Clear Sky Science · en
DFT study of benzothiadiazole based small molecules for high efficiency organic photovoltaics
Why Better Solar Materials Matter
Solar panels are becoming a familiar sight on rooftops and in fields, but the technology behind them is still rapidly evolving. Today’s most efficient commercial panels are made from rigid silicon wafers, which are effective but costly, heavy, and hard to integrate into curved surfaces or lightweight devices. This paper explores a new class of tailor‑made organic molecules that could power thinner, cheaper, and more flexible solar cells—potentially turning windows, clothing, or portable gadgets into power sources.
From Rigid Panels to Flexible Films
Traditional silicon solar cells are excellent at turning sunlight into electricity, but they come with trade‑offs: they are brittle, require high‑temperature manufacturing, and are difficult to adapt to lightweight or bendable products. Organic solar cells, built from carbon‑based molecules, promise something different. They can be printed like ink, tuned by chemistry, and laid down as ultrathin films on flexible plastic. To reach their full potential, however, they need light‑absorbing materials that grab more of the solar spectrum and move electrical charges with minimal loss. This study focuses on designing such materials on a computer before they are ever made in the lab.
Designing New Building Blocks on a Screen
The researchers started from a known small molecule used in organic electronics and simplified it into a reference structure, called REF. This reference acts as a backbone made of a central “donor” segment flanked by two “acceptor” segments. The team then created eight new variants (G1–G8) by swapping out the chemical groups on the ends of the molecule. These terminal groups are like adjustable knobs: by choosing stronger or weaker electron‑pulling ends, the scientists can shift how the molecule absorbs light and how easily it passes charges along. Using quantum‑mechanical simulations (a branch of theory known as density functional theory), they predicted each molecule’s color absorption, electrical energy levels, and how efficiently it might work inside a solar cell.
Capturing More Sunlight, Wasting Less Energy
The virtual experiments showed that all eight new designs outperform the original backbone in key ways. Their energy gaps—the difference between the levels where electrons sit and where they can move freely—are smaller than in REF, which means they can absorb redder and near‑infrared light that silicon and many older organics leave unused. One standout, labeled G7, absorbs light strongly around 803 nanometers, deep in the red region, and achieves an almost perfect light‑harvesting efficiency close to 100% in the simulations. Several molecules also show very low “reorganization energies,” a measure of how much the molecular structure has to flex as charges move. Lower values here translate into faster, smoother charge transport and fewer losses inside a working device.
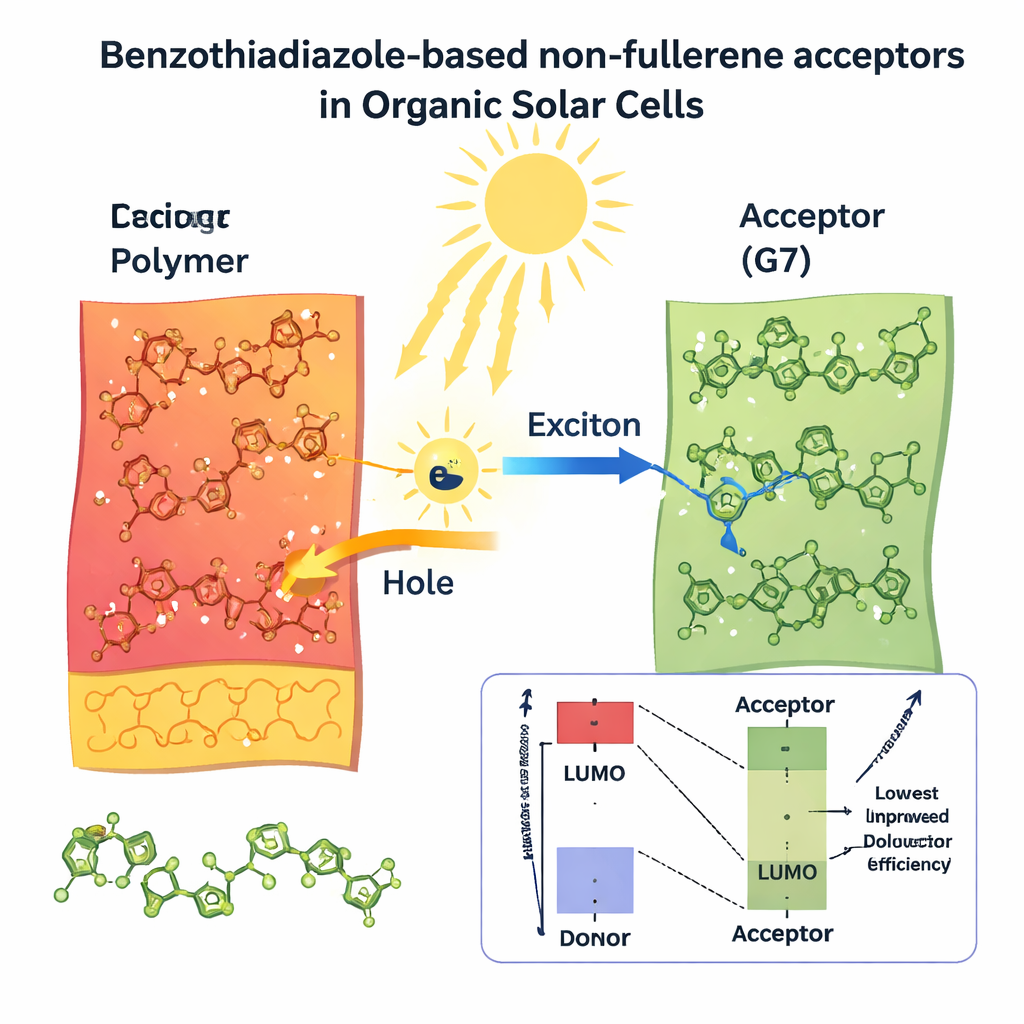
Balancing Voltage, Current, and Overall Output
Good solar materials must do more than just absorb light; they must also generate high voltage, deliver strong electrical current, and keep resistive losses small. The authors estimated these practical performance metrics—open‑circuit voltage, fill factor, and overall power conversion efficiency—by combining their quantum calculations with established device models. They predict that all eight new molecules could, in principle, reach efficiencies above 20%, far above the 12% estimated for the original reference structure. Two candidates stand out for different reasons. G7 offers the highest predicted current because it captures the broadest slice of sunlight, making it attractive for tandem or low‑light applications. G5, on the other hand, strikes the best balance: in the model it delivers strong current, high voltage, and an excellent fill factor, leading to a projected efficiency of about 37% under standard sunlight.
What This Means for Future Solar Tech
For non‑experts, the key takeaway is that chemistry can be used like a fine‑tuning dial for solar materials. By changing only the small groups at the ends of an otherwise similar molecule, the researchers were able to predict large gains in how much sunlight can be captured and how efficiently it can be turned into electricity. While these results are theoretical and still need to be proven in the lab, they point toward a clear design recipe for next‑generation organic solar cells: engineer terminal units that extend light absorption, encourage clean separation of charges, and keep molecular motion during charge transport to a minimum. Among the virtual candidates, G7 shines for its light‑harvesting power, while G5 offers the most practical all‑around performance, making both strong contenders for future flexible, high‑efficiency solar films.
Citation: Ghaffar, A., Yousuf, A., Qureshi, M.Z. et al. DFT study of benzothiadiazole based small molecules for high efficiency organic photovoltaics. Sci Rep 16, 5859 (2026). https://doi.org/10.1038/s41598-026-35432-6
Keywords: organic solar cells, non-fullerene acceptors, benzothiadiazole, photovoltaic efficiency, molecular design