Clear Sky Science · en
Efficient and sustainable nanocatalyst based on dithiocarbamate ionic liquid functionalized magnetic graphene oxide for green synthesis of 1,4-dihydropyridins
A Faster, Cleaner Way to Make Druglike Molecules
Chemists spend enormous effort finding ways to make useful molecules—especially those related to medicines—without creating unnecessary waste. This paper introduces a new microscopic catalyst that helps build a family of druglike compounds quickly and cleanly, while also being easy to fish out of the reaction with a simple magnet and use again.
Stacked Sheets and Tiny Magnets
The heart of the work is a carefully engineered nanocatalyst, a material built from components only billionths of a meter in size. The base is graphene oxide, a single-atom-thick carbon sheet decorated with oxygen atoms. This sheet offers a huge surface area for reactions. The researchers anchor tiny iron oxide particles to it, turning the sheets into magnetic flakes that can be moved or collected with a magnet. On top of this, they attach a thin layer of an “ionic liquid”, a salt that is liquid-like at relatively low temperatures, and further decorate it with sulfur- and nitrogen‑containing groups called dithiocarbamates. Each part plays a role: the sheet spreads everything out, the magnet allows easy recovery, and the surface groups help grab and activate the reacting molecules. 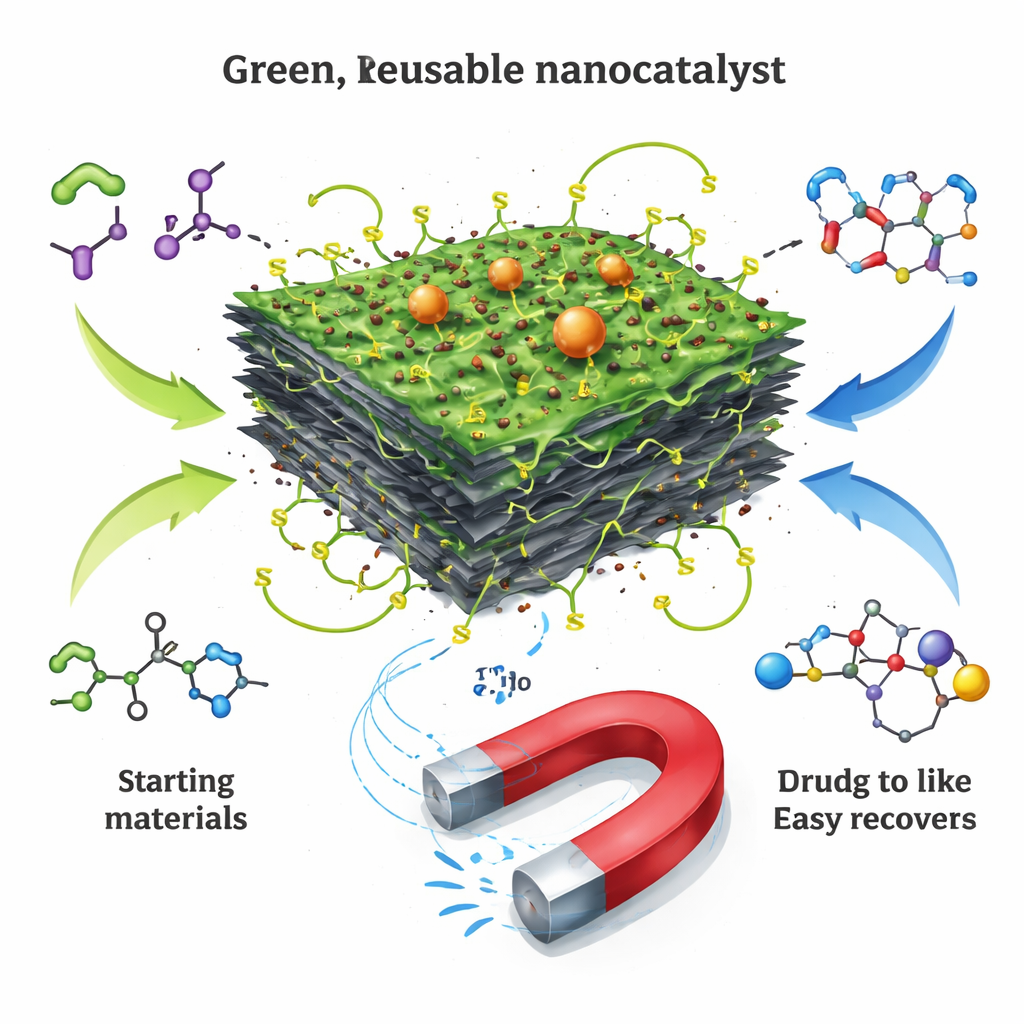
Why This Family of Molecules Matters
The catalyst is used to build 1,4-dihydropyridines, a class of ring-shaped molecules that form the backbone of many widely used drugs. Members of this family help treat high blood pressure and chest pain and are being explored for effects on the brain, cancer, inflammation, and blood sugar. Traditionally, making these compounds in the lab has required harsh conditions, complicated purification steps, or catalysts that are difficult to recycle. A reliable, gentle, high-yield route to these structures can therefore speed up both basic research and the early stages of drug discovery.
