Clear Sky Science · en
Sequential platinum and PARP Inhibition enhances PD1 immunotherapy efficacy in murine Brca2 mutated pancreatic cancer
Why this research matters for patients
Pancreatic cancer is one of the deadliest cancers, and even when doctors know a tumor carries a helpful vulnerability—such as a mutation in the BRCA2 gene—today’s treatments rarely keep the disease under control for long. This study uses a realistic mouse model to ask a practical question that many patients and oncologists now face: after strong chemotherapy and a PARP inhibitor, can adding modern immunotherapy meaningfully extend survival, and why do tumors so often come back?
A stubborn cancer with a genetic weak spot
Pancreatic ductal adenocarcinoma is notoriously difficult to treat, with five‑year survival still in the single digits. A small fraction of patients carry inherited or tumor‑specific defects in BRCA2, a gene needed to repair broken DNA. These tumors are unusually sensitive to drugs that damage DNA, like platinum‑based chemotherapy, and to PARP inhibitors, which further cripple cancer cells’ ability to repair DNA. Current standard care for advanced BRCA‑mutated pancreatic cancer is months of platinum chemotherapy followed by maintenance treatment with a PARP inhibitor. Yet in the key POLO clinical trial, this strategy did not extend overall survival, underscoring the need to understand resistance and to design better maintenance approaches.
Building a more lifelike mouse model
Many existing laboratory models strip away the immune and stromal components that surround real tumors in patients. To overcome this, the authors engineered mice that develop BRCA2‑deficient pancreatic tumors in the setting of a fully functioning immune system. From these spontaneous tumors they derived clonal cell lines that could be implanted into genetically matched mice. These Brca2‑mutant tumors looked and behaved a lot like human pancreatic cancers: they were densely fibrotic, largely excluded T cells, and showed high levels of spontaneous DNA damage. Crucially, the tumors were highly sensitive to a common platinum combination (gemcitabine plus cisplatin), whereas PARP inhibitor treatment alone had only modest and short‑lived effects in animals, despite strong activity against the same cells in a dish. 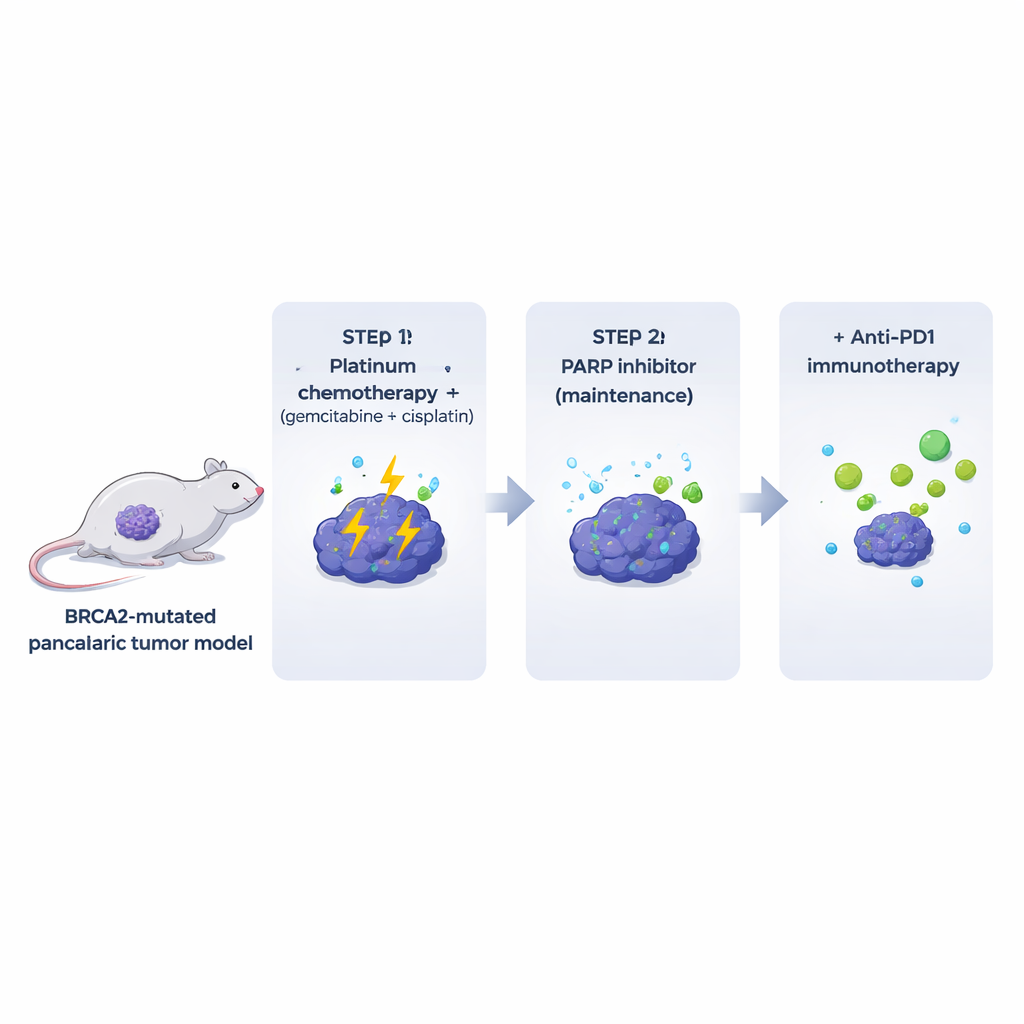
