
Turning glass slides into digital clues
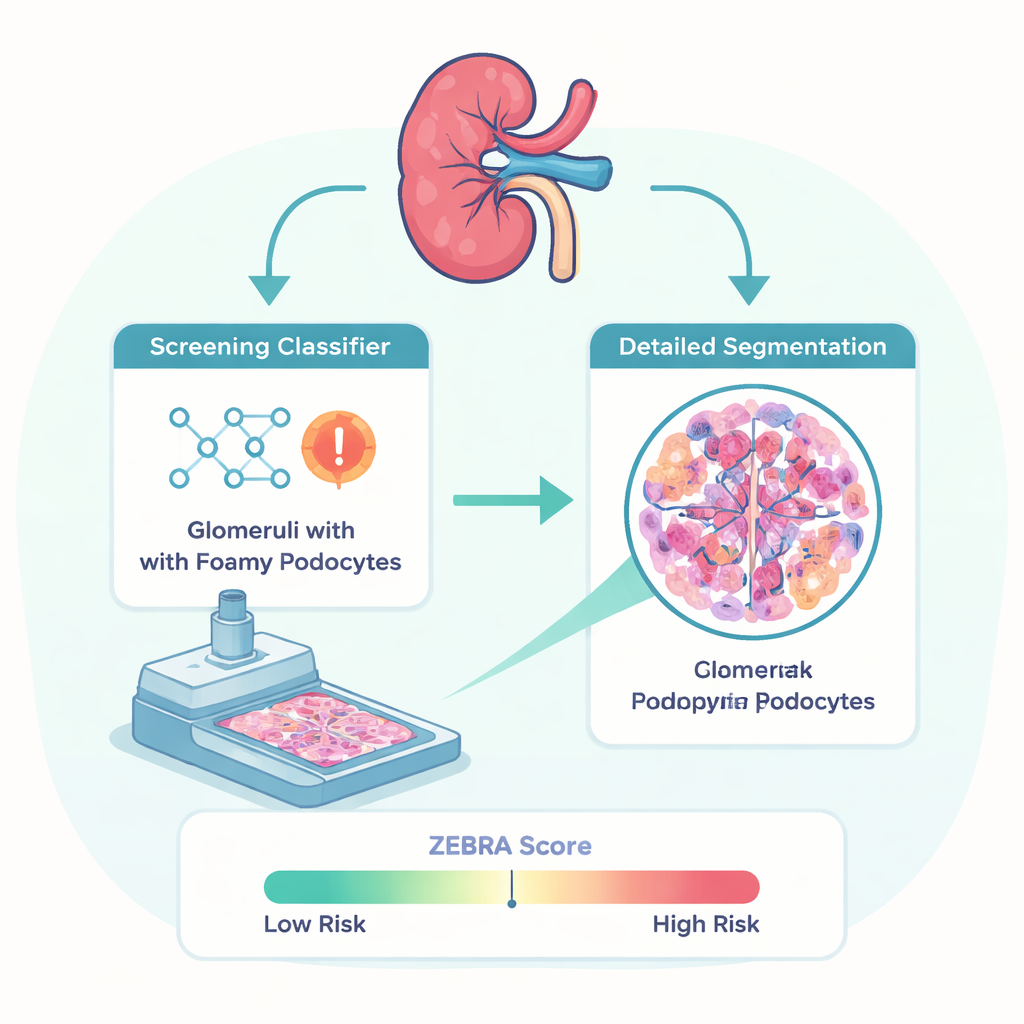
To tackle this problem, researchers from several Italian centers collected kidney biopsy samples from 37 people with genetically proven Fabry nephropathy and 40 patients with other kidney diseases. The slides were scanned to create high-resolution digital images. Expert kidney pathologists then carefully marked each glomerulus (the tiny filtering unit in the kidney) and outlined individual foamy podocytes. Using these detailed markings as a reference, the team trained two types of AI models: a “classification” model to decide whether a glomerulus contains foamy podocytes, and a “segmentation” model to trace exactly where these abnormal cells sit within each glomerulus.
Teaching computers to see what experts see
The classification model that performed best, called EfficientNetB2, correctly labeled glomeruli with or without foamy podocytes about four out of five times. Importantly, at the patient level it detected all Fabry cases in the independent test group, though it sometimes misflagged glomeruli from non-Fabry diseases as suspicious. This makes it especially useful as a high-sensitivity screening tool that can alert pathologists to cases that deserve a closer look. The segmentation model, based on a modern transformer architecture (SegFormerB4), was less perfect in tracing exact borders but was very sensitive at recognizing when foamy podocytes were present at all. Together, these models form the ZEBRA pipeline, which is released as free software that can plug into commonly used digital pathology platforms.
