Clear Sky Science · en
The emerging role of SPHK1 at the immune-metabolic interface: a pan-cancer integrative analysis
Why this hidden cancer switch matters
Cancer is no longer seen as just a mass of runaway cells. Tumors also rewire how they use energy and how they hide from the immune system. This study focuses on a single enzyme, called SPHK1, that appears to sit right at the crossroads of these two processes. By examining data from 33 different cancer types, the authors suggest that SPHK1 may act like a "master dimmer switch" that helps tumors grow faster while at the same time turning down anti-cancer immune responses. Understanding this switch could open new doors for diagnosis and treatment across many cancers, not just one.
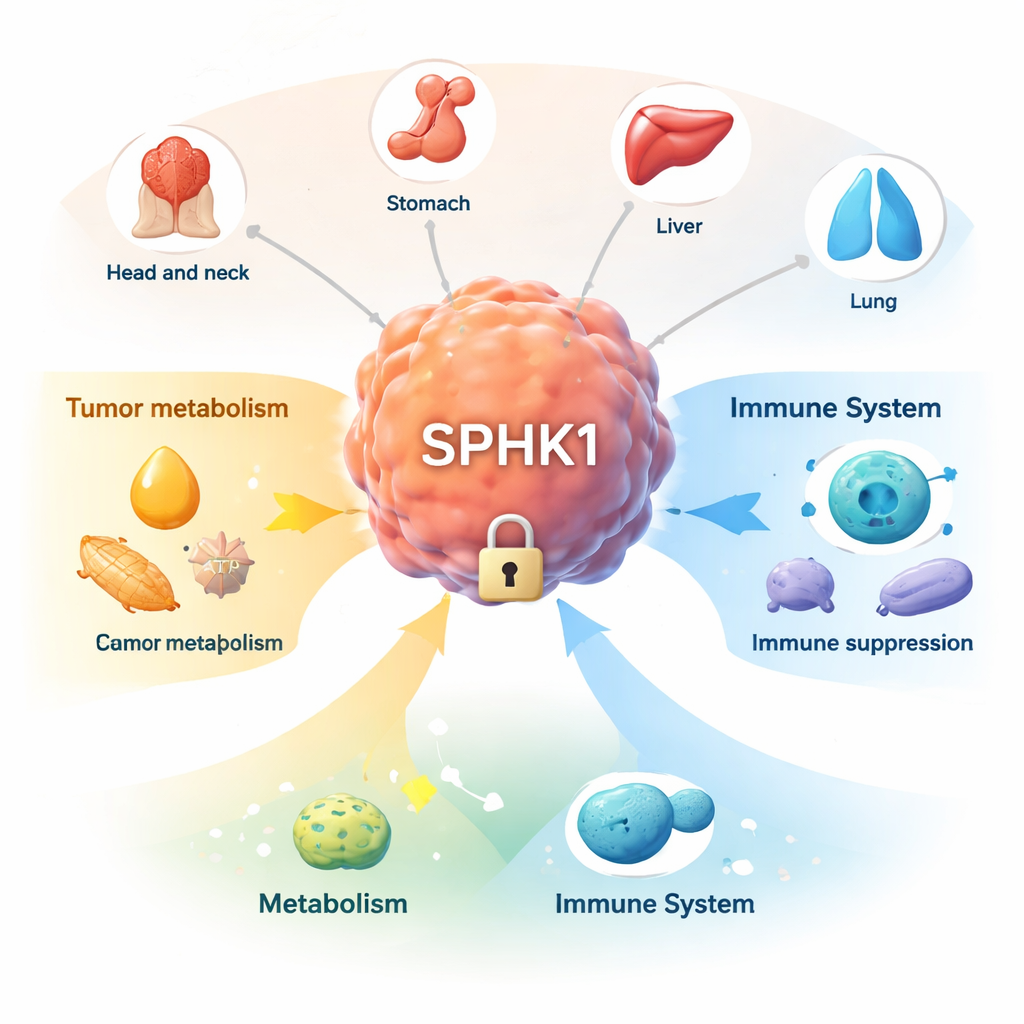
A common thread across many cancers
Instead of studying one tumor type in isolation, the researchers performed a pan-cancer analysis, using large public datasets of tumor and normal tissues. They measured how much SPHK1 is produced in 33 cancers and compared it to nearby healthy tissue. In more than a dozen cancer types – including cancers of the head and neck, stomach, liver, lung, kidney, bladder and colon – SPHK1 levels were clearly higher in tumors. Only one cancer type showed lower levels. The team also examined clinical samples from patients with head and neck, stomach and liver cancers, confirming at both the RNA and protein level that SPHK1 is consistently more abundant in tumor tissue than in normal tissue. When they reduced SPHK1 levels in cancer cell lines grown in the lab, those cells grew more slowly, hinting that tumors may be addicted to this enzyme.
