Clear Sky Science · en
Interaction between ELMO1 DNA methylation and Med31 promotes H. pylori-induced gastric cancer EMT and intestinal metaplasia via M2 polarization
Why stomach bacteria matter for cancer risk
Most of us carry billions of bacteria in our digestive tract, and one of them—Helicobacter pylori—can quietly live in the stomach for decades. For some people this infection is harmless, but for others it becomes the first step toward gastric cancer, one of the world’s deadliest cancers. This study explores how a tiny chemical change to DNA in stomach cells helps turn a long‑lasting infection into precancerous damage and, eventually, an environment where cancer can grow.
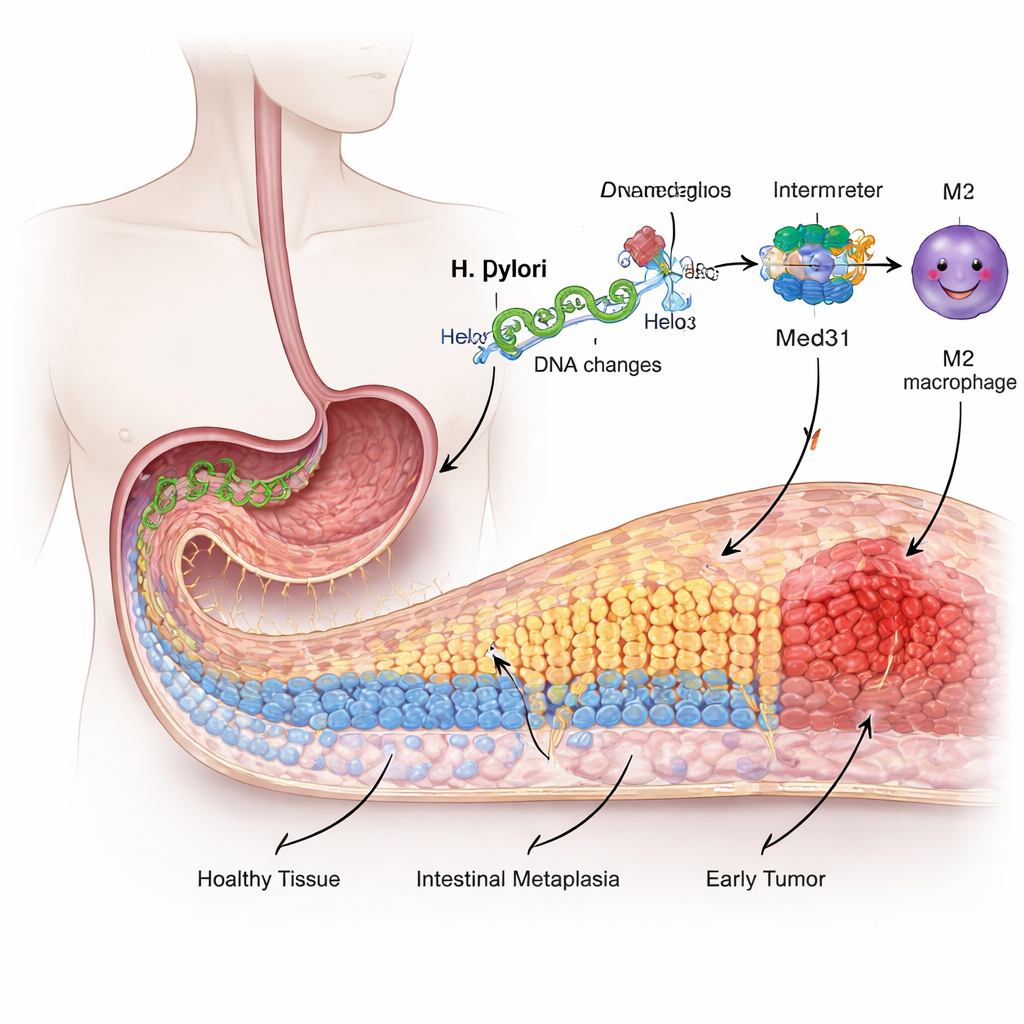
From friendly lining to risky makeover
The inner surface of the stomach is lined with specialized cells that are adapted to handle strong acid, not food digestion like the intestine. Over years of irritation and inflammation from H. pylori, some of these stomach cells start to “redecorate” themselves, taking on features of intestinal cells in a process called intestinal metaplasia. This change is considered a precancerous stage. The researchers used a human gastric cell line to mimic infection and found that exposure to H. pylori made these cells grow faster, move more easily, and switch on intestinal‑type markers, all signs that the lining was shifting away from its normal identity.
Small chemical tags with big consequences
Our DNA can be marked with tiny chemical tags called methyl groups that act like on–off switches for genes without altering the genetic code itself. The team focused on a gene called ELMO1, already known to be involved in cell movement and cancer spread. They showed that H. pylori infection increased methylation of the ELMO1 gene and, somewhat counter‑intuitively, boosted its activity and protein levels in gastric cells. When they used a drug that strips away these methyl tags, the cells lost many of the infection‑driven changes: they divided less, moved less, and showed fewer signals of intestinal metaplasia. This pointed to ELMO1 methylation as a crucial switch flipped by the bacterium.
