Clear Sky Science · en
Preparation and characterization of low-cost chemically activated carbons using H3PO4, ZnCl2 and KOH for CO2 adsorption applications
Turning Waste Wood into Climate Helpers
Rising carbon dioxide (CO2) levels are a main driver of global warming, and much of this gas comes from power plants and factories that burn fossil fuels. Capturing CO2 before it reaches the atmosphere is one promising way to slow climate change, but current methods often use costly or corrosive liquids. This study explores a simpler idea: transforming low-value Persian ironwood, a common tree in northern Iran, into highly porous charcoal-like materials that can trap CO2 efficiently and cheaply.
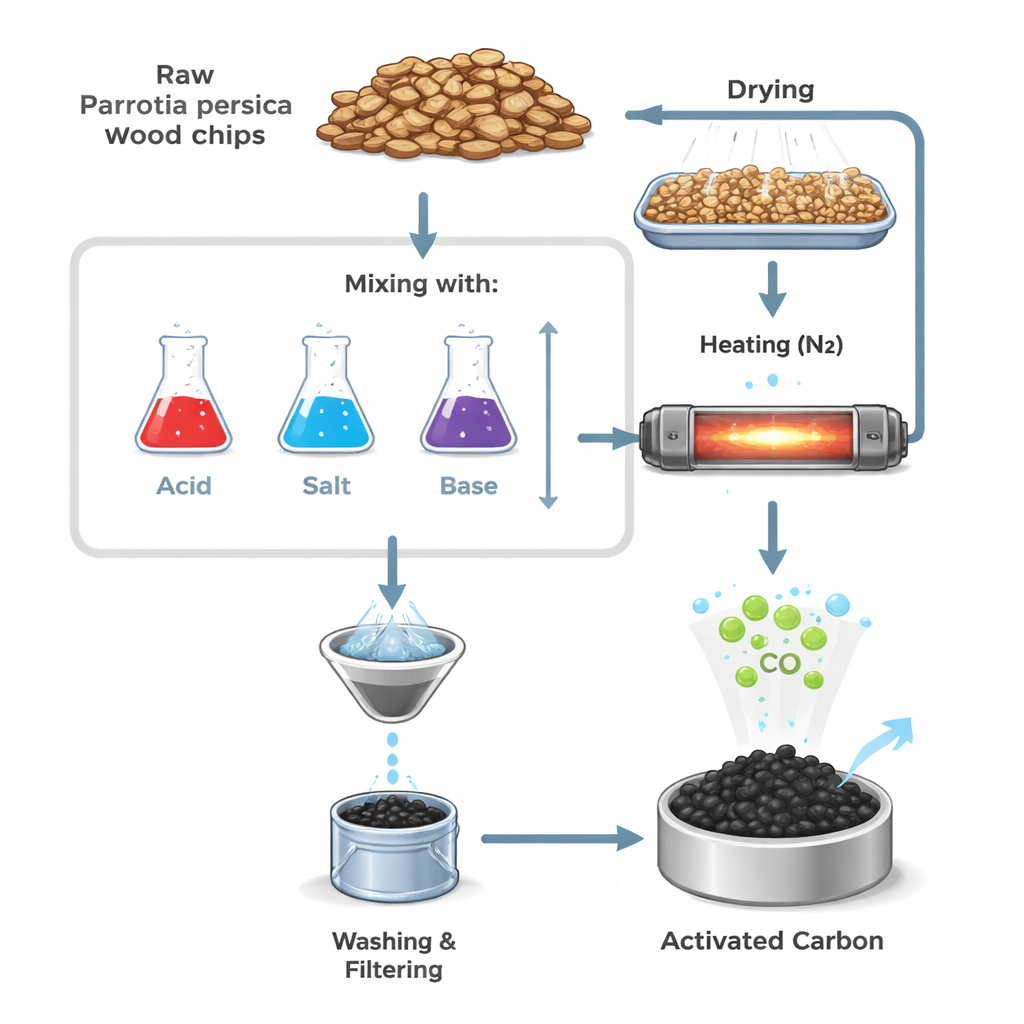
From Forest Byproduct to Engineered Charcoal
The researchers started with Parrotia persica wood, which grows abundantly in the Hyrcanian forests yet has low commercial value. After cleaning and grinding the wood, they heated it in the absence of oxygen to turn it into a carbon-rich solid, then treated it with three different chemicals: a strong acid (phosphoric acid), a zinc salt (zinc chloride), and a strong base (potassium hydroxide). Each chemical interacts differently with the wood’s natural polymers during heating, carving out a network of tiny pores and tuning how much surface area is available for gas to cling to. By varying how much chemical they used and the heating temperature, they created a family of activated carbons with distinct pore structures.
Designing Tiny Cavities for CO2
Why do pores matter? Gas capture on solid materials works because gas molecules stick to surfaces through weak electrical forces. The more internal surface area and the more appropriately sized cavities a material has, the more molecules it can hold. The team measured pore size and surface area with nitrogen gas and microscopy. Zinc chloride produced the highest surface area—about 1,925 square meters per gram, comparable to spreading a tennis court on a sugar cube. Phosphoric acid, however, created carbons with especially large pore volumes and a mix of ultra-small and slightly larger pores, as well as many oxygen-containing surface groups. These chemical features strengthen interactions with CO2, which is slightly electrically uneven and attracted to polar or basic sites on the carbon surface.
How Well Do These Sorbents Capture CO2?
