Clear Sky Science · en
EEG-based classification of alzheimer’s disease and frontotemporal dementia using functional connectivity
Why brainwave patterns matter
Dementia affects millions of families, but even doctors can struggle to tell different forms apart. Alzheimer’s disease and frontotemporal dementia often look similar in clinics, yet call for different care plans and research trials. This study asks a simple but powerful question: can we use a quick, non-invasive brain test—electroencephalography, or EEG—to read the brain’s communication patterns at rest and help distinguish these conditions from healthy aging, and from each other?
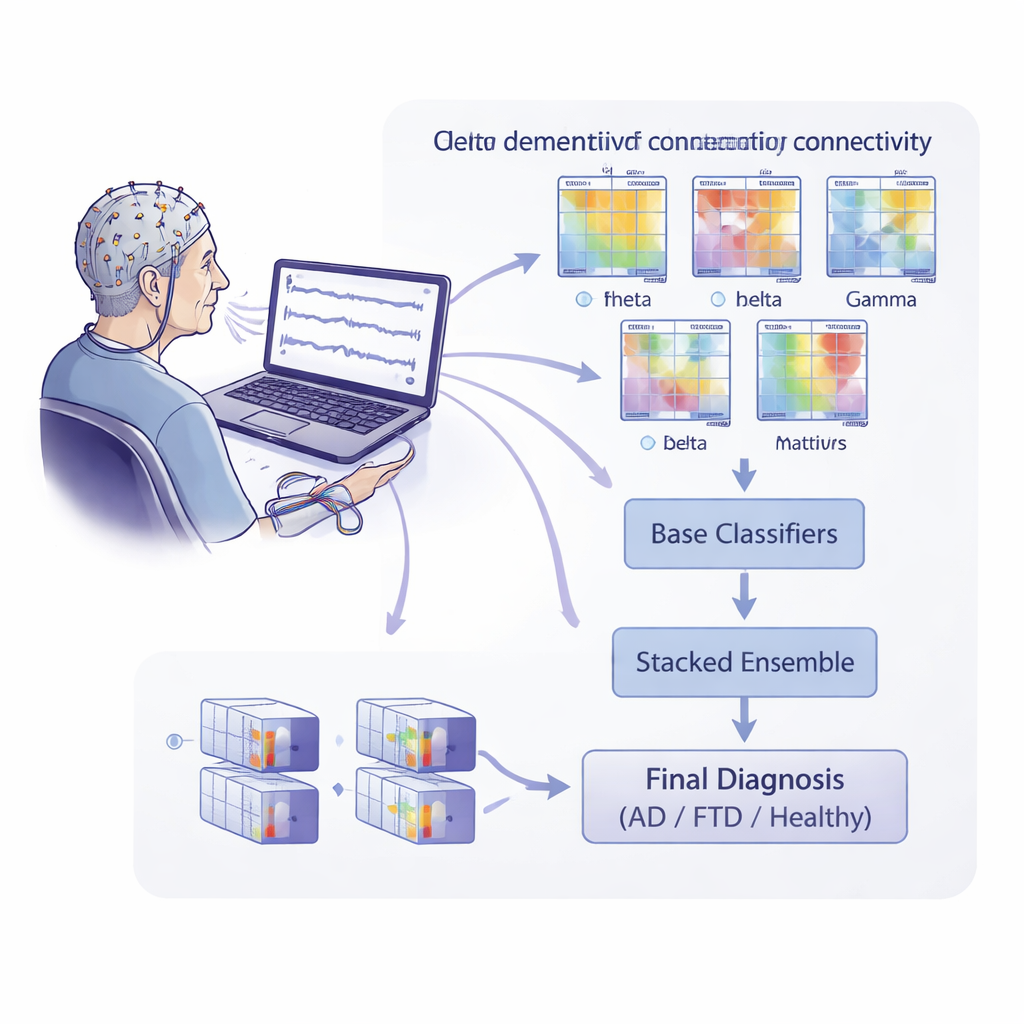
Looking at brain conversations, not just brain activity
EEG usually measures how strong brain waves are in different frequency bands, such as slow delta waves or faster alpha waves. Here, the researchers went a step further and examined how different parts of the brain “talk” to each other. They used recordings from 88 people: 36 with Alzheimer’s disease, 23 with frontotemporal dementia, and 29 healthy older adults. With 19 scalp electrodes, they recorded brain activity at rest with eyes closed, then split each recording into many short time segments. For each segment and for several frequency bands, they calculated a series of connectivity measures—mathematical descriptions of how tightly different EEG channels are linked in time, frequency, and phase.
Teaching algorithms to spot dementia patterns
To turn these connectivity maps into predictions, the team trained a large set of basic machine-learning models, each focused on one combination of frequency band and connectivity measure. These base models used specialized mathematical tools to compare connectivity patterns across individuals. The output of all base models was then fed into a higher-level “stacked” model that tried to learn which combinations of features were most informative. Crucially, the researchers evaluated performance at the level that matters clinically: a single diagnosis per person. They used a strict testing scheme that always kept one person’s data entirely separate from the training data, reducing the risk of overly optimistic results due to subtle data leakage.
