Clear Sky Science · en
Modifying graphene oxide with magnetic nanoparticles and Mg-Al LDHs and its application as an efficient catalyst in organic reactions
Smart Powders That Make Medicine and Materials
Imagine a tiny, reusable powder that can help chemists snap together new drug-like molecules quickly, then leap out of the liquid when you wave a magnet nearby. This paper describes just such a material: a layered, magnetic form of graphene oxide that speeds up important chemical reactions while being easy to recover and reuse. The work sits at the crossroads of nanotechnology, green chemistry, and drug discovery, showing how careful design at the nanoscale can simplify complex chemistry in the lab and, eventually, in industry.
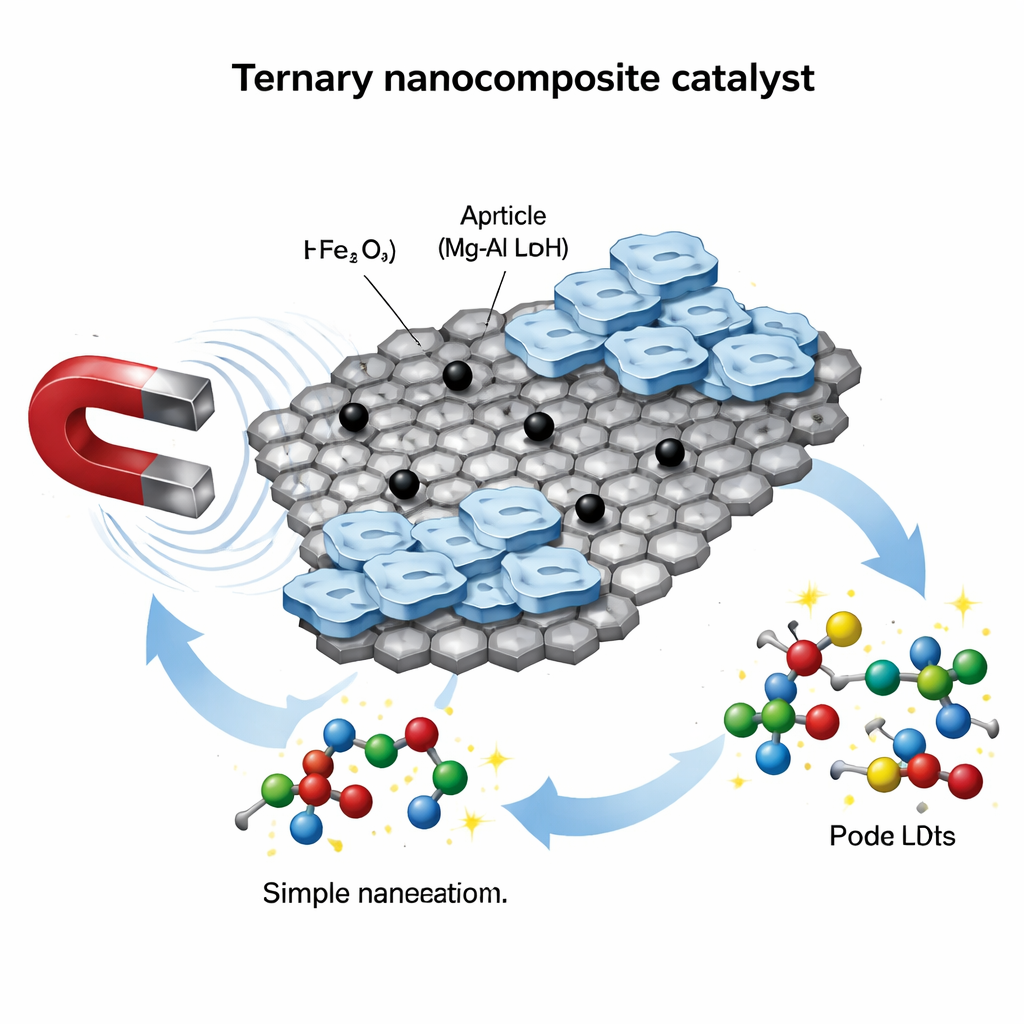
Building a Three-in-One Nano Helper
The researchers started from graphene oxide, a single-atom-thick carbon sheet decorated with oxygen groups. On its own, graphene oxide spreads well in water and offers a huge surface for reactions, but it is hard to separate once a reaction is done. To fix this, the team first anchored tiny iron oxide particles—magnetic nanoparticles—onto the graphene sheets. These particles give the hybrid material a strong magnetic response, so it can be pulled out of a mixture with a simple magnet. Next, they added a third component: thin platelets of a magnesium–aluminum material known as a layered double hydroxide. These plates bring basic (alkaline) sites and ion-exchange abilities, turning the whole structure into a versatile chemical “workbench.”
Seeing and Measuring the New Material
To confirm that their three-in-one structure had really formed, the scientists used a suite of standard materials tests. Electron microscope images showed clumps of nearly spherical particles smaller than 100 nanometers—thousands of times thinner than a human hair. Elemental analysis maps revealed that carbon, iron, magnesium, aluminum, and oxygen were all present and well mixed, indicating that the building blocks were evenly distributed rather than separated into patches. X-ray measurements gave diffraction patterns matching all three ingredients, while magnetic tests showed that the final powder remained strongly attracted to a magnetic field, even though its magnetization dropped compared with pure iron oxide because of the added non-magnetic layers.
