Clear Sky Science · en
Rubiadin-1-methyl ether alleviates bleomycin induced pulmonary fibrosis
Why a plant compound for scarred lungs matters
Idiopathic pulmonary fibrosis is a deadly lung disease in which normal, spongy lung tissue is gradually replaced by stiff scar tissue, making every breath harder. Current drugs can slow this scarring but rarely stop it and often cause side effects. This study explores whether rubiadin‑1‑methyl ether (RBM)—a natural compound found in the roots of several common plants—can ease lung scarring in a well‑established mouse model. The work also probes how RBM acts on the immune cells that help drive this damage.
A closer look at scarring in the lungs
In pulmonary fibrosis, the lungs fill with excess connective tissue, known as extracellular matrix, which distorts their delicate architecture. Patients often live only three to five years after diagnosis, and options are limited to drugs that slow decline rather than truly repairing the lungs. Scientists know that a signaling molecule called TGF‑β1 and a process called epithelial‑to‑mesenchymal transition are central to this scarring. But many details—especially how immune cells and plant‑derived molecules influence these pathways—are still being uncovered.
A plant molecule with a medical past
RBM belongs to a family of natural chemicals called anthraquinones, which are found in Rubiaceae plants such as Rubia cordifolia and Morinda species. Compounds in this group have been studied for effects ranging from laxative action to anti‑tumor and anti‑malarial activity. Earlier work showed that RBM can damp down inflammatory signals like nitric oxide and certain interleukins, and can influence fat cell development. Extracts rich in RBM and related molecules were recently shown to blunt lung fibrosis in animals, hinting that RBM itself might be a key active ingredient. Until now, however, its direct impact on lung scarring and its detailed mode of action were unclear.
Testing RBM in a mouse model of lung injury
To mimic human pulmonary fibrosis, the researchers gave mice a single dose of the chemotherapy drug bleomycin into the windpipe, which reliably triggers lung inflammation and scarring. After this injury, animals received different oral doses of RBM (3, 10, or 30 mg per kilogram of body weight). Over three weeks, the team tracked body weight, lung weight relative to body size, and microscopic changes in lung tissue using standard stains that highlight damage and collagen deposits. They also examined lung wash samples to count inflammatory cells and measure key signaling molecules, and used molecular methods to assess genes tied to scarring and to a particular immune cell state known as the M2 macrophage.
Less inflammation, fewer scar markers, and calmer immune cells
The 10 mg/kg dose of RBM emerged as the sweet spot. Mice receiving bleomycin alone lost weight, developed heavy, collagen‑packed lungs, and showed severe distortion of their air sacs. RBM at 10 mg/kg partly reversed these changes: weight loss was reduced, lung structure looked closer to normal, and the amount of collagen‑rich scar tissue fell. In lung wash fluid, RBM cut the total number of inflammatory cells by nearly half, with especially strong reductions in neutrophils, a type of white blood cell linked to acute injury. Genes coding for major scar‑building proteins—fibronectin, type I collagen, and alpha‑smooth muscle actin—were all markedly lower after RBM treatment. Importantly, RBM also reduced levels of inflammatory and pro‑fibrotic messengers such as TGF‑β1, IL‑6, IL‑1β, and TNF‑α, and shifted macrophages away from the M2 state that is known to promote fibrosis. Markers of M2 macrophages on the cell surface and inside the cell both dropped substantially.