Chemotherapy reshapes the tumor neighborhood
The team then asked what happens inside the tumor after platinum chemotherapy. Ten days after treatment, tumors had shrunk but were not eradicated. Gene‑expression analyses revealed a striking shift toward immune‑related activity: pathways linked to T cell activation and adaptive immunity were turned on. More T cells were found inside treated tumors, and many of these T cells bore markers of exhaustion—signs that they had been activated but were becoming ineffective. Similar patterns of increased T cell presence were observed in human pancreatic cancers with BRCA or related PALB2 mutations after patients received platinum‑based chemotherapy before surgery. At the same time, tumor cells increased levels of PD‑L1, a surface protein that can switch off T cells by engaging the PD‑1 receptor, hinting that the tumors were defending themselves against this new immune pressure.
Why PARP alone is not enough—and where resistance emerges
Mirroring patient care, the researchers gave mice an initial course of gemcitabine and cisplatin, followed by maintenance with the PARP inhibitor olaparib. This sequence improved tumor control and survival compared with chemotherapy alone, confirming that prior chemotherapy can make tumors more susceptible to PARP inhibition. However, no mice were cured: tumors eventually regrew under PARP maintenance. Resistant tumors showed a more differentiated appearance and strongly increased levels of the developmental regulator CDX2. This suggests that, at least in this model, resistance does not simply arise from fixing the original DNA repair defect, but may involve a shift in the tumor’s cellular identity. CDX2 could potentially serve as a biomarker of emerging resistance in patients.
Unlocking immunotherapy with the right sequence