Speeding Up the Construction of Bioactive Rings
With the structure in hand, the team turned to its job: catalyzing organic reactions. They chose two families of ring-shaped molecules—called isoxazolones and 2-aminothiophenes—that often appear in pharmaceuticals, pesticides, and other bioactive compounds. Using their magnetic powder as a solid catalyst in warm ethanol, they could join simple starting materials in one pot to build these rings in minutes, usually with very high yields. Tests showed that the new catalyst matched or outperformed many previously reported catalysts, while offering a major practical edge: after the reaction, it could be removed instantly with a magnet instead of by filtration or extraction, then washed and reused.
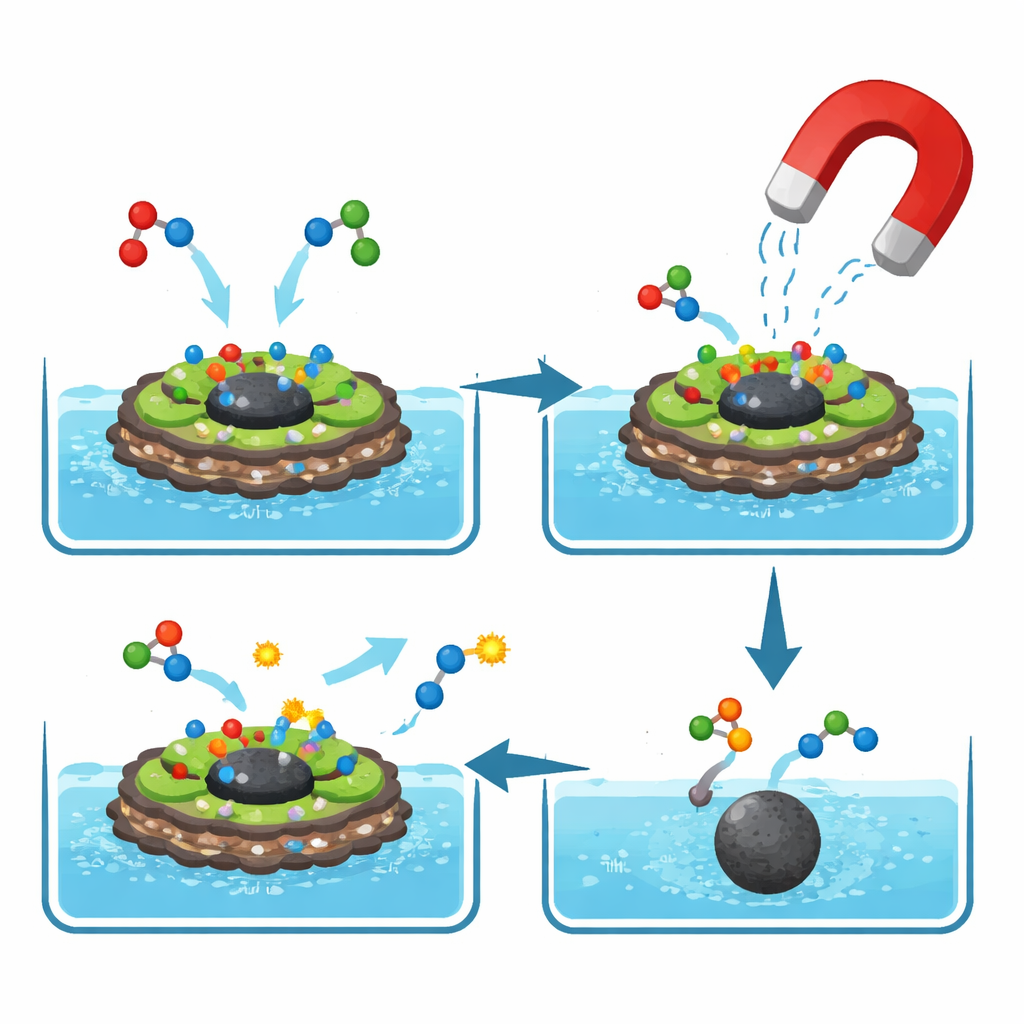
How the Catalyst Guides the Reaction
Although the reactions happen invisibly in solution, the authors propose clear step-by-step pathways. Basic sites on the magnesium–aluminum layers activate acidic hydrogen atoms and make carbon–oxygen groups more reactive, helping the building blocks snap together and shed water or alcohol molecules to form the final rings. The broad graphene surface spreads out the molecules and stabilizes charged intermediates, while the iron oxide core simply makes the whole particle easy to handle. For the sulfur-containing 2-aminothiophenes, the same basic surface first joins a ketone or aldehyde to an activated nitrile, then helps elemental sulfur insert and close the ring, again in a compact, magnetically collectible package.
Reusable Tools for Cleaner Chemistry
To test durability, the researchers ran the same isoxazolone reaction five times, each time reclaiming the catalyst with a magnet, washing, and drying it. Even after the fifth cycle, the product yield had dropped by only about nine percentage points, showing that the material remains active and structurally sound. In simple terms, this work demonstrates a tough, reusable nano-catalyst that combines the strengths of graphene sheets, magnetic particles, and layered minerals. Such smart powders could help chemists make complex, biologically important molecules more efficiently, with less waste and easier cleanup, supporting greener and more economical chemical production.
Citation: Rezaeian, M., Tajbakhsh, M. & Naimi-Jamal, M.R. Modifying graphene oxide with magnetic nanoparticles and Mg-Al LDHs and its application as an efficient catalyst in organic reactions. Sci Rep 16, 6823 (2026). https://doi.org/10.1038/s41598-026-35283-1
Keywords: graphene oxide, magnetic nanocomposite, heterogeneous catalysis, isoxazole synthesis, Gewald reaction