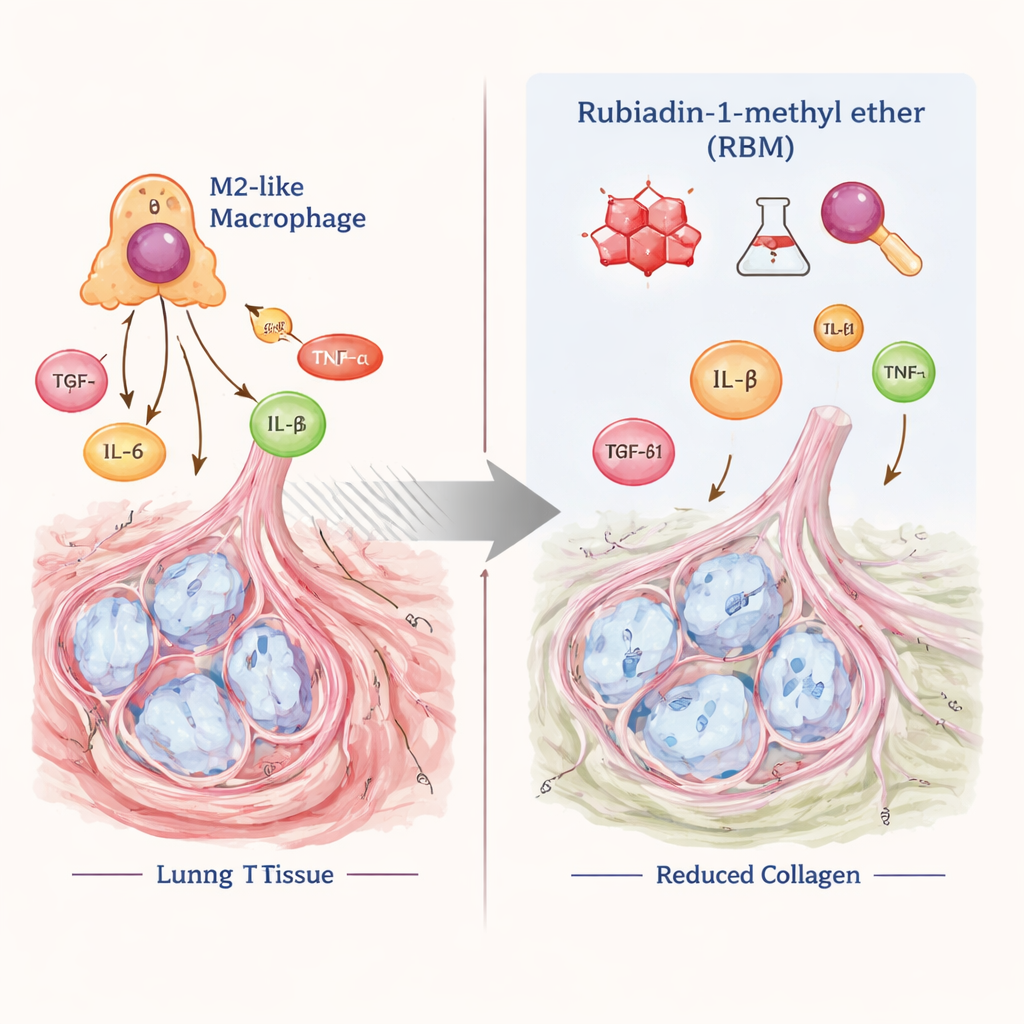
How RBM may be re‑tuning the lung environment
Macrophages are immune cells that can help heal tissue, but in chronic disease they can also push organs toward fibrosis. The M2 type of macrophage, while anti‑inflammatory in some settings, is increasingly recognized as a driver of lung scarring because it secretes TGF‑β1 and other growth factors that stimulate collagen‑producing cells. In this study, RBM appears to act as a brake on this harmful loop: by limiting M2 polarization and lowering TGF‑β1 output, the compound likely weakens the signals that tell lung cells to lay down more scar tissue. The authors suggest that RBM interferes with the TGF‑β1/Smad signaling pathway, a central hub in fibrosis, though they note that more mechanistic work is needed to confirm the exact molecular targets.
What this could mean for future treatments
For non‑specialists, the takeaway is that a naturally occurring plant compound was able to make damaged mouse lungs less inflamed, less scarred, and populated with fewer pro‑fibrotic immune cells. RBM did not cure fibrosis outright, and the highest dose did not work better—possibly because excessive amounts may introduce new stresses to lung tissue. Still, the findings position RBM as a promising candidate for further development as a gentler, plant‑derived therapy for idiopathic pulmonary fibrosis, a condition with few good options. Before it can be tested in people, researchers will need to clarify how the compound is absorbed and processed in the body, test it in larger and more human‑like animal models, and explore whether it can safely boost the effects of existing drugs that slow lung scarring.
Citation: Zhen, X., Xinpeng, L., Jing, S. et al. Rubiadin-1-methyl ether alleviates bleomycin induced pulmonary fibrosis. Sci Rep 16, 4864 (2026). https://doi.org/10.1038/s41598-026-35352-5
Keywords: idiopathic pulmonary fibrosis, macrophage polarization, natural product therapy, TGF-beta signaling, lung fibrosis