Clear Sky Science · en
Modeling and experimental verification of polycaprolactone nanoparticle precipitation
Why tiny plastic spheres matter for medicine
Imagine packing a powerful drug inside a speck so small it can slip through blood vessels and release its cargo exactly where it is needed. Such specks, called polymer nanoparticles, are at the heart of many emerging treatments and imaging tools. But making them with the right and reproducible size is surprisingly hard. This study shows how a simple physics-based computer model can predict and tune the size of a widely used biodegradable nanoparticle, potentially cutting years of trial‑and‑error from the development of new nanomedicines.
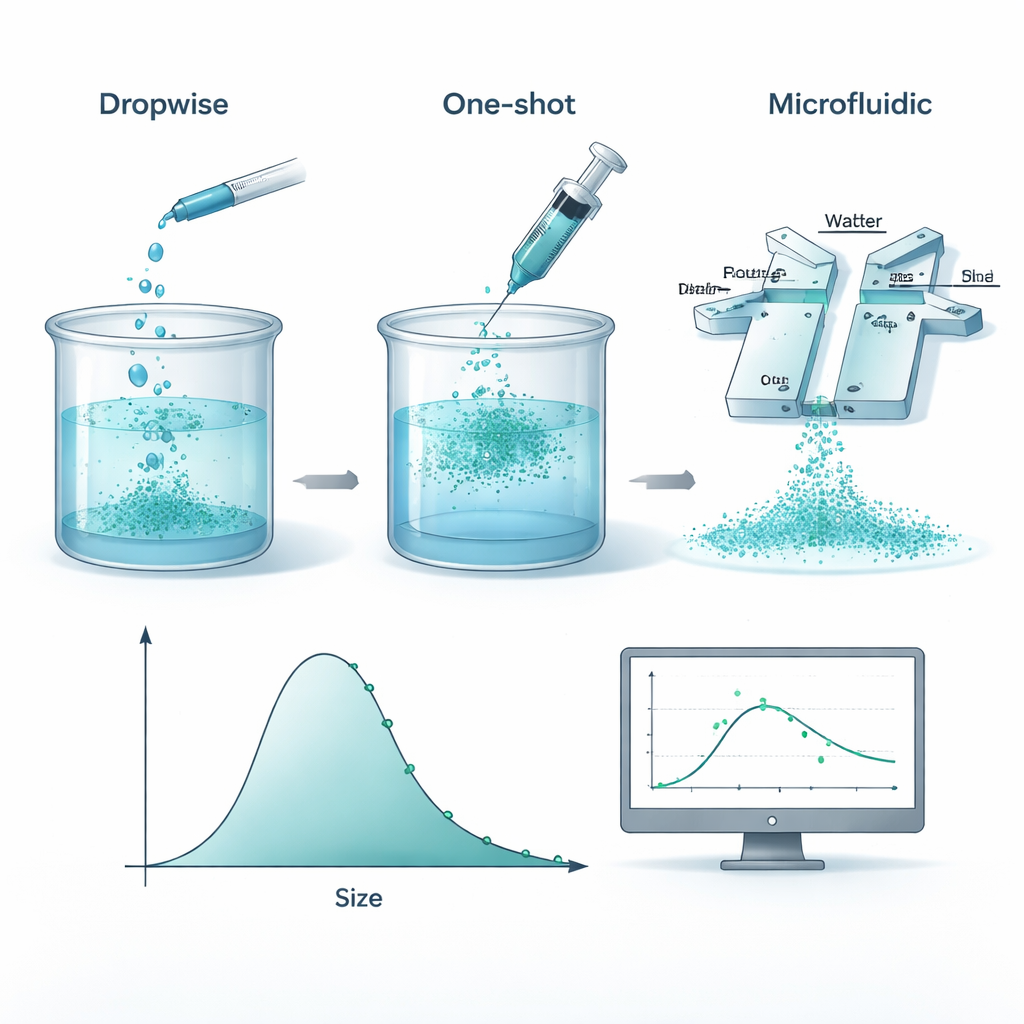
From kitchen mixing to lab precision
To create these nanoparticles, researchers dissolve a biodegradable plastic called polycaprolactone (PCL) in an organic solvent and then mix it with water so that the polymer “falls out” of solution and forms tiny spheres. The team compared three practical ways of doing this: slowly adding the polymer solution drop by drop, dumping it in all at once, and pushing both liquids through a small microfluidic chip where they meet in narrow channels. Under carefully controlled conditions, all three approaches produced particles with very similar average sizes and size spreads. This means that, at least in the ranges studied, what matters more than the exact mixing gadget is what you put in—polymer and stabilizer amounts—rather than how you stir.
How thickness and helper molecules shape the particles
The researchers then explored how the recipe ingredients control the final particle size. Increasing the amount of PCL in the organic phase makes that liquid thicker, or more viscous. Think of trying to mix syrup into water instead of juice: thicker syrup breaks into larger droplets. Here, thicker polymer solutions led to bigger nanoparticles and slightly broader size distributions. Adding a second solvent, ethanol, helped keep the process stable even at very high polymer levels, but at the cost of making particles larger at the upper concentration range. A second ingredient, a surfactant called Pluronic F‑127, acts like a molecular anti‑clumping agent. At low surfactant levels, particles tend to bump into each other and stick, growing larger and less uniform. As the surfactant concentration increases, it coats the particle surfaces, prevents them from merging, and yields smaller, more stable nanoparticles—up to a point, beyond which further gains are modest and the size distribution can become more mixed.