The researchers tested CO2 uptake at pressures up to 14 bar and near room temperature, conditions similar to those in industrial exhaust streams. All samples showed the strongest CO2 uptake at lower temperatures, consistent with a physical “sticking” process: as gas molecules warm up, they move faster and are less likely to remain attached. Among the materials, the phosphoric-acid-activated carbon with the highest treatment ratio (labeled ACH3) achieved the best CO2 capacity at 1 bar and 25 °C, slightly outperforming the zinc-chloride sample despite its slightly lower surface area. This edge came from its larger pore volume and richer surface chemistry. Analysis of the heat released during adsorption confirmed that CO2 was held mainly by physical forces rather than by forming new chemical bonds, which is important because it means the material can be regenerated with modest heating and reused many times.
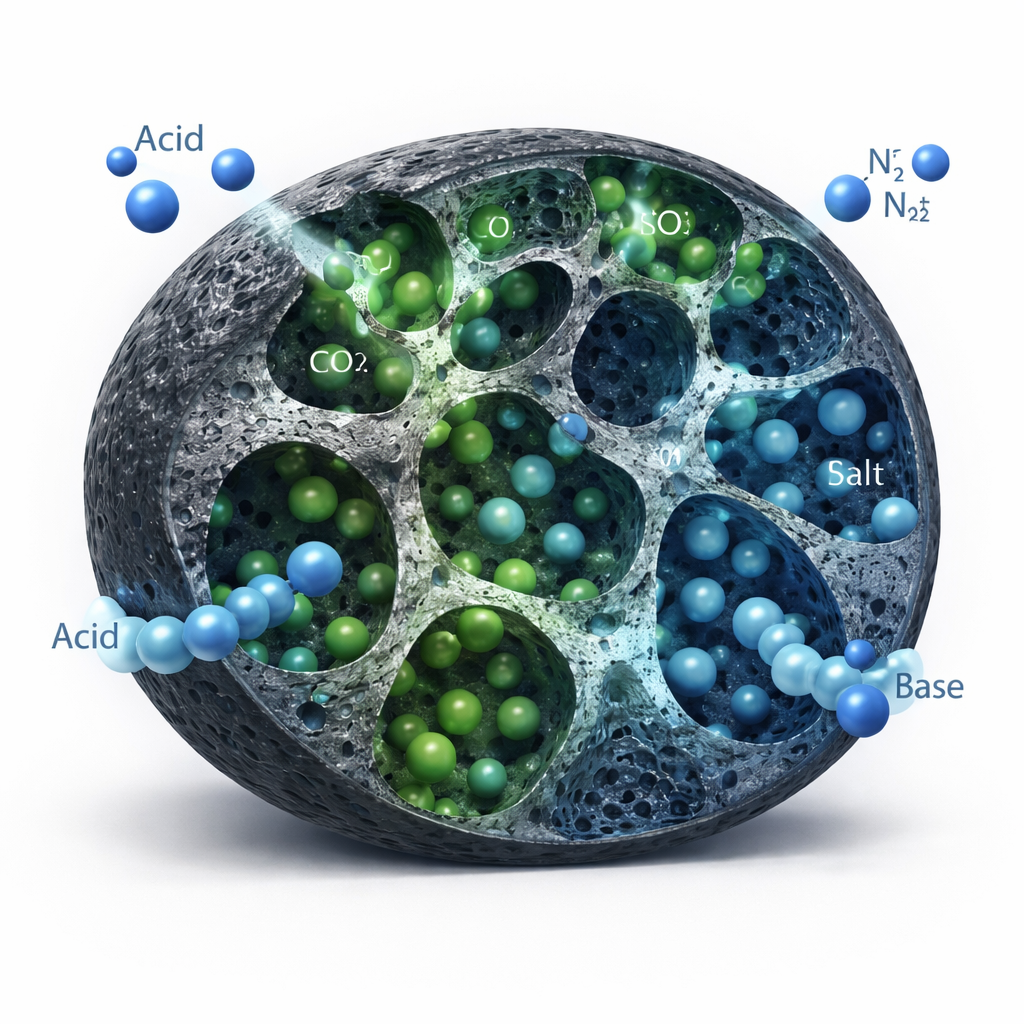
Separating CO2 from Ordinary Air Components
Capturing CO2 from flue gas is not just about how much it can hold, but how well it prefers CO2 over other gases such as nitrogen (N2), which makes up most of the air. By combining measurements of how each gas behaves on the carbons with a well-established predictive theory, the team estimated how selectively the materials would adsorb CO2 from a CO2/N2 mixture. Both phosphoric-acid and zinc-chloride carbons showed strong selectivity, favoring CO2 over N2 by roughly a factor of 20 at atmospheric pressure. The potassium-hydroxide-based sample was less selective, likely because its pore network was more coarsely etched and partially blocked, giving fewer ideal-sized sites for CO2 to nestle into. Importantly, all of the best-performing carbons maintained nearly constant performance over several adsorption–desorption cycles, suggesting they could withstand repeated use in real systems.
What This Means for Future CO2 Capture
To a non-specialist, the take-home message is straightforward: a low-value forest byproduct can be upgraded into a finely tuned, sponge-like carbon that captures CO2 efficiently, prefers it strongly over nitrogen, and can be reused multiple times. Among the tested approaches, phosphoric-acid and zinc-salt treatments of Persian ironwood produced especially promising materials, balancing high surface area, well-matched pore sizes, and favorable surface chemistry. While the study’s predictions of gas-mixture performance still need verification in full-scale flow tests, the work shows that carefully engineered “charcoal” from local biomass could become a practical, low-cost tool for cutting industrial greenhouse gas emissions.
Citation: Bandani, M., Najafi, M., Khalili, S. et al. Preparation and characterization of low-cost chemically activated carbons using H3PO4, ZnCl2 and KOH for CO2 adsorption applications. Sci Rep 16, 6288 (2026). https://doi.org/10.1038/s41598-026-35319-6
Keywords: carbon capture, activated carbon, biomass, CO2 adsorption, porous materials