Clear Sky Science · en
Optimization and adsorption mechanisms of vanadium removal by Fe-Pillared bentonite as an efficient adsorbent
Why cleaning vanadium from water matters
Modern industries such as steelmaking and oil refining quietly release a little-known metal called vanadium into water. At high levels, vanadium can be toxic to people and aquatic life, yet it is also a valuable resource worth recovering. This study explores a low-cost way to clean vanadium from water using a naturally abundant clay that has been “propped open” with iron atoms to make it a more effective sponge for pollution.
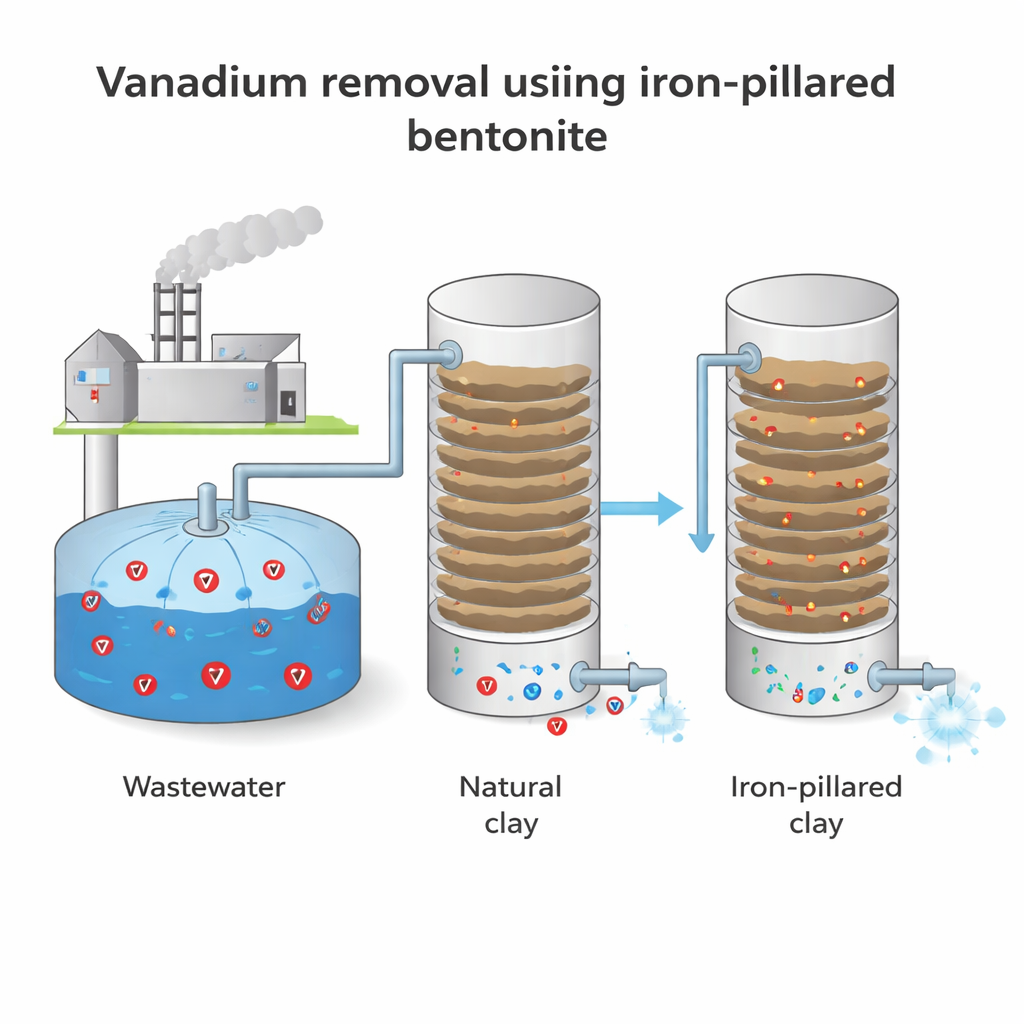
Turning common clay into a smarter sponge
The researchers started with bentonite, a soft clay already used in cat litter, drilling muds and environmental clean-up. On its own, this clay can trap metal ions, but its internal spaces are limited. To boost its performance, the team created “iron-pillared bentonite.” They soaked purified clay in an iron-rich solution and then heated it so that tiny iron oxide clusters formed permanent pillars between the clay layers. Tests using X-ray, infrared light, surface area measurements and electron microscopy confirmed that the layers were pushed farther apart, the surface area grew, and the overall structure became more porous and fluffy. In short, the clay’s internal “apartment building” gained extra floors and bigger hallways for vanadium to occupy.
Testing how well the new material cleans water
Next, the scientists checked how effectively the modified clay removes vanadium from water. They focused on three practical knobs an operator can turn: the water’s acidity (pH), the starting amount of vanadium, and how much clay is added. Using a statistical tool called response surface methodology, they mapped how these factors work together. At the best conditions they found – slightly acidic water (around pH 5.8), low starting vanadium (50 milligrams per liter), and a relatively high clay dose (6 grams per liter) with three hours of contact time – the iron-pillared bentonite removed about 60 percent of the vanadium. That is roughly a 20 percent improvement over the same clay in its natural form, showing that the structural upgrade translates into real-world performance gains.