Immune‑checkpoint drugs such as anti‑PD1 antibodies have transformed treatment for several cancers, but pancreatic cancer has remained largely unresponsive. Consistent with this, anti‑PD1 (with or without another checkpoint drug, anti‑CTLA4) failed to help when given to treatment‑naive BRCA2‑mutant tumors, and also did little when simply added to PARP inhibitor therapy. The picture changed when the full sequence was used: platinum chemotherapy first, then PARP inhibition plus anti‑PD1 as maintenance. In this setting, tumors shrank more deeply and mice lived longer than with chemotherapy plus PARP inhibitor or anti‑PD1 alone. Adding anti‑CTLA4 to PARP maintenance did not yield similar benefits, suggesting that the chemotherapy‑primed tumor environment is specifically poised to respond to PD‑1 blockade. 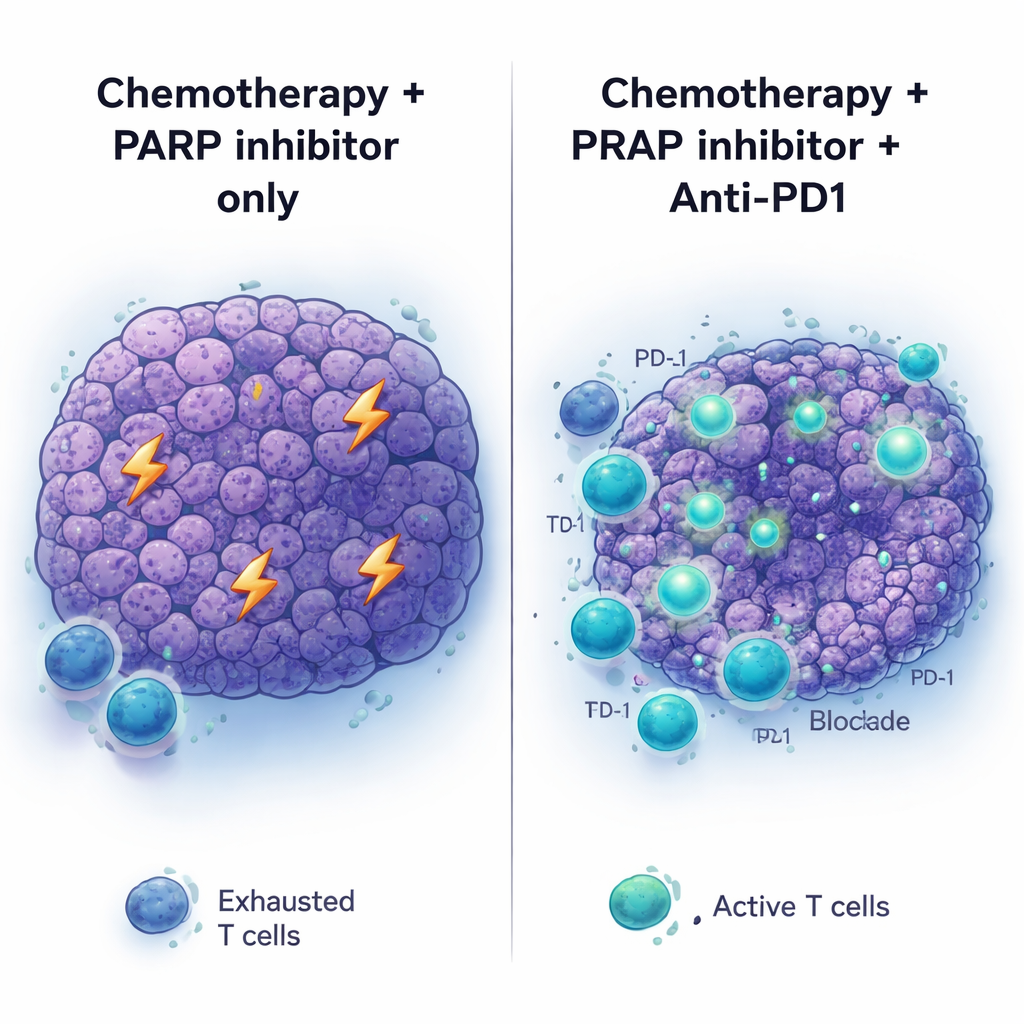
What this could mean for future care
Taken together, the study argues that for BRCA‑mutated pancreatic cancer, the order and combination of therapies matter greatly. Strong DNA‑damaging chemotherapy not only attacks tumor cells directly but also stirs up an immune response, even if that response is initially blunted by exhaustion and PD‑L1 defenses. PARP inhibitors can extend the benefit but are unlikely to be curative on their own and may drive tumors toward a resistant, CDX2‑high state. By layering anti‑PD1 immunotherapy onto PARP maintenance after platinum chemotherapy, it may be possible to convert this briefly inflamed, exhausted immune landscape into a more effective and durable anti‑tumor attack. Ongoing clinical trials combining PARP inhibitors with immunotherapy in BRCA‑related pancreatic cancer will be key to learning whether this carefully timed strategy can translate into longer, better lives for patients.
Citation: McVey, J.C., Wattenberg, M.M., Coho, H. et al. Sequential platinum and PARP Inhibition enhances PD1 immunotherapy efficacy in murine Brca2 mutated pancreatic cancer. Sci Rep 16, 6808 (2026). https://doi.org/10.1038/s41598-026-35423-7
Keywords: pancreatic cancer, BRCA2 mutation, platinum chemotherapy, PARP inhibitor, PD-1 immunotherapy