Clear Sky Science · en
Effects of microplastic on submerged nanofiltration for advanced drinking water treatment
Why tiny plastics in water matter to you
Invisible pollutants are increasingly finding their way into the water we drink. Among them are microplastics—fragments smaller than a grain of pepper—that can carry other contaminants and may pose health risks. At the same time, water utilities are turning to new filtration technologies to provide cleaner tap water. This study looks at how one such advanced filter, a submerged nanofiltration membrane, handles both natural organic substances and added microplastics in real reservoir water, and what that means for the safety and quality of drinking water.
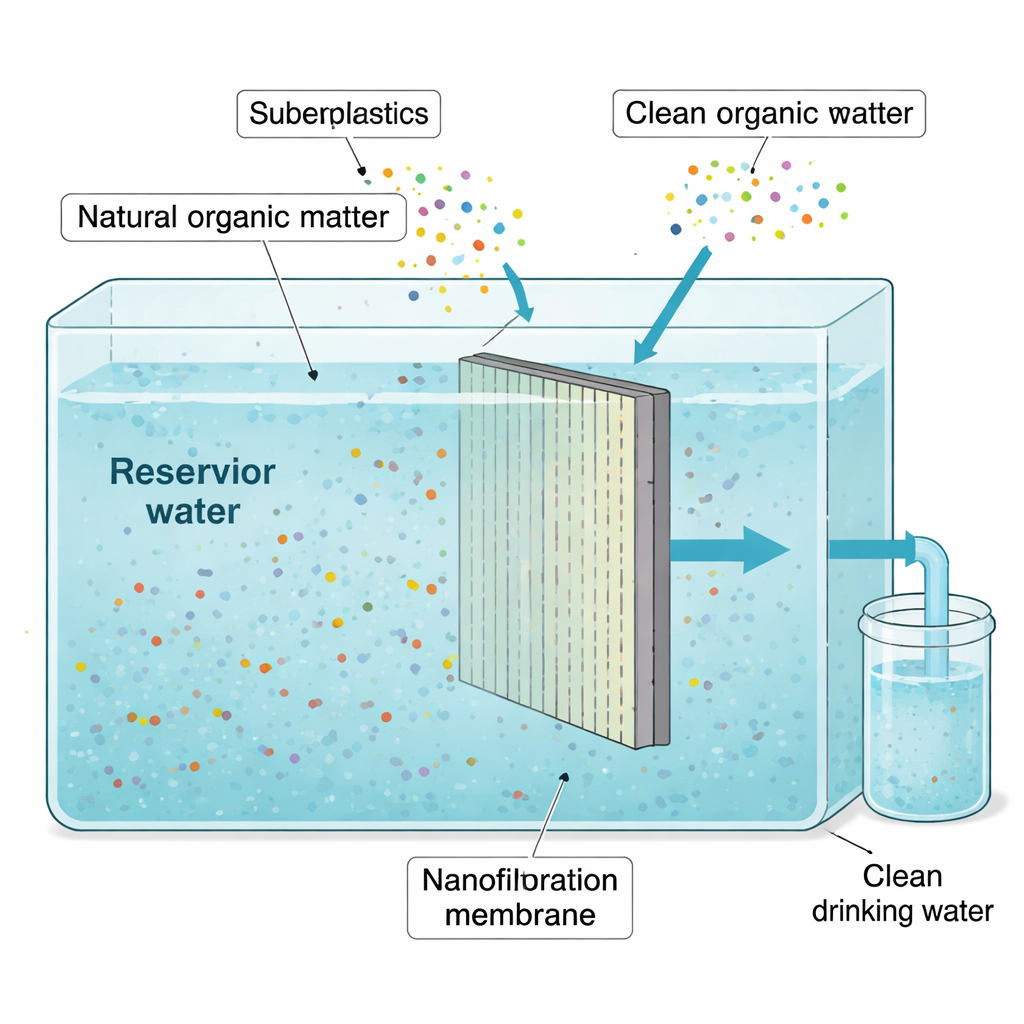
A closer look at a new kind of filter
Traditional drinking water treatment—coagulation, settling, sand filters, and chlorine—does a good job in many cases, but it often leaves behind some natural organic matter. When chlorine reacts with these leftover compounds, it can form unwanted by-products such as trihalomethanes, which are linked to health concerns. Nanofiltration, a pressure-driven membrane process with extremely fine pores, is being explored as an add-on or alternative because it can remove much more of this organic matter. In this research, engineers tested a commercial nanofiltration membrane (called NF270) immersed directly in water from a university reservoir in Thailand, asking how well it could remove dissolved organic material and how its performance changed when microplastics were present.
Simulating real-world microplastic pollution
To mimic contaminated surface waters, the team added polyethylene terephthalate (PET) particles—similar to those from common plastic bottles—to the reservoir water at increasing levels over four filtration cycles: none, then low, medium, and very high microplastic concentrations. Each cycle ran for four days. The submerged membrane was operated at relatively low pressure, and the researchers measured how fast water passed through, how much dissolved organic carbon and light-absorbing organic compounds were removed, and how many salts and dissolved solids were filtered out. They also examined the membrane surface with electron microscopes to see how microplastics and organic material built up over time.
