Clear Sky Science · en
Sustainable α-AO@CS composite for effective humic acid elimination from water
Why cleaning "natural" water is harder than it looks
Even crystal-clear lakes and rivers contain invisible brownish organic matter that can make drinking water harder and more expensive to treat. One major culprit is humic acid, a complex mixture formed as plants and microbes break down. It is harmless at low levels but, in large amounts, it stains water, interferes with disinfection, and can lead to potentially harmful by-products when chlorine is used. This study reports a new, eco-friendly material made from alumina and chitosan that traps humic acid more efficiently and can be reused many times, offering a practical way to improve drinking water safety.
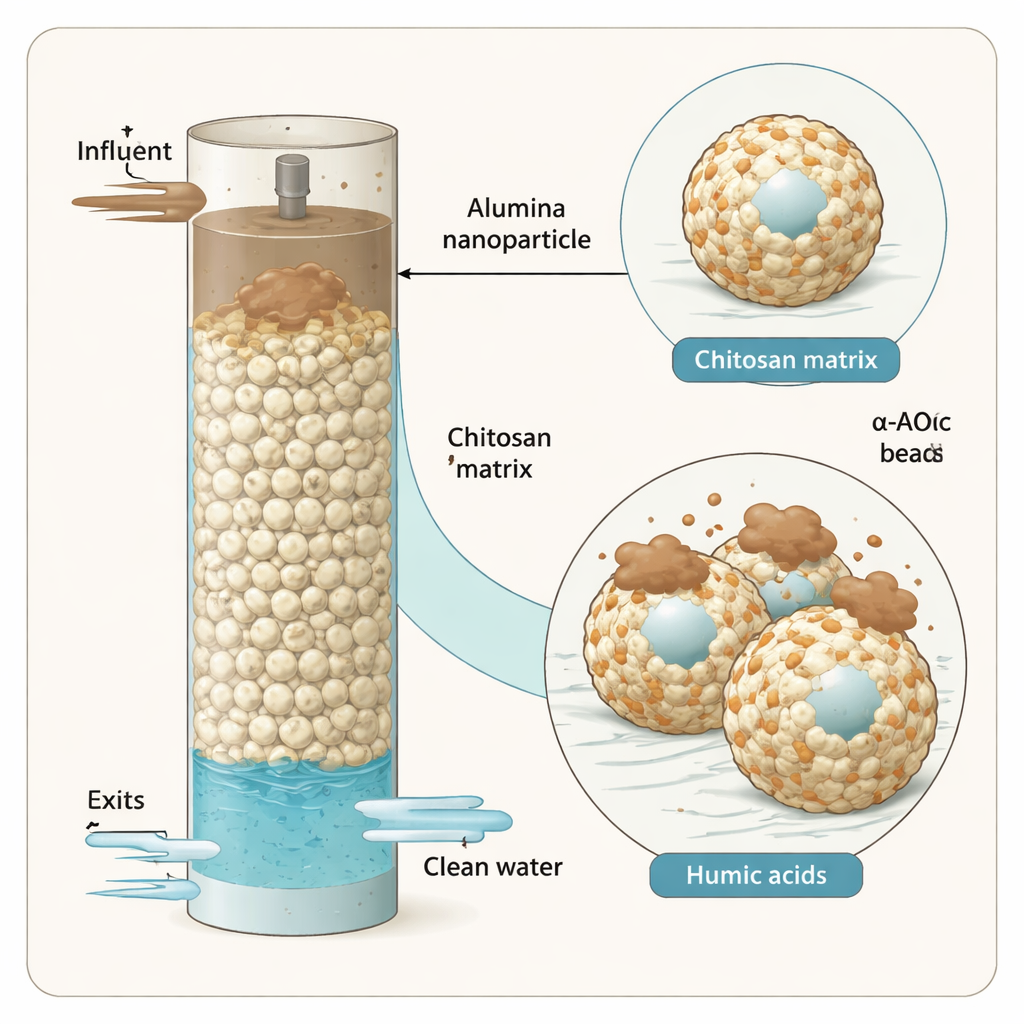
The hidden troublemaker in brown-tinted water
Humic acid belongs to a family of natural substances that give some waters a tea-like color. Because it carries many reactive chemical groups, it can grab onto metals, react with disinfectants, and shield germs from ultraviolet light. When utilities chlorinate water that contains a lot of humic acid, they often generate disinfection by-products such as trihalomethanes, some of which are linked to cancer risk. Standard treatment methods like coagulation, membranes, or advanced oxidation can remove humic acid, but they may be costly, energy-intensive, or produce extra waste. Adsorption—using a solid material that selectively captures contaminants—offers a simpler, potentially cheaper route if the right adsorbent can be designed.
Building a greener sponge for dirty water
The researchers created millimeter-sized beads made from chitosan, a biodegradable polymer obtained from crustacean shells, and alumina, a common aluminum oxide powder. They mixed alumina nanoparticles into a chitosan solution and then dripped this mixture into an alkaline bath so that solid beads formed, locking the particles inside a flexible matrix. These beads were then washed, dried, and optionally cross-linked to improve strength. A suite of analytical tools—including infrared spectroscopy, X-ray diffraction, electron microscopy, and surface charge measurements—confirmed that alumina was uniformly embedded in the chitosan and that the resulting composite had a stable, heterogeneous surface well-suited for binding humic acid.
