Clear Sky Science · en
Identifying the potential anti-lung cancer targets of Baicalein using a network pharmacology approach
Why a plant compound matters for lung cancer
Lung cancer remains the leading cause of cancer deaths worldwide, and many patients eventually stop responding to their medicines. This study explores whether baicalein, a natural substance extracted from the roots of the traditional Chinese herb Scutellaria baicalensis, could help. Instead of testing just one protein or one pathway, the researchers used large biological databases and computer simulations to see how baicalein might act on many targets at once inside lung cancer cells and within the body’s immune defenses.
From ancient remedy to digital investigation
Baicalein has long been known for its anti‑inflammatory and anti‑tumor properties, and experiments in lung cancer models have shown that it can slow cell growth, block invasion, and boost anti‑tumor immunity. But exactly which molecules it binds, and how this translates into benefit for people with lung cancer, has been unclear. In this work, the team combined information from several online resources that list drug‑like compounds and their likely protein partners, and cross‑matched these with thousands of genes linked to lung cancer. This digital filtering narrowed the search to 92 shared targets—proteins that are both related to lung cancer and predicted to interact with baicalein.
Mapping the cancer control network
To understand how these 92 proteins work together, the researchers built a "conversation map" of protein–protein contacts, known as a network. In this map, some proteins sat at busy crossroads, connecting to many others. Ten such hubs stood out as especially important, and five of them—commonly involved in controlling cell growth, death, and stress responses—were highlighted as central to baicalein’s potential action. Many of the connected proteins clustered in a signaling route called the PI3K–AKT pathway. Although the name is technical, the idea is simple: when this pathway is overly active, cancer cells receive strong "stay alive and keep dividing" messages, resist chemotherapy, and are better able to spread.
How baicalein grips its molecular targets
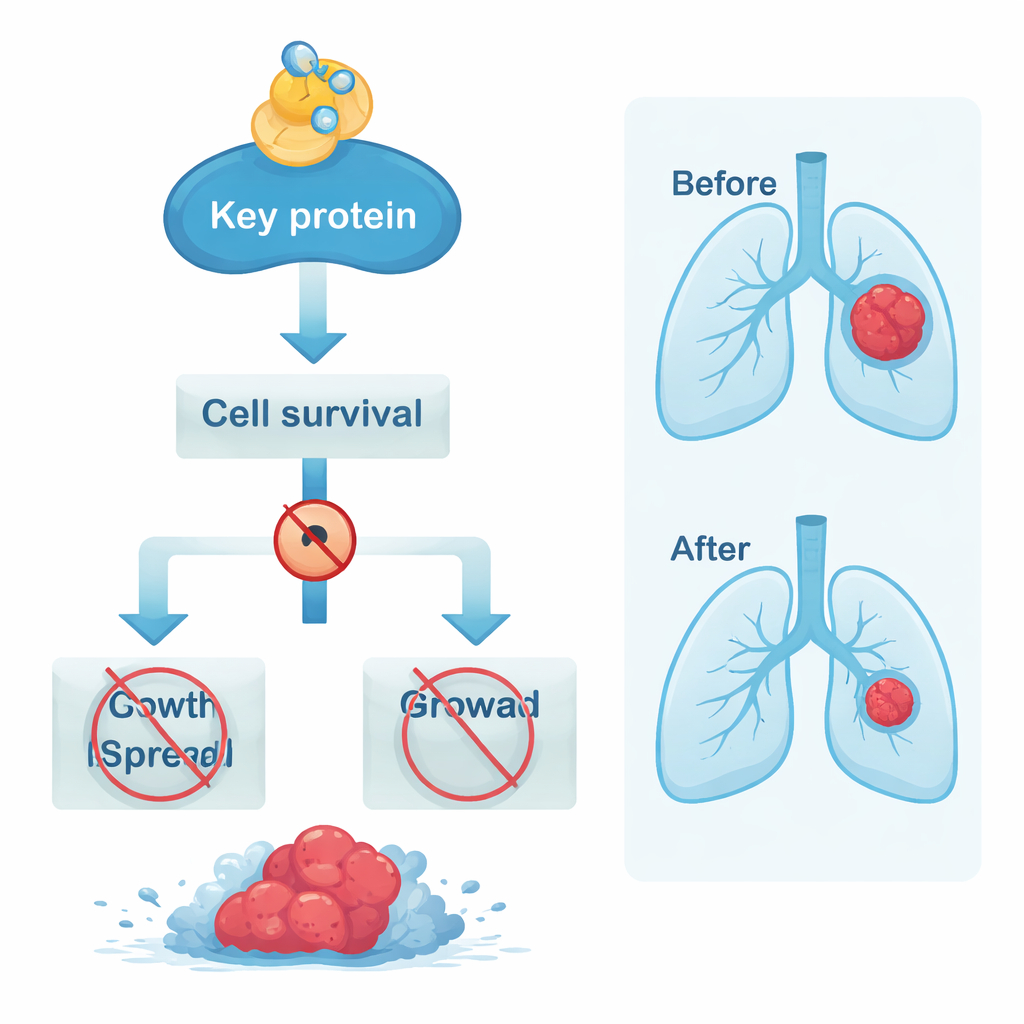
The team then used 3D computer docking, a technique similar to testing how a key fits into a lock, to see whether baicalein could physically bind to these core proteins. All five key targets showed strong predicted binding, but one protein in particular—AKT1, a major switch in the PI3K–AKT pathway—stood out. Baicalein was predicted to latch onto both normal AKT1 and a cancer‑promoting mutant form with especially high affinity. Detailed simulations that mimic the jostling of molecules in a cell over time showed that the baicalein–AKT1 complex remained stable. The analysis suggested that the snug fit and water‑repelling contact surfaces between baicalein and AKT1, rather than classic hydrogen bonds alone, are what keep the complex tightly held together.
Shaping the immune neighborhood around tumors
Beyond tumor cells themselves, lung cancers live within a complex community of immune cells that can either attack or protect the tumor. Using data from hundreds of lung adenocarcinoma samples in a large public cancer database, the researchers estimated the mix of immune cells in and around tumors and compared this with nearby normal tissue. They found that many immune cell types differed between the two, and that the five main target genes linked to baicalein had distinct patterns of mutation and activity in tumors. Certain targets, such as AKT1 and MAPK3, were positively associated with immune cells that often help tumors grow, including specific macrophage types and regulatory T cells. This suggests that by acting on these targets, baicalein might also help to remodel the local immune environment in a more tumor‑fighting direction.
What this could mean for future treatments
In plain terms, this study proposes that baicalein does not work through a single silver bullet, but rather by nudging a whole network of signals that lung cancers depend on—especially the PI3K–AKT route centered on AKT1—and by influencing the immune cells that surround tumors. The findings come entirely from computational and database‑driven methods, so they need to be confirmed in laboratory and animal studies, and eventually in people. Still, they provide a detailed roadmap for where to look next, and suggest that baicalein, alone or combined with existing drugs, might one day help overcome treatment resistance and improve outcomes for patients with lung cancer.
Citation: Chen, X., Chen, K., Ma, X. et al. Identifying the potential anti-lung cancer targets of Baicalein using a network pharmacology approach. Sci Rep 16, 5527 (2026). https://doi.org/10.1038/s41598-026-35351-6
Keywords: lung cancer, baicalein, PI3K-AKT pathway, network pharmacology, AKT1