Clear Sky Science · en
Efficient solvent-free amide synthesis via Ritter reaction catalyzed by a reusable Fe3O4/g-C3N4/ NTMPA nanocomposite
Why cleaner chemistry matters
Many of the medicines we take, the plastics around us, and even high‑performance fibers are built from a simple type of chemical bond called an amide bond. Making these bonds on an industrial scale usually demands strong, corrosive acids and large volumes of solvent, which create waste and safety problems. This article describes a new, magnet‑responsive solid catalyst that can make amides in high yield without using any solvent, pointing toward safer and more sustainable manufacturing of drugs and materials.
A key link in drugs and materials
Amides are the links that hold proteins together and are also found in countless pharmaceuticals, agrochemicals, and polymers such as nylon. Chemists have many ways to build amide bonds, but most methods either need pre‑activated starting materials or harsh conditions. The Ritter reaction stands out because it joins a simple alcohol (or alkene) with a nitrile directly in one step. However, in its classic form the Ritter reaction depends on concentrated mineral acids like sulfuric or hydrochloric acid. These liquid acids are corrosive, hard to separate from products, and difficult to recycle, making them poorly suited to green chemistry.
A tiny magnet you can stir
The researchers designed a solid, magnetically separable catalyst that could replace these liquid acids. Their material combines three components: iron oxide nanoparticles (Fe3O4), which give magnetic properties; a layered carbon‑ and nitrogen‑rich solid (graphitic carbon nitride, g‑C3N4), which acts like a protective support; and a strongly acidic molecule called nitrilotri(methylphosphonic acid) (NTMPA), which supplies the reaction‑driving acidity. These pieces are assembled so that NTMPA is anchored onto the g‑C3N4 surface, while tiny Fe3O4 particles are embedded throughout. Because the whole composite responds to a magnet, it can be pulled out of the reaction mixture simply by placing a magnet outside the flask.
Proving the catalyst’s structure
To confirm that they had built what they intended, the authors used a suite of materials‑science tools. Infrared spectroscopy showed signals from the phosphonic acid groups, the carbon‑nitrogen framework, and the iron–oxygen bonds, all present together in the final composite. X‑ray diffraction indicated that the magnetic iron oxide kept its crystalline form, while the carbon nitride remained as a layered, somewhat disordered solid. Electron microscopy revealed sheet‑like particles decorated with evenly dispersed spheres in the 10–20 nanometer range, and elemental mapping showed iron, carbon, nitrogen, oxygen, and phosphorus spread uniformly. Measurements of surface area and pore size confirmed a mesoporous structure—full of nanoscale channels that allow reactants to reach the active sites—while thermal analysis showed the material remains stable up to several hundred degrees Celsius.
Fast, solvent‑free reactions
With the structure established, the team tested the catalyst in the Ritter reaction between various alcohols and nitriles. They found that the best conditions were surprisingly simple: equal amounts of an alcohol and a nitrile, a small dose of the solid catalyst, heating to 80 °C, and no added solvent. Under these conditions, many different starting materials were converted to their corresponding amides in high to excellent yields, often above 90%. Tertiary and benzylic alcohols (which more easily form the reactive intermediate needed for the Ritter reaction) reacted in as little as 1–4 hours, while more demanding substrates needed somewhat longer. Both aromatic and aliphatic nitriles worked well, with strongly electron‑withdrawing substituents on aromatic nitriles making the reaction even more efficient. Overall, the study demonstrated that the solid catalyst can match or outperform many liquid acids, while avoiding corrosive media and extra solvents.