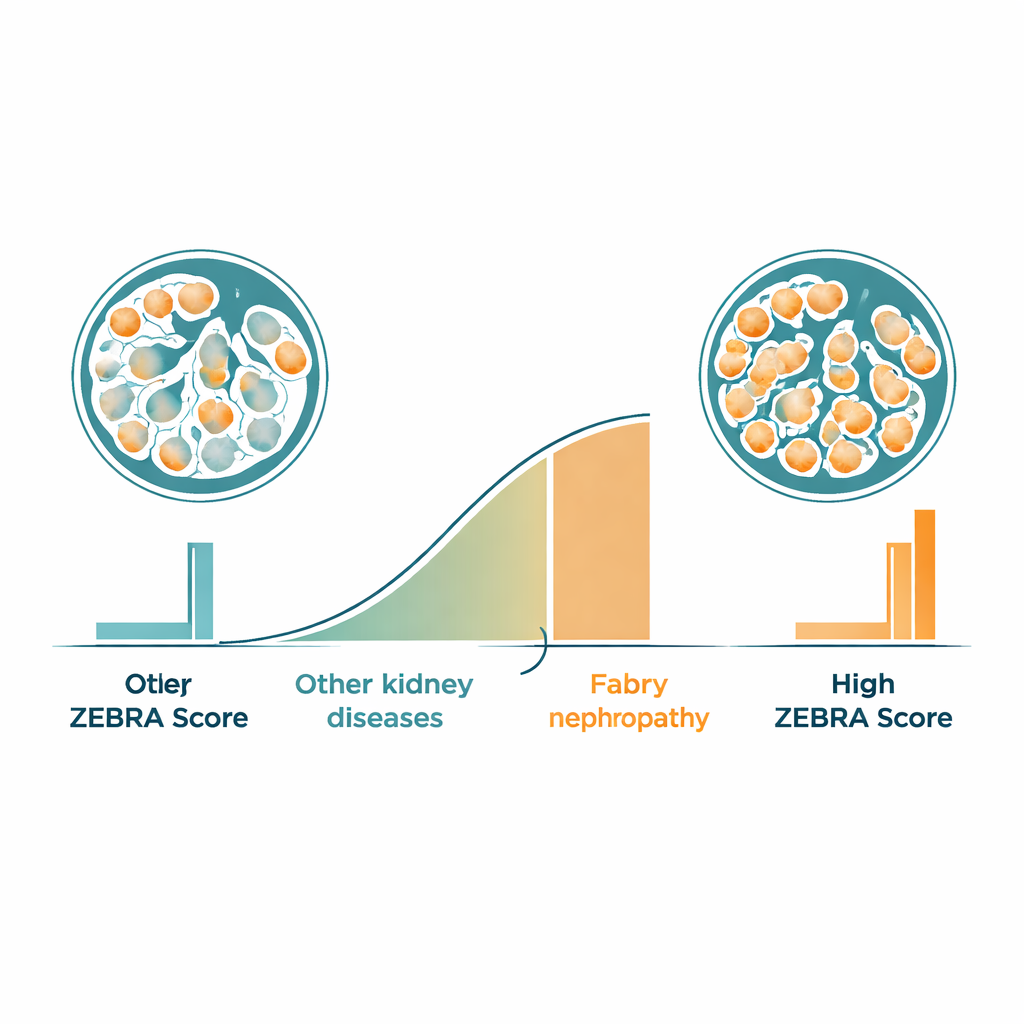
From pixels to a simple risk score
Using the segmentation results, the researchers created a new numeric measure called the ZEBRA score. For each glomerulus, the software calculates what fraction of the area is occupied by foamy podocytes and then summarizes this for every patient. When they compared people with Fabry nephropathy to those with other kidney diseases, the ZEBRA score clearly separated the two groups, with almost no overlap. A proposed cutoff value could distinguish Fabry from non-Fabry cases with high sensitivity and good specificity. The score also tracked reasonably well with manual grading by pathologists and showed modest links to kidney function and protein leakage in the urine, even in patients whose lab tests still looked relatively normal.
What this means for patients and care teams
This work shows that AI can act as an extra pair of highly attentive eyes on routine kidney biopsy slides, helping pathologists notice patterns that might otherwise be missed. While the ZEBRA score is not meant to replace genetic testing or specialist judgment, it can flag high-risk cases, prompt further testing, and support more consistent reporting across hospitals. With larger studies and long-term follow-up, this digital tool could eventually help doctors not only diagnose Fabry disease earlier but also monitor how well treatments protect the kidneys over time.
Citation: Cazzaniga, G., Carbone, M., Barretta, R. et al. Zebra bodies recognition by artificial intelligence (ZEBRA): a computational tool for Fabry nephropathy. Sci Rep 16, 5072 (2026). https://doi.org/10.1038/s41598-026-35466-w
Keywords: Fabry disease, kidney biopsy, digital pathology, artificial intelligence, ZEBRA score