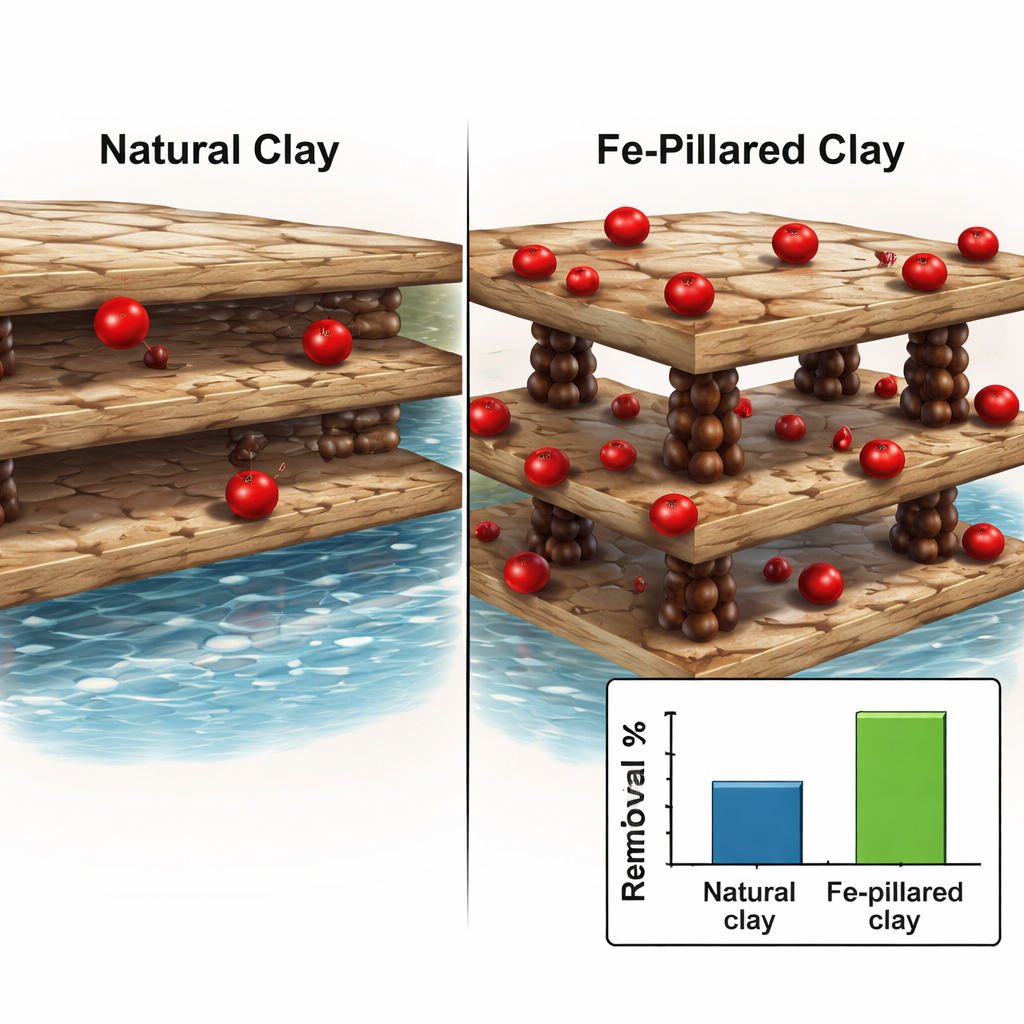
What happens at the tiny scale
To understand what is happening at the microscopic level, the team analyzed how vanadium sticks to the clay surface and how fast this happens. The data match a pattern known as a Langmuir isotherm, which is consistent with a single, organized layer of vanadium ions forming on a fairly uniform surface rather than piling up in random clusters. The time-dependent behavior fits a so‑called second-order kinetic model, which points to a process controlled by chemical bonding, such as ion exchange between vanadium in the water and reactive sites on the iron-pillared clay. Additional tests of energy changes showed that the process is spontaneous (it tends to happen on its own), becomes more favorable at higher temperatures, and slightly increases disorder at the boundary between water and solid, all signs of a robust and efficient adsorption process.
Designing for real-world treatment
The authors also explored how changes in vanadium concentration and clay dose affect removal, using three-dimensional response surfaces to visualize performance. As expected, raising the initial vanadium level eventually overwhelms the available sites on the clay, lowering the removal percentage. Increasing the amount of adsorbent improves removal, but only up to a point where the surface becomes saturated. The optimal pH window arises because both the charge on the clay and the chemical form of vanadium change with acidity; in the identified range, the surface of the iron-pillared bentonite is negatively charged and strongly attracts positively charged vanadium species. Together, these insights provide a guide for engineers to tune treatment systems for different industrial wastewaters.
What this means for safer, cleaner water
In accessible terms, this work shows that a cheap, natural clay can be smartly re-engineered with iron to become a better magnet for a problematic metal in water. By propping open the clay layers and creating new chemical “grab points,” the iron-pillared bentonite removes more vanadium than raw clay and does so in a predictable, controllable way. While it does not eliminate vanadium completely, it offers a promising, low-cost step toward cleaner industrial effluents and easier recovery of a useful metal, especially in regions where advanced treatment technologies are too costly or complex to deploy.
Citation: Etaati, A., Soleimani, M. Optimization and adsorption mechanisms of vanadium removal by Fe-Pillared bentonite as an efficient adsorbent. Sci Rep 16, 4915 (2026). https://doi.org/10.1038/s41598-026-35348-1
Keywords: vanadium removal, iron pillared bentonite, water treatment, metal adsorption, industrial wastewater