Clear Sky Science · en
Signal recognition particle-dependent secretome in humans
How Cells Decide Which Proteins Leave the Cell
Our cells constantly ship out proteins that form hormones, antibodies, and the scaffolding between tissues. But not every protein is meant to travel. This study asks a basic yet unanswered question: how do human cells decide which proteins rely on a key helper called the signal recognition particle (SRP) to reach the cell’s internal shipping center, and which can get there without it? By mapping this decision across thousands of proteins at once, the authors reveal who depends on SRP, who does not, and what happens when this system breaks down—a topic relevant to many diseases tied to misrouted or missing secreted proteins.
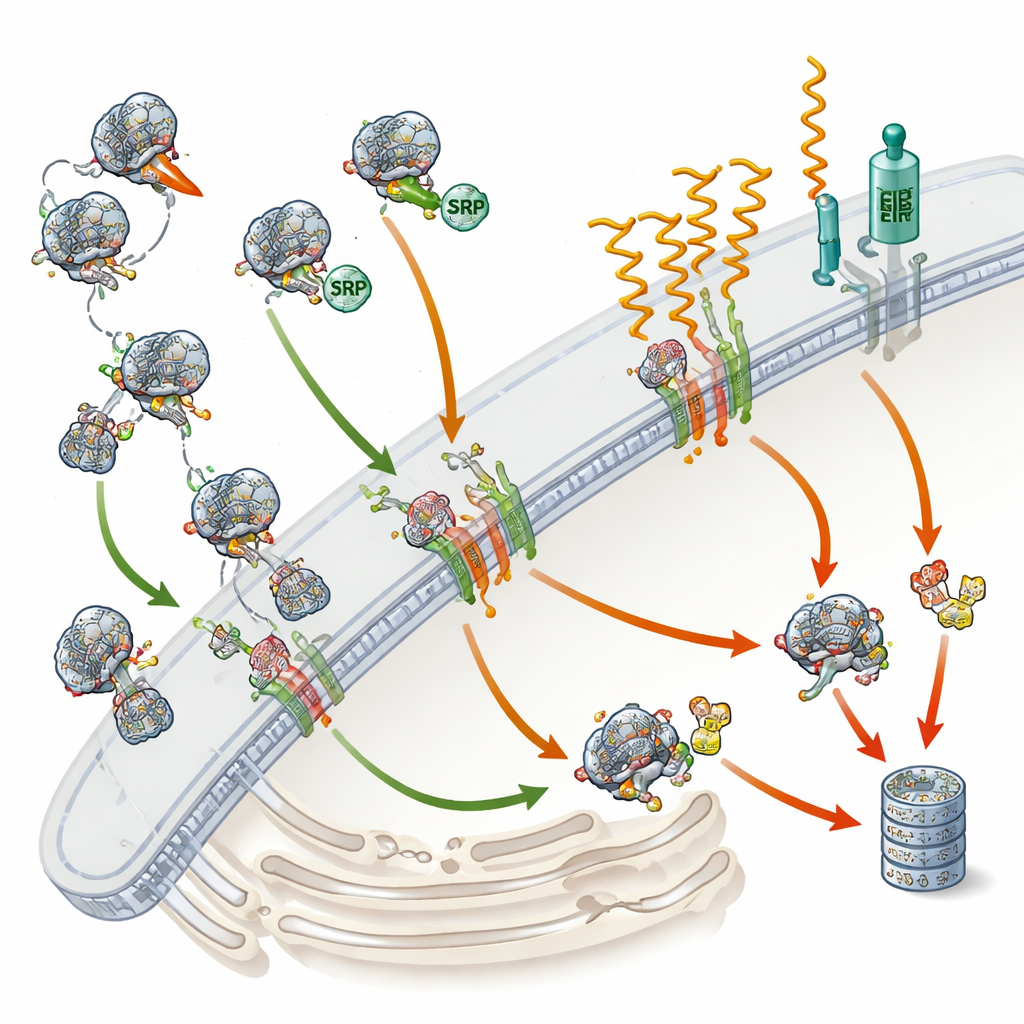
The Cell’s Shipping Dock and Its Main Guide
Roughly one out of every three human proteins is destined either for the cell surface, cellular membranes, or for release outside the cell. To reach the right place, most of these proteins must first enter a network of internal membranes called the endoplasmic reticulum, the cell’s shipping dock. SRP acts as a guide: it recognizes a small “address tag” at the beginning of newly made proteins and temporarily pauses their production. This pause gives the ribosome—the protein-making machine—time to dock on the endoplasmic reticulum, where production resumes and the growing protein is threaded into or through the membrane. Some proteins, however, are known to use alternative routes that do not require SRP, especially very small proteins or those anchored at their tail ends. Until now, scientists did not know, on a global scale, which human proteins truly require SRP and which can bypass it.
Turning SRP Down to See What Breaks
To answer this, the researchers selectively reduced levels of one essential SRP component, SRP54, in human HeLa cells. They then used a sensitive technique called mass spectrometry to measure over 6,000 proteins inside the cells and nearly 2,000 proteins present in the surrounding liquid, known as conditioned medium. Comparing cells with normal SRP54 to those with depleted SRP54 allowed them to see which proteins became less abundant, which increased, and which were unchanged. A strong drop in a protein’s level—especially both inside the cell and in the medium—was taken as a sign that this protein depends on SRP to be made and routed correctly.