From growth driver to warning sign
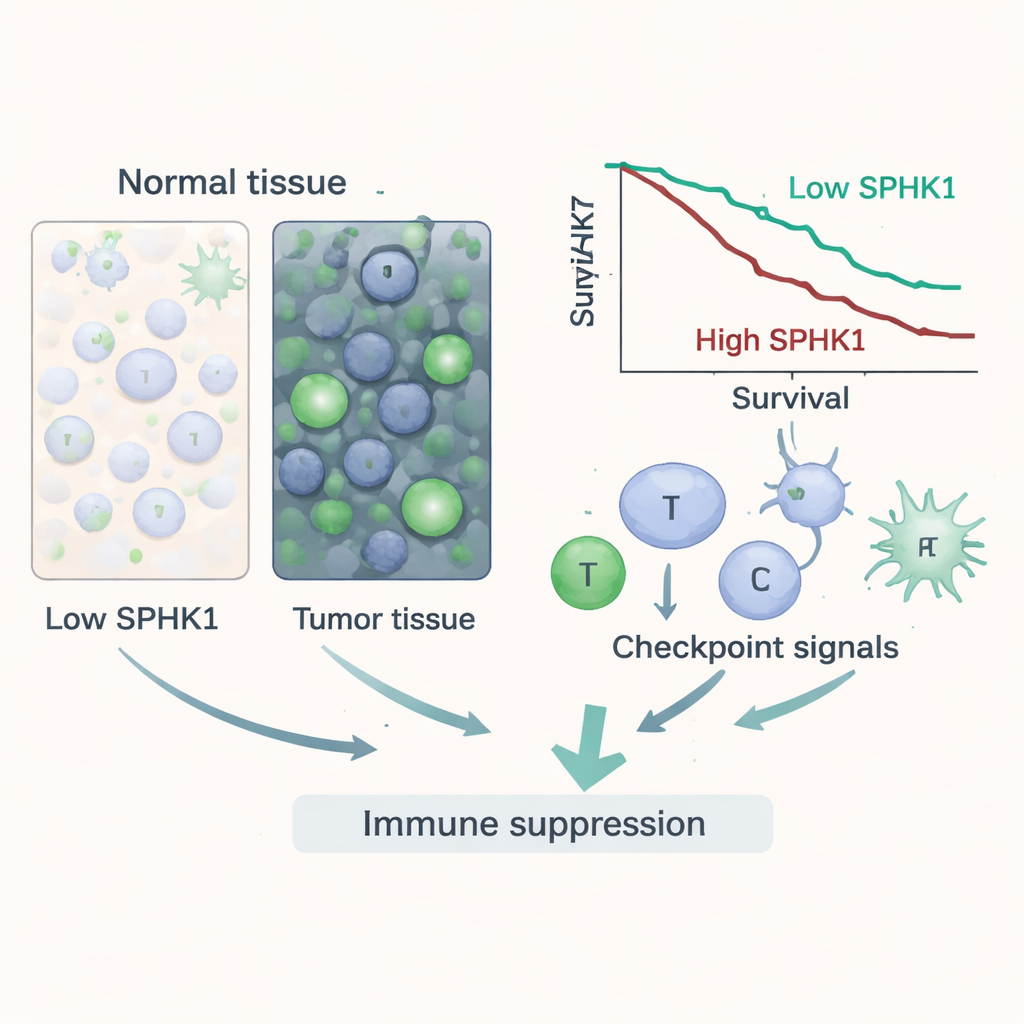
Beyond simple overproduction, SPHK1 carried important information about patient outcomes. Using survival data from thousands of patients, the authors found that higher SPHK1 was often linked to shorter overall survival and faster disease progression in several cancers, such as kidney, liver, lung and certain brain tumors. Even after adjusting for age, tumor stage and grade, SPHK1 remained an independent risk factor in multiple cancer types. In diagnostic tests that measure how well a marker can distinguish cancer from normal tissue, SPHK1 showed strong performance for several tumors, especially bile duct cancer and cancers of the head and neck, stomach and thyroid. Together, these findings position SPHK1 as both a growth promoter and a potential biomarker to help identify high-risk disease.
How tumor fuel and immune escape are connected
SPHK1 controls a balance between two fatty molecules inside cells, tipping the scale toward a signal that favors survival and inflammation. The study shows that when SPHK1 is high, tumors not only grow better but also reshape their surroundings to keep immune cells at bay. Across many cancers, SPHK1 levels tracked with features of genetic chaos such as tumor mutation burden and microsatellite instability, which are often linked to how tumors respond to immunotherapy. More strikingly, tumors rich in SPHK1 tended to harbor immune cells in a state that is less effective at killing cancer. They also showed increased activity of well-known "brakes" on the immune system, including several checkpoint molecules that exhaust T cells. SPHK1 was tied to chemical signals that attract suppressive immune cells and to factors like TGF-β that further dampen immune attack, suggesting that this enzyme helps build an immunosuppressive stronghold around tumors.
Clues for smarter combination treatments
Because SPHK1 sits at the interface of cancer metabolism and immune control, it may shape how tumors respond to drugs. By linking SPHK1 levels with drug sensitivity data from large cell-line panels, the authors found that tumors with more SPHK1 can be more or less sensitive to specific medicines, including some targeted agents and chemotherapy drugs. This pattern was not one-directional, underscoring that SPHK1’s effects depend on context and on the type of therapy used. Still, the results hint that measuring SPHK1 could help guide which treatments might work best and suggest that drugs that block SPHK1, or the signals it produces, could be combined with existing immune checkpoint inhibitors to overcome resistance in certain patients.
What this means for future cancer care
Overall, the study argues that SPHK1 is more than a simple cancer gene; it functions as a "metabolic immune checkpoint" that links how tumors fuel themselves to how they fend off immune attack. For non-specialists, this means that a single enzyme helps cancer both press the gas pedal and cut the brake lines of the immune system. By targeting SPHK1, future treatments might simultaneously slow tumor growth and reawaken immune cells, leading to more effective, tailored immunotherapy strategies across many different cancers.
Citation: Wang, L., Zhong, G., Luo, H. et al. The emerging role of SPHK1 at the immune-metabolic interface: a pan-cancer integrative analysis. Sci Rep 16, 5528 (2026). https://doi.org/10.1038/s41598-026-35350-7
Keywords: cancer metabolism, tumor immune evasion, SPHK1, pan-cancer analysis, immunotherapy