What the brainwaves revealed—and what they did not
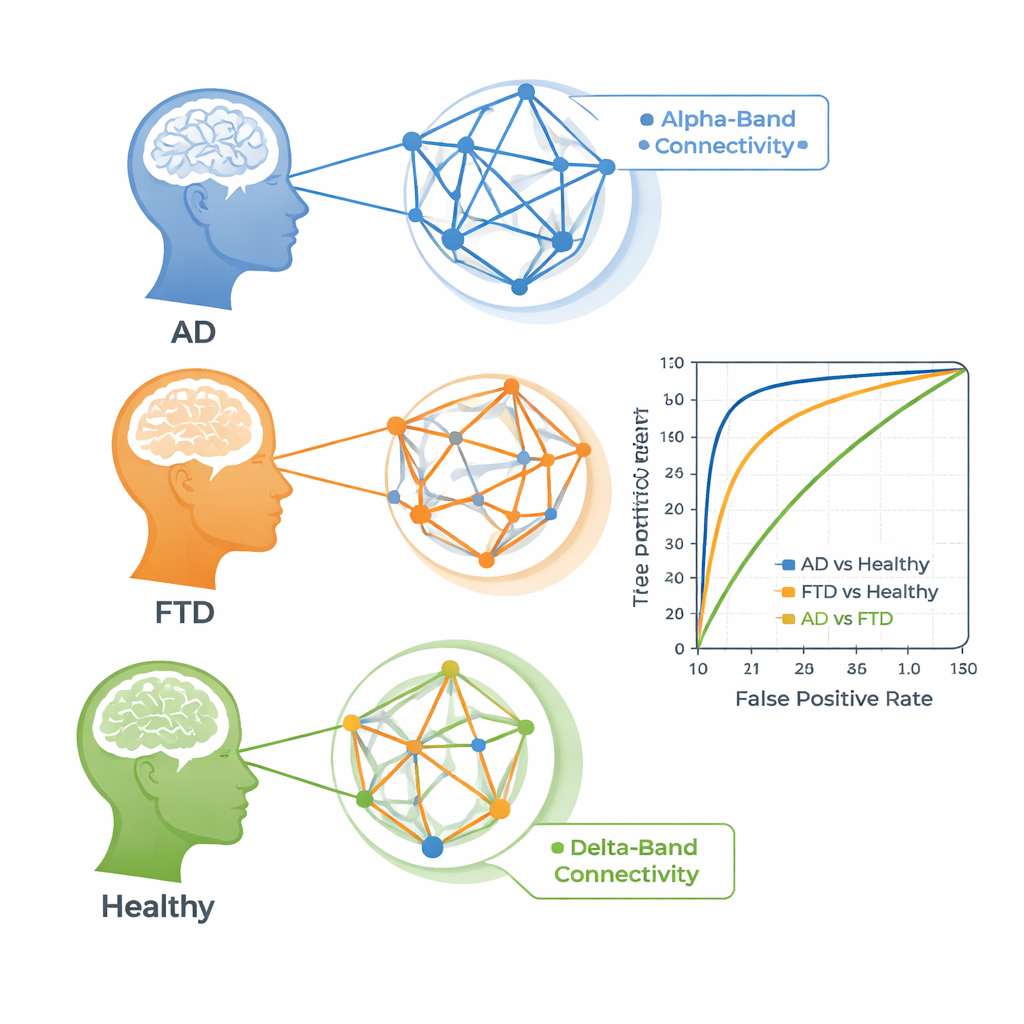
The connectivity patterns did help separate people with dementia from healthy volunteers. For Alzheimer’s versus healthy controls, some individual connectivity features achieved area-under-the-curve scores above 85%, meaning they were fairly good at ranking who had the disease. The same was true, though slightly weaker, for frontotemporal dementia versus healthy aging. The most informative signals in both cases came from the alpha band, a rhythm linked to relaxed wakefulness that is often disrupted in dementia. By contrast, telling Alzheimer’s and frontotemporal dementia apart was much harder. The best measures for this task came from the slow delta band and reached only modest accuracy, suggesting that the two diseases share many of the same large-scale network disruptions in these EEG recordings.
When more complexity does not mean better answers
One hope of ensemble methods is that combining many weak predictors can yield a stronger, more reliable model. In this study, however, the stacked ensemble did not outperform the best single connectivity features. In fact, the top stand‑alone models were often slightly better. Additional analyses showed that many base models were making similar kinds of errors, so the ensemble had limited truly new information to exploit. The authors also tested different ways of measuring distances between connectivity matrices, including advanced “manifold” geometry methods, but found little advantage over simpler approaches for this between‑person diagnostic problem.
What this means for patients and clinicians
For people facing memory or behavior changes, these findings offer cautious optimism. Resting‑state EEG, a quick, inexpensive, and widely available test, does capture meaningful signatures of dementia in how brain regions connect with one another. At the same time, the work underscores that clever mathematics alone cannot fully disentangle closely related diseases when data are limited and brain changes overlap. The authors argue that carefully chosen, interpretable EEG features, evaluated with strict subject‑level testing, may be more reliable than highly complex models. Future progress, they suggest, will likely come from combining EEG connectivity with other biomarkers such as brain scans, blood tests, and cognitive profiles, to build more precise and trustworthy tools for diagnosing different forms of dementia.
Citation: Mlinarič, T., Van Den Kerchove, A., Barinaga, Z.I. et al. EEG-based classification of alzheimer’s disease and frontotemporal dementia using functional connectivity. Sci Rep 16, 4903 (2026). https://doi.org/10.1038/s41598-026-35316-9
Keywords: EEG, functional connectivity, Alzheimer’s disease, frontotemporal dementia, machine learning