Who Depends on SRP and Who Manages Without It
The analysis showed that most of the proteins normally secreted into the medium clearly rely on SRP. When SRP54 was reduced, the majority of these secreted proteins, particularly those carrying a classic signal “address tag,” dropped sharply. Many membrane proteins inside the cell also decreased, revealing them as SRP clients. A smaller set of proteins with similar address tags was largely unaffected, marking them as SRP-independent; these likely use alternative routes to reach the endoplasmic reticulum. The researchers also discovered that specific features of the address tag—such as a positively charged region at its beginning—were more common among SRP-dependent proteins, hinting at subtle rules that help SRP choose its cargo. At the same time, some proteins involved in tagging damaged proteins for destruction and in mitochondrial function increased, suggesting that cells activate backup systems and energy production when their main shipping guide is compromised.
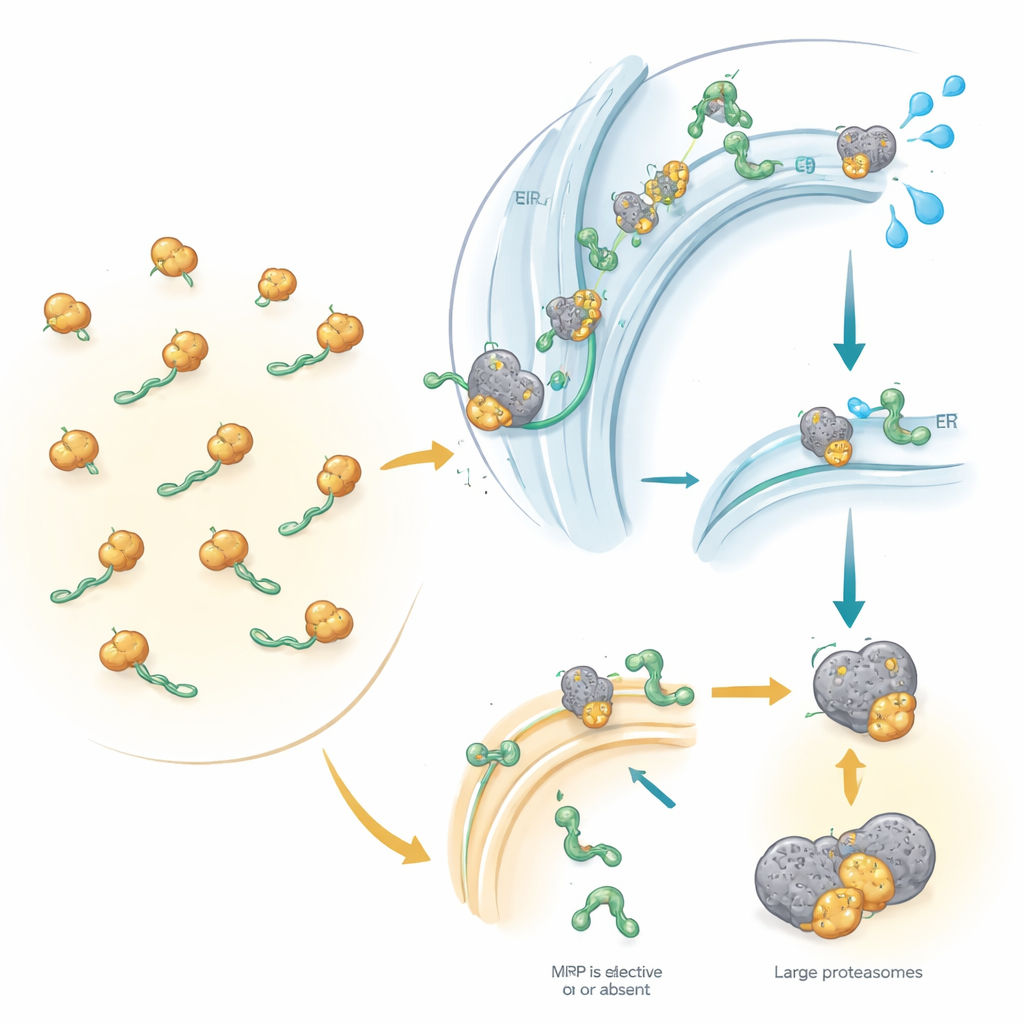
Quality Control When Targeting Goes Wrong
The study went beyond counting proteins: it compared these changes to shifts in messenger RNA, the blueprints used to make proteins. Many SRP-dependent proteins lost not only protein but also their RNA blueprints, implicating a quality-control pathway known as RAPP. When SRP cannot properly engage an emerging secretory or membrane protein, this pathway appears to trigger the breakdown of the corresponding RNA, preventing further production of a misrouted protein. At the same time, incompletely made proteins that fail to reach the endoplasmic reticulum are likely tagged with small molecular flags and fed to cellular “shredders” called proteasomes. Interestingly, classical stress pathways in the endoplasmic reticulum were not strongly activated, hinting that defective SRP provokes a distinct type of cellular stress response.
What This Means for Health and Disease
By charting, for the first time in human cells, which secreted and membrane proteins are SRP-dependent and which are SRP-independent, this work provides a reference map of the cell’s shipping rules. The findings show that most exported proteins truly rely on SRP and that, when SRP falters, cells respond by degrading both the proteins and their blueprints while turning on alternative coping mechanisms. Because many disease-causing mutations affect the signal “address tags” on secreted proteins, understanding exactly how SRP recognizes its clients and how RAPP removes faulty ones may help explain why certain mutations lead to hormone deficiencies, immune problems, or neurodegeneration—and may eventually point to new ways to fine-tune protein routing in human health.
Citation: Miller, S.C., Tikhonova, E.B., Rodríguez-Almonacid, C.C. et al. Signal recognition particle-dependent secretome in humans. Sci Rep 16, 8760 (2026). https://doi.org/10.1038/s41598-026-35427-3
Keywords: protein secretion, signal recognition particle, secretome, protein quality control, endoplasmic reticulum