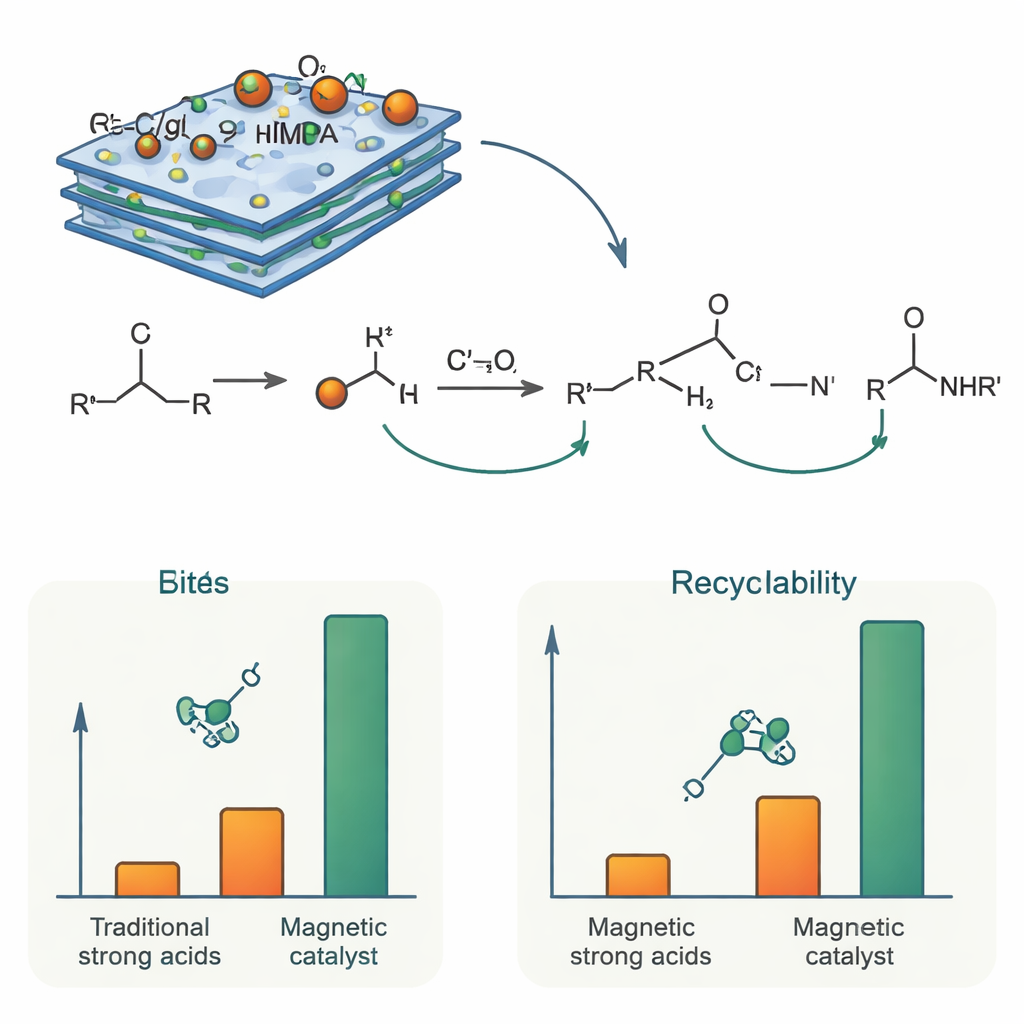
How it works and why it lasts
Chemically, the catalyst’s role is to briefly protonate the alcohol and help it lose water, generating a short‑lived positively charged species. A nitrile molecule then attacks this intermediate to form a new carbon–nitrogen bond, which is finally converted into an amide by reaction with water formed in the process. The phosphonic acid groups in NTMPA provide controlled acidity, strong enough to drive these steps but moderated by the surrounding carbon‑nitride surface so that unwanted side reactions are minimized. Because the active NTMPA units are chemically tethered to the solid support, they do not wash away during reaction. Magnetization measurements confirmed that the particles remain strongly magnetic, allowing the spent catalyst to be rapidly removed with a magnet. In repeat‑use tests, the same batch of catalyst was employed at least six times with only a small drop in activity, and structural analyses after use showed that its composition and morphology were largely unchanged.
What this means for greener manufacturing
For a non‑specialist reader, the key takeaway is that the authors have developed a reusable, magnet‑responsive powder that can help chemists assemble important amide bonds without relying on the harsh liquid acids and extra solvents normally required. This approach cuts down on waste, simplifies product purification, and makes it easier to recycle the catalyst, all of which are important for greener industrial chemistry. While the work focuses on one type of reaction, the same design principles—anchoring strong acid groups on a robust, magnetic support—could be applied to many other transformations that underpin the production of medicines and advanced materials.
Citation: Karimitabar, H., Sardarian, A.R. Efficient solvent-free amide synthesis via Ritter reaction catalyzed by a reusable Fe3O4/g-C3N4/ NTMPA nanocomposite. Sci Rep 16, 6494 (2026). https://doi.org/10.1038/s41598-026-35371-2
Keywords: amide synthesis, Ritter reaction, magnetic nanocatalyst, solvent-free chemistry, green catalysis