A One-Pot Recipe in Green Solvent
The team combines four simple building blocks in one flask: an aromatic aldehyde, dimedone, a β-keto ester (ethyl acetoacetate), and ammonium acetate as a nitrogen source. In ordinary ethanol—a relatively safe, familiar solvent—the new catalyst brings the pieces together into the desired 1,4-dihydropyridine rings. Under mild heating, most reactions finish within 10–15 minutes, delivering isolated yields of 85–95 percent for a broad range of starting aldehydes, including those with electron‑donating or electron‑withdrawing groups. Careful comparison with many previously reported methods shows that this system achieves similar or better yields in shorter times and under gentler, more environmentally friendly conditions.
Built to Last and Easy to Reuse
Because the catalyst is magnetic, separation after the reaction is as simple as holding a magnet to the side of the flask and pouring off the liquid. The authors show that the material can be washed, dried, and reused at least five times with only a minor drop in performance. Laboratory tests using infrared light, thermal analysis, electron microscopy, and magnetic measurements confirm that the structure of the catalyst survives these cycles with little change. A “leaching” study—where the solid is removed halfway through a reaction—demonstrates that almost all catalytic activity stays bound to the magnetic solid, rather than dissolving into the solution. 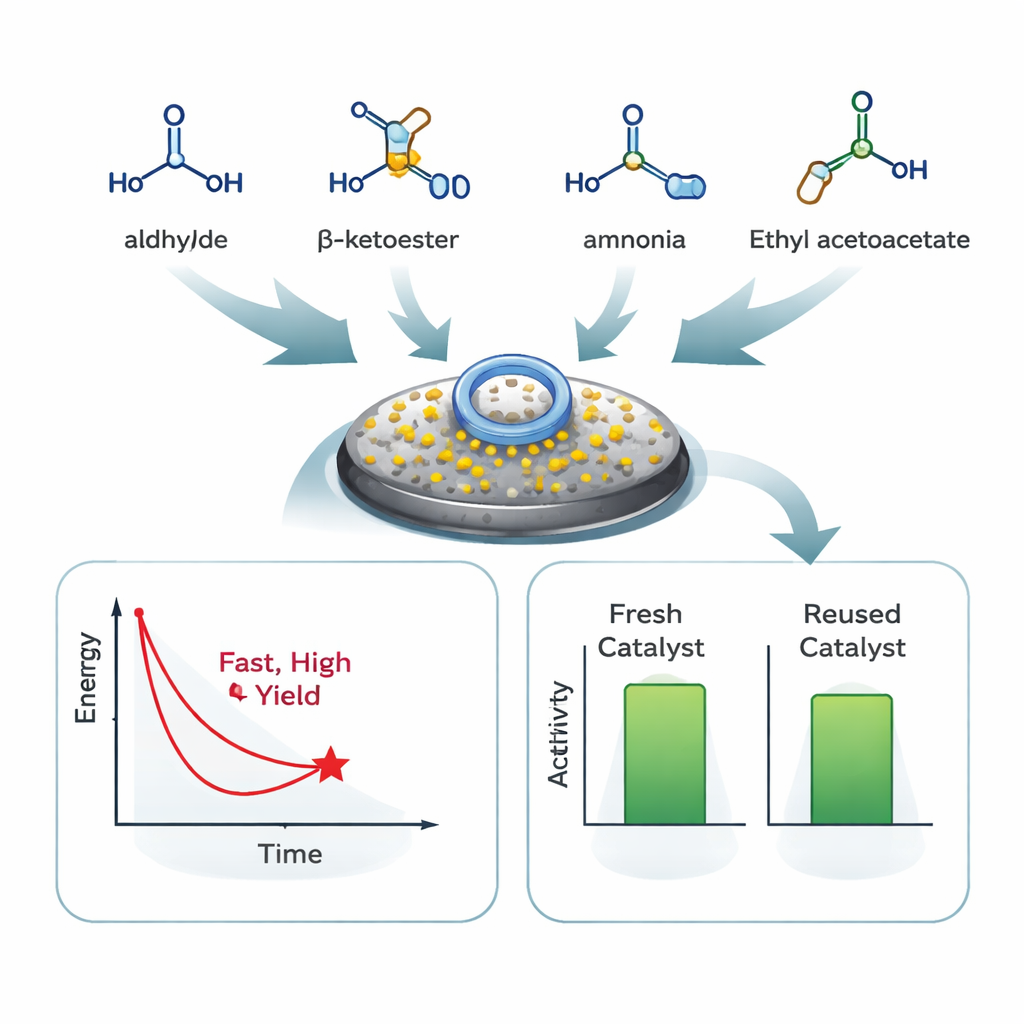
Toward Greener Chemical Manufacturing
For non-specialists, the key message is that the researchers have designed a smart, modular surface that makes an important class of druglike molecules quickly, in a relatively safe solvent, and with minimal waste. The ability to pull the catalyst out with a magnet and use it again and again is particularly attractive for industrial processes, where separation and disposal steps often dominate cost and environmental impact. This work shows how combining advanced carbon materials, magnetic particles, and tailored surface chemistry can bring the ideals of green chemistry—efficiency, reusability, and reduced pollution—closer to everyday practice in the lab and, potentially, in manufacturing.
Citation: Ghorbannia, R., Baharfar, R. & Maleki, B. Efficient and sustainable nanocatalyst based on dithiocarbamate ionic liquid functionalized magnetic graphene oxide for green synthesis of 1,4-dihydropyridins. Sci Rep 16, 7532 (2026). https://doi.org/10.1038/s41598-026-35422-8
Keywords: green chemistry, nanocatalyst, magnetic graphene oxide, ionic liquids, 1,4-dihydropyridines