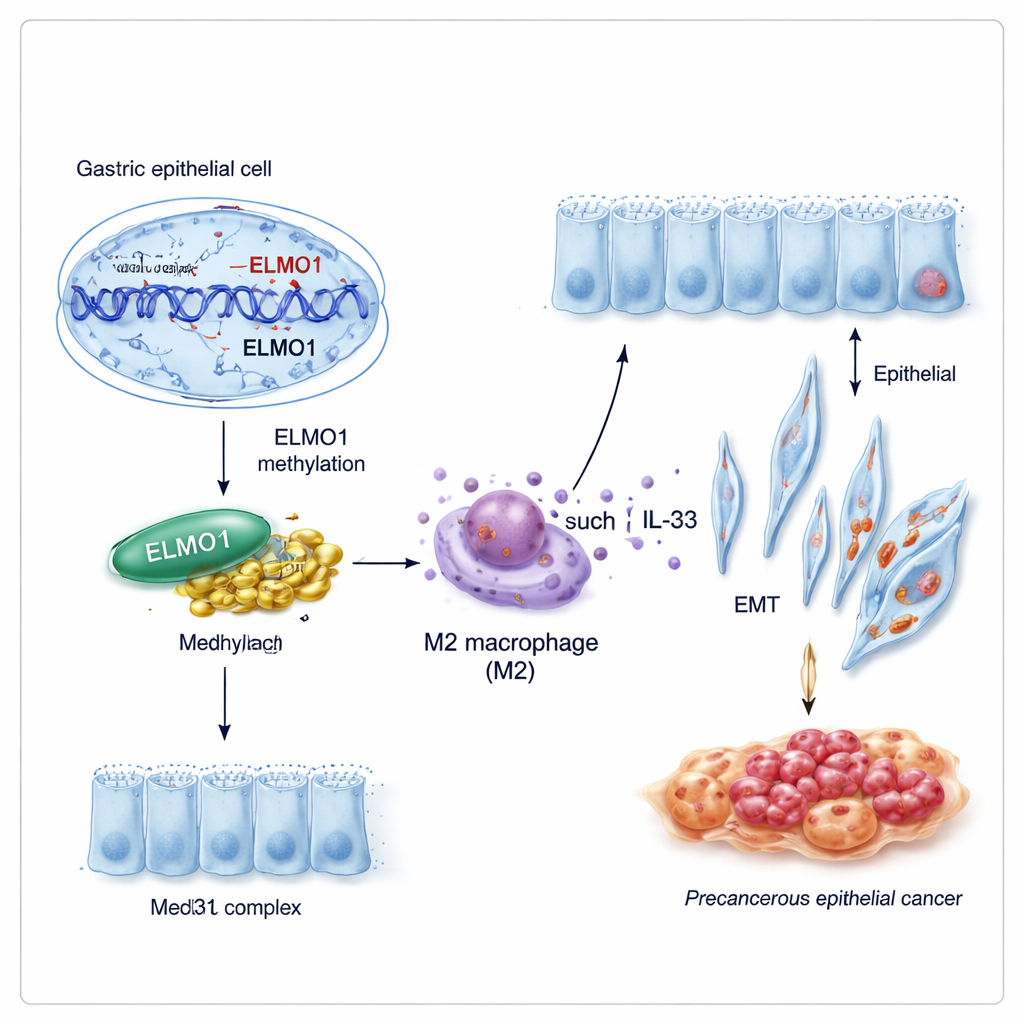
A new partnership that rewires cell behavior
Genes rarely act alone. The researchers discovered that methylated ELMO1 favors a partnership with another protein, Med31, part of a large “control board” that helps decide which genes a cell turns on. Under conditions that promoted DNA methylation, ELMO1 levels rose, Med31 increased, and the two physically bound to each other, while an older known partner of ELMO1, DOCK10, was sidelined. This new ELMO1–Med31 team appears to shift the gene‑expression program of stomach cells in ways that encourage them to adopt intestinal features and behave more aggressively.
How immune cells are pushed to help the tumor
Cancer does not develop in isolation; it grows within a complex neighborhood of immune and support cells. The study shows that infected gastric cells with methylated ELMO1 release higher levels of IL‑33, a signal that nudges nearby immune cells called macrophages into a tumor‑friendly “M2” state. Using a co‑culture system, the authors found that macrophages exposed to these signals strongly tilted toward the M2 type and, in turn, secreted factors that pushed fresh gastric cells to become more mobile, more invasive, and more like intestinal cells. Markers of epithelial‑mesenchymal transition—when stationary lining cells transform into wandering, invasion‑capable cells—rose sharply under the influence of these M2 macrophages.
What this means for preventing stomach cancer
Taken together, the work paints a chain of events: H. pylori infection alters DNA methylation of ELMO1, this modified ELMO1 teams up with Med31, infected cells send out signals that convert nearby immune cells into M2 helpers, and these helpers then drive both intestinal metaplasia and a more invasive cell state. For non‑specialists, the key idea is that a common stomach bacterium can reshape both the stomach lining and the local immune response through reversible chemical marks on DNA. Understanding this pathway may open doors to new blood‑based tests that detect risky methylation patterns early, as well as drugs that interrupt the ELMO1–Med31–M2 axis before a chronic infection progresses to gastric cancer.
Citation: Lu, T., Yu, T., He, C. et al. Interaction between ELMO1 DNA methylation and Med31 promotes H. pylori-induced gastric cancer EMT and intestinal metaplasia via M2 polarization. Sci Rep 16, 5201 (2026). https://doi.org/10.1038/s41598-026-35314-x
Keywords: gastric cancer, Helicobacter pylori, DNA methylation, intestinal metaplasia, tumor microenvironment