What happens on the membrane surface
Despite the rising microplastic load, the water flow through the membrane stayed fairly stable, with only small changes in flux and pressure. This suggests that, under the tested conditions, microplastics did not rapidly clog the system. Instead, they formed a loose “cake layer” on top of the membrane, together with natural organic matter, sediment, bacteria, and algae. This layer acted like an extra pre-filter, trapping larger polysaccharide-like substances and most of the microplastics themselves. However, the same build-up changed how smaller, more mobile organic molecules behaved near the surface: their concentration right at the membrane increased, making it easier for some of them to slip through the pores.
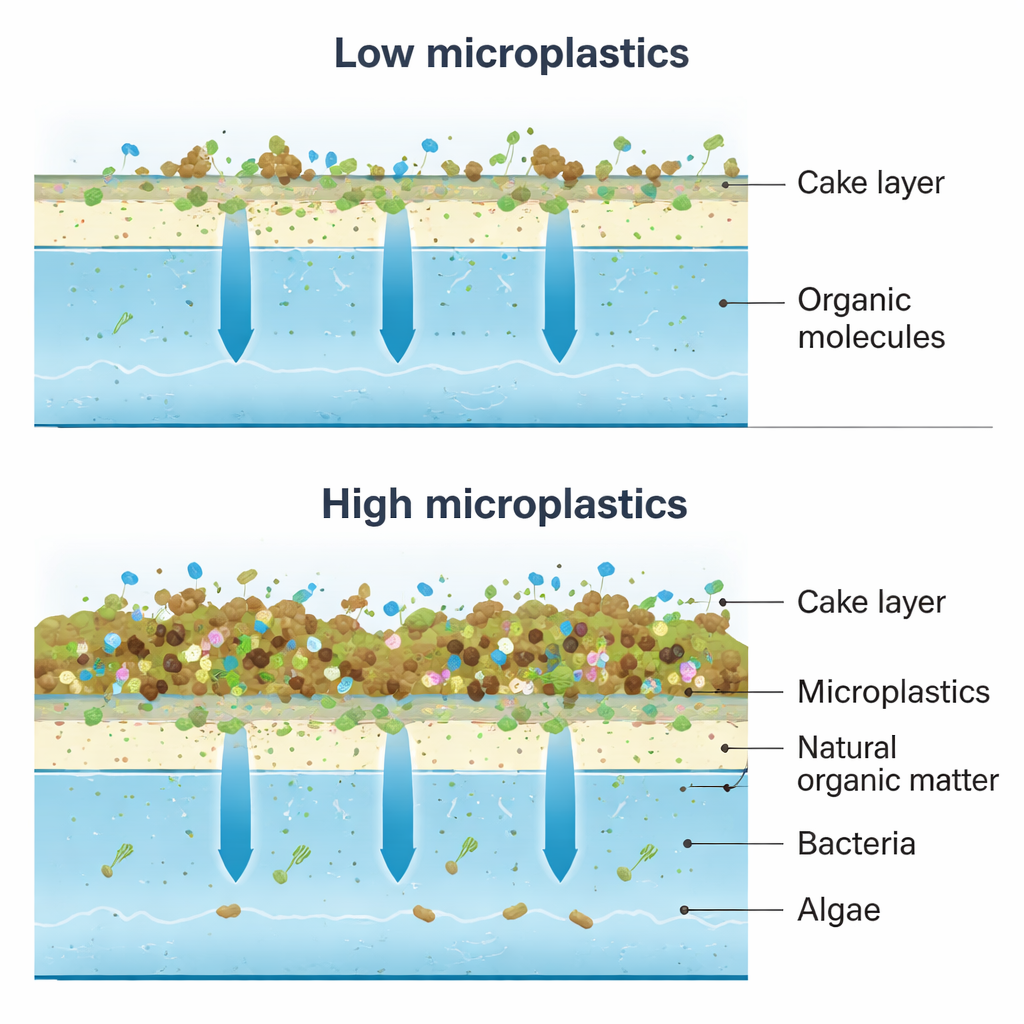
Cleaner water, but changing chemical risks
Overall, the submerged membrane removed dissolved organic matter very effectively, cutting ultraviolet-absorbing compounds by about 90–98% and dissolved organic carbon by roughly 87% across all test conditions. Salts and total dissolved solids were also reduced by about half, keeping the treated water well within World Health Organization guidelines. Yet, as microplastic levels rose, removal of certain organic components dipped slightly, and the remaining organic mix in the treated water became more “reactive” toward chlorine. When the researchers simulated disinfection, the potential to form trihalomethanes per unit of remaining carbon actually increased after filtration, especially when microplastics were present, even though the total amount of organic matter was much lower.
What this means for future drinking water systems
For a layperson, the central message is reassuring but nuanced: this type of submerged nanofiltration can reliably strip out most natural organic matter, salts, and microplastics from reservoir water, even under heavy microplastic contamination, without severe clogging. At the same time, the few organic molecules that do pass through can still react with chlorine to form disinfection by-products, and microplastics may slightly worsen this tendency by altering what reaches the membrane and what remains in the permeate. The study suggests that pairing nanofiltration with careful control of chlorination—and, over the long term, improved membrane cleaning—could offer a powerful way to safeguard drinking water in a world where microplastic pollution is likely to keep rising.
Citation: Kaewjan, T., Sittisom, P., Fujioka, T. et al. Effects of microplastic on submerged nanofiltration for advanced drinking water treatment. Sci Rep 16, 5198 (2026). https://doi.org/10.1038/s41598-026-35398-5
Keywords: microplastics, drinking water treatment, nanofiltration, membrane fouling, disinfection by-products