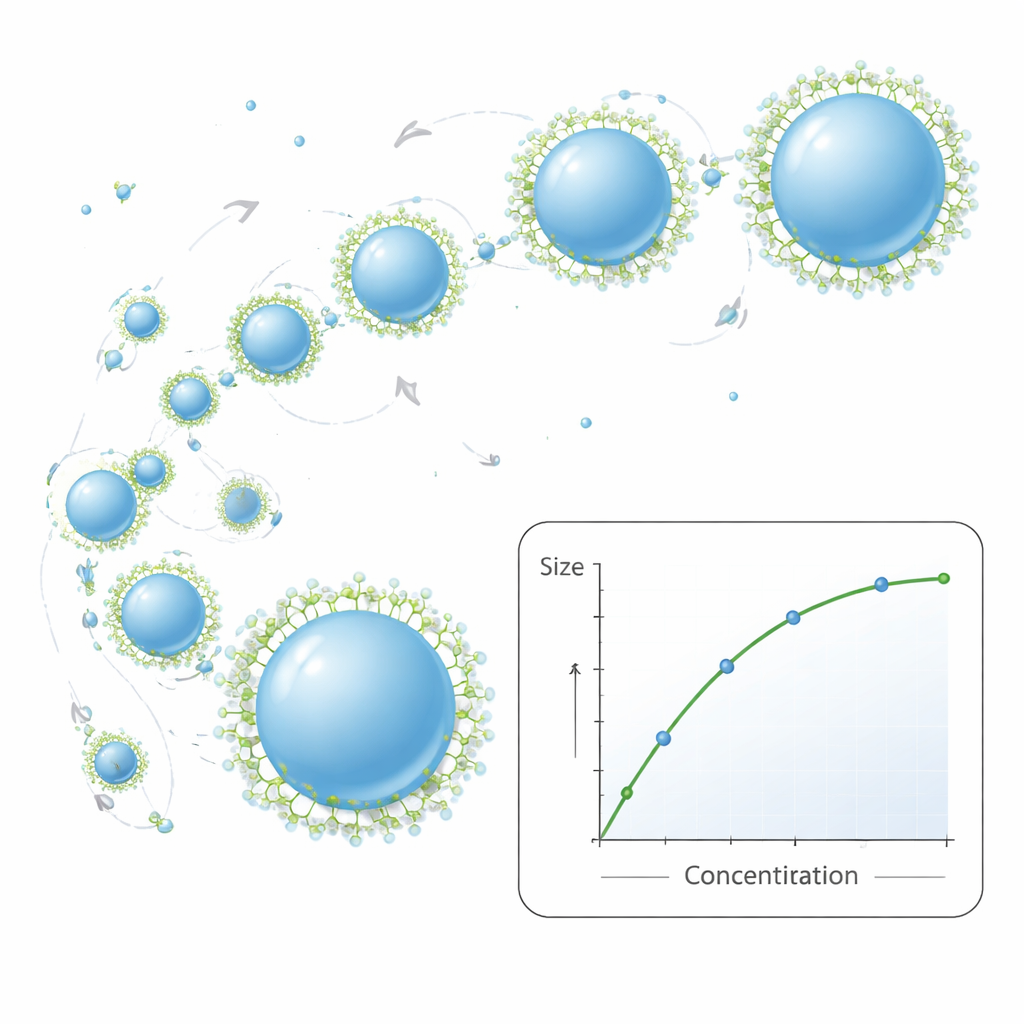
A simple growth rule that matches reality
At the heart of the work is a compact mathematical model that describes how nanoparticles grow after they first appear. It treats their motion as random jiggling in liquid, where collisions sometimes lead to two particles fusing into a larger one. Earlier models assumed that whenever particles met they merged instantly and became perfect spheres. The new model adds two realistic twists: first, it allows a finite time for two touching particles to relax from an elongated shape into a single sphere; second, it lets surfactant molecules gradually “block” the surface, slowing or stopping further merging. With only a few measurable inputs—such as temperature, liquid viscosity, and initial polymer concentration—the model predicts how average particle size should change with recipe conditions. Across many experiments, the predicted sizes closely matched those measured by light scattering, while also capturing general trends in how broad the size distribution becomes.
Using the model as a design tool
To test whether this approach is truly useful, the team flipped the problem: instead of asking the model to explain past data, they asked it to suggest recipes that should yield three specific nanoparticle sizes and degrees of uniformity. They then prepared these “designed” formulations in the lab. The measured particle diameters differed from the targets by only 1–7 percent—well within typical experimental variation—showing that the model can reliably steer formulation choices. Predictions for how narrow or broad the size distribution would be were less exact, but still good enough to distinguish relatively tight from more spread‑out populations. Compared with heavy‑duty simulation methods that track every molecule or every flow eddy, this streamlined model runs quickly on modest computers and is easy to adapt to other polymers and processing setups.
What this means for future nanomedicine
For non‑specialists, the key message is that making useful nanoparticles no longer has to rely solely on painstaking trial‑and‑error in the lab. By capturing the essential physics of how small plastic droplets move, collide, and are protected by surfactants, this work delivers a practical “map” from formulation choices—how much polymer, which solvent mix, how much stabilizer—to the final particle size. Because PCL is a common biodegradable material used in drug delivery, implants, and imaging agents, this model could help researchers design safer, more effective nanomedicines faster and with fewer wasted materials. The same principles can be extended to other polymers, bringing more predictability and efficiency to the world of nanoscale therapies.
Citation: Rybak, E., Trzciński, J., Gac, J. et al. Modeling and experimental verification of polycaprolactone nanoparticle precipitation. Sci Rep 16, 6613 (2026). https://doi.org/10.1038/s41598-026-35286-y
Keywords: polymeric nanoparticles, nanoprecipitation, drug delivery, numerical modeling, polycaprolactone