How well the new beads clean water
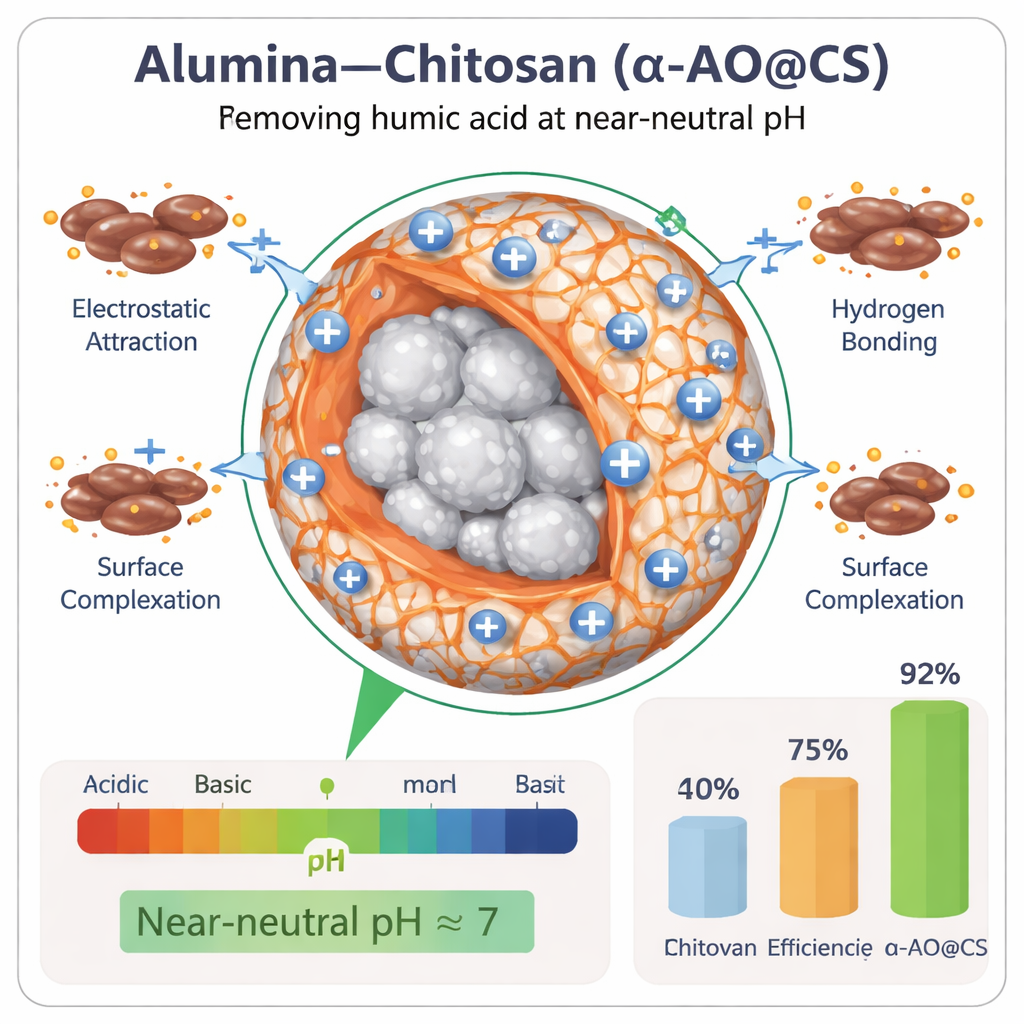
To test performance, the team shook the alumina–chitosan beads with humic-acid-containing water under different conditions of pH, contact time, dosage, temperature, and competing ions such as calcium and magnesium. At a pH close to that of natural waters (around 7), the new beads removed about 91.7% of humic acid, clearly outperforming plain alumina (49.2%) and pure chitosan (74.9%). The material kept working effectively over a broader pH range than either ingredient alone and still showed strong removal even when natural background salts and other organic matter were added. Mathematical models of how quickly and how strongly humic acid attached to the beads pointed to a chemically driven process rather than simple physical trapping, and showed that the composite behaves as a surface with many different types of binding sites.
Why the bead chemistry matters
The key to the composite’s success lies in how charge and chemical groups line up at the water–solid interface. At near-neutral pH, the bead surface carries a slight positive charge, while humic acid molecules are mostly negative, encouraging electrostatic attraction. Alumina contributes hydroxyl groups that can form tight surface complexes with humic acid, while chitosan provides amino and hydroxyl groups that participate in hydrogen bonding and additional charge-based interactions. Temperature-dependent tests showed that adsorption is spontaneous and slightly heat-favored, and detailed modeling indicated that humic acid tends to form more than a single uniform layer on the bead surface. Importantly, after five cycles of adsorption and cleaning with a mild alkaline solution, the beads still retained about 83% of their original capacity, much better than alumina or chitosan alone, which lost more than half their effectiveness.
From lab bench to real-world taps
For non-specialists, the bottom line is that combining a common mineral (alumina) with a natural biopolymer (chitosan) yields robust, sand-sized beads that can strip troublesome natural organics from water near drinking-water pH, and then be regenerated and reused. The beads work better than either ingredient alone, tolerate realistic water chemistries, and can be handled easily in filters or packed columns without the dust and recovery issues of fine powders. While full-scale economic and engineering studies are still needed, this work suggests a promising, scalable, and environmentally friendly option for making brown, humic-rich waters safer to drink.
Citation: Al-Mur, B.A., Jamal, M.T. Sustainable α-AO@CS composite for effective humic acid elimination from water. Sci Rep 16, 5529 (2026). https://doi.org/10.1038/s41598-026-35347-2
Keywords: humic acid removal, water purification, chitosan composite, alumina adsorbent, natural organic matter