Clear Sky Science · en
Effectiveness and safety of cyclosporine A in moderate to severe COVID-19: a randomized, open-label trial
Why this matters beyond the hospital
Even as vaccines and new drugs have changed the course of the COVID-19 pandemic, doctors still need better ways to help people whose illness turns dangerously inflammatory. This study asks a practical question with worldwide relevance: can an old, widely available transplant drug called cyclosporine A safely calm the immune “storm” in moderate to severe COVID-19 and help patients recover faster?
A familiar drug in a new role
Cyclosporine A has been used for decades to prevent organ rejection and treat certain immune diseases. Scientists have also learned that it can block the replication of several viruses, including coronaviruses, and dial down some of the very immune signals that drive the severe, body‑wide inflammation seen in critical COVID-19. Because the drug is already approved, inexpensive, and taken by mouth, it is an attractive candidate to repurpose for patients whose immune systems are overreacting to SARS‑CoV‑2.
How the trial was carried out
The researchers ran a randomized, open‑label phase III trial at Alexandria University Hospital in Egypt, a major referral center during the pandemic. Out of more than 1,500 adults screened between January and September 2022, 75 patients with confirmed COVID-19 and clear signs of heightened inflammation were enrolled; 66 completed the study. Participants were randomly assigned either to receive cyclosporine A capsules in addition to the national standard treatment, or to receive the standard treatment alone. All patients were between 18 and 65 years old and had moderate to severe disease but did not yet require mechanical ventilation.
Tracking recovery and inflammation
Doctors followed patients daily in the hospital or via telemedicine. They tracked symptoms, oxygen levels measured by fingertip probe, and a World Health Organization seven‑point scale that ranges from fully recovered to death. Blood tests were done at the start, day 7, and day 14 to measure markers linked to the so‑called cytokine storm: C‑reactive protein (CRP), interleukin‑6 (IL‑6), ferritin, and D‑dimer. These markers reflect how inflamed the body is and how at risk patients might be for clotting and organ damage. Safety was closely monitored, including serious side effects and new bacterial or fungal infections.
What the researchers found
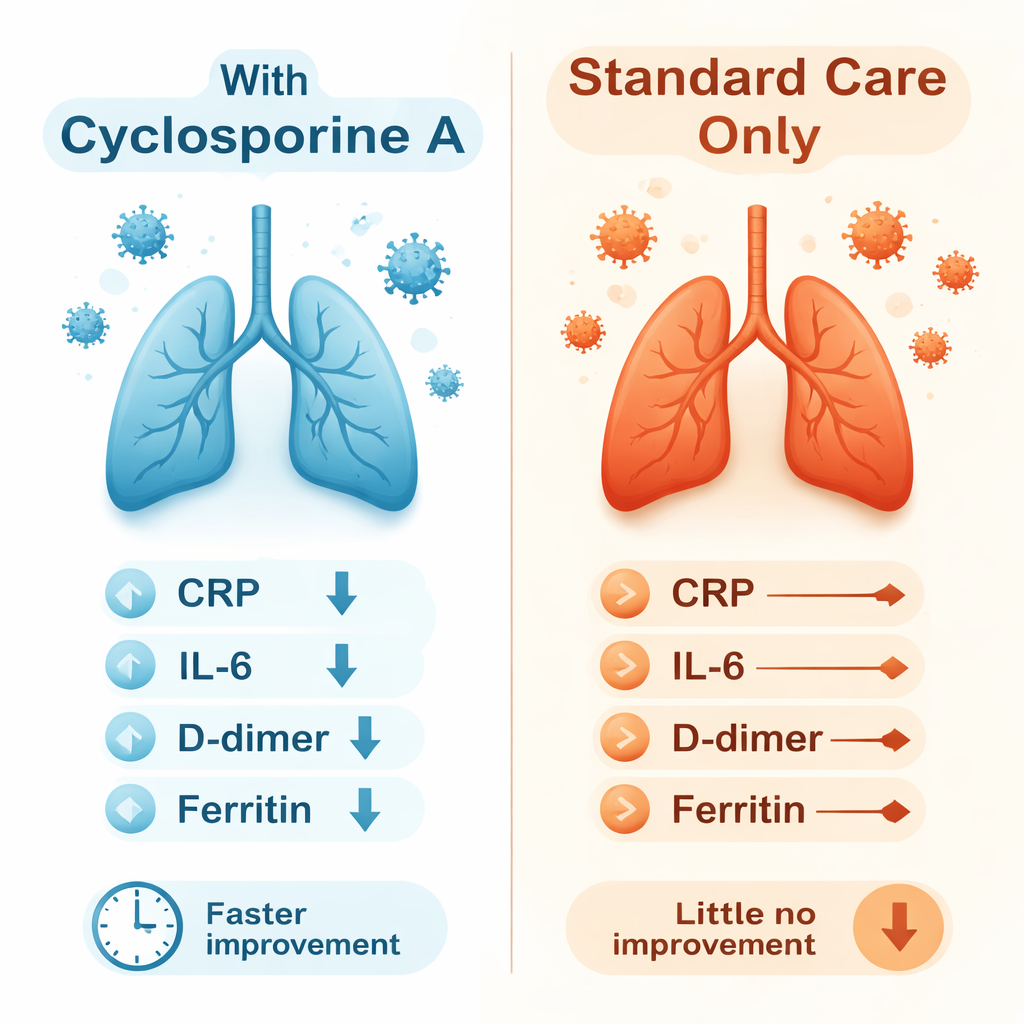
Cyclosporine did not dramatically change the big clinical milestones captured by the WHO scale: in both groups, no patients needed high‑flow oxygen, mechanical ventilation, or died during the 28‑day follow‑up. Where the drug did make a difference was in speed of improvement and in blood chemistry. Patients who received cyclosporine improved, on average, about one day sooner than those on standard care alone. By day 7, their oxygen levels were slightly higher, and by days 7 and 14 they had substantially lower CRP and IL‑6 levels, signaling a quieter immune response. By day 14, levels of D‑dimer and ferritin were also lower in the cyclosporine group than in controls, suggesting less clotting activity and iron‑related inflammation.
Safety and limits of the study
Importantly, the drug appeared safe in this setting. No serious drug‑related problems or secondary bacterial or fungal infections were seen during the 28 days of follow‑up, aside from one case of significant diarrhea. The relatively small number of patients and the careful selection of individuals without severe organ problems mean the findings may not apply to all people with COVID‑19, especially the sickest patients on ventilators. The open‑label design also leaves room for subtle bias in how symptoms were assessed. The authors emphasize that larger studies, direct comparisons with other immune‑modifying drugs, and different dosing strategies are needed.
What this means for patients and clinicians
For lay readers, the bottom line is that cyclosporine A did not turn out to be a miracle cure that keeps people off ventilators or guarantees survival. However, it did seem to help patients with moderate to severe COVID‑19 feel better a bit faster while measurably calming the dangerous inflammatory signals in their blood, without major safety concerns in this carefully chosen group. Because cyclosporine is already widely available in pill form and relatively affordable, these findings support its potential as an add‑on treatment to help tame the immune overreaction in COVID‑19. Future, larger trials will be needed to confirm whether this modest benefit can translate into fewer complications or deaths in the broader population.
Citation: Zidan, A.A., Gad, A.Y.S., Zakaria, N.H. et al. Effectiveness and safety of cyclosporine A in moderate to severe COVID-19: a randomized, open-label trial. Sci Rep 16, 6722 (2026). https://doi.org/10.1038/s41598-026-35292-0
Keywords: cyclosporine A, COVID-19 treatment, cytokine storm, anti-inflammatory therapy, clinical trial