Clear Sky Science · en
Synthesis of 3:2 mullite ceramics from silica-enriched filter cake waste via diphasic gels method
Turning Industrial Waste into High-Value Materials
Industries around the world create mountains of mineral waste that often end up in landfills. This study shows how one such by-product, a silica-rich "filter cake" from an Ethiopian chemical factory, can be transformed into a valuable high‑performance ceramic called mullite. Because mullite is widely used in furnaces, electrical insulators, and advanced electronics, learning to make it cheaply from waste could lower costs, reduce pollution, and conserve natural resources.
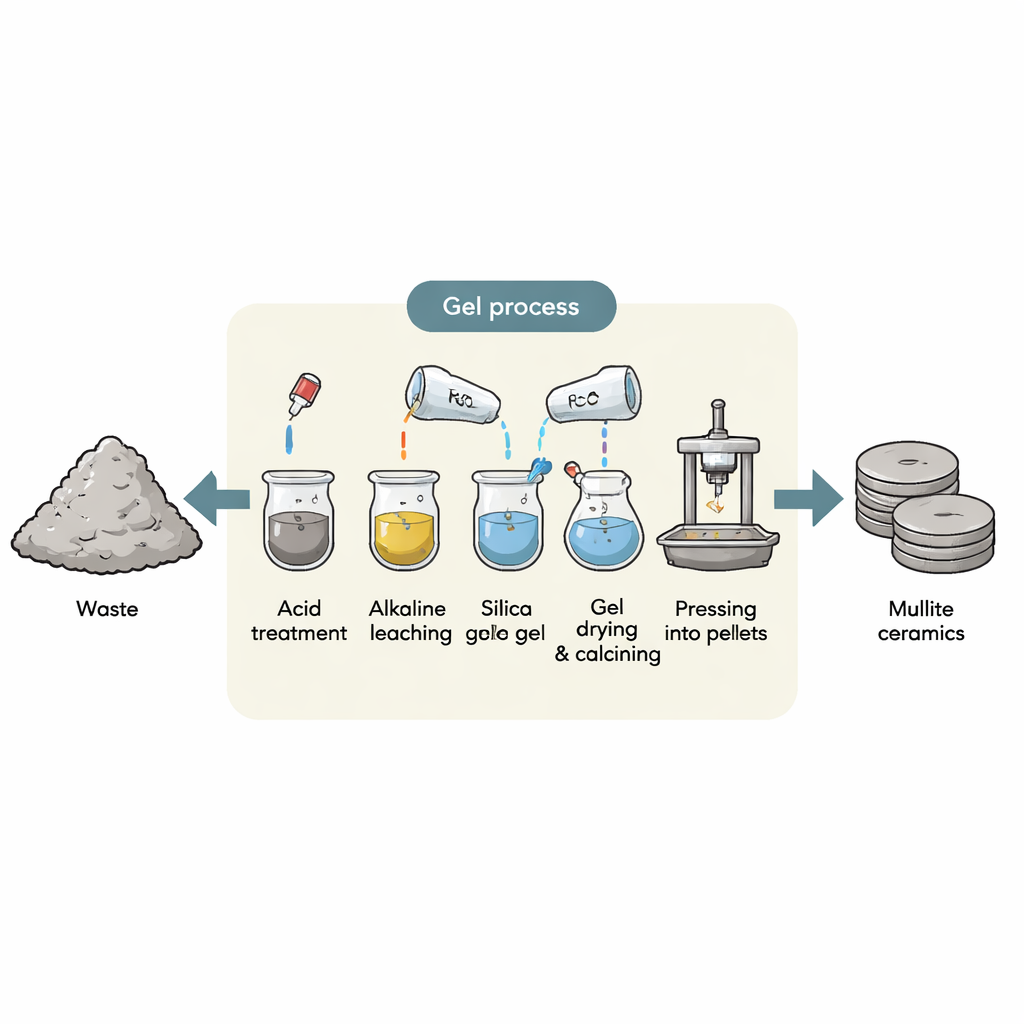
From Factory Sludge to Useful Powder
The researchers began with filter cake waste left over from producing aluminum sulfate. This material contains more than 65% silica, the same basic ingredient found in sand and glass. Instead of discarding it, they cleaned it with acid to remove impurities, then heated it and treated it with a strong base so that the silica dissolved and formed a sodium silicate solution. By carefully adding an acid again, they caused pure silica gel to form and then washed and stored this gel for later use. Chemical analysis confirmed that the resulting silica was very pure, making it a promising replacement for expensive commercial silica.
Building a New Ceramic Through a Two-Phase Gel
To make mullite, the team needed both silica and alumina (aluminum oxide). They mixed the waste‑derived silica gel with a solution of aluminum nitrate using a technique called the diphasic gel method. In this approach, tiny domains of silica and alumina—tens of nanometers across—are mixed so closely that atoms can move and react over very short distances when heated. The mixture was turned into a gel, dried, gently pre‑heated to remove water and nitrates, ground into a fine powder, pressed into small discs, and then fired at temperatures between 1150 °C and 1350 °C. This careful sequence produced what materials scientists call an aluminosilicate precursor, the starting point for mullite.
Watching the Material Transform as It Heats
Using a range of analytical tools, the scientists tracked how this precursor changed as the temperature rose. Thermal analysis showed two key events: around 970 °C, an intermediate phase called spinel formed, and at about 1147 °C, mullite crystals began to appear. X‑ray diffraction confirmed that at an optimized composition and a firing temperature of 1250 °C, the material turned into almost pure mullite with very few unwanted phases. Electron microscope images revealed how the structure evolved: at lower temperatures, small rod‑ and flake‑shaped mullite crystals started to form; at 1250 °C they became dominant; and by 1350 °C the structure was much denser, with grains packed closely together. Chemical mapping showed that aluminum and silicon were evenly distributed, a sign of good mixing and uniform properties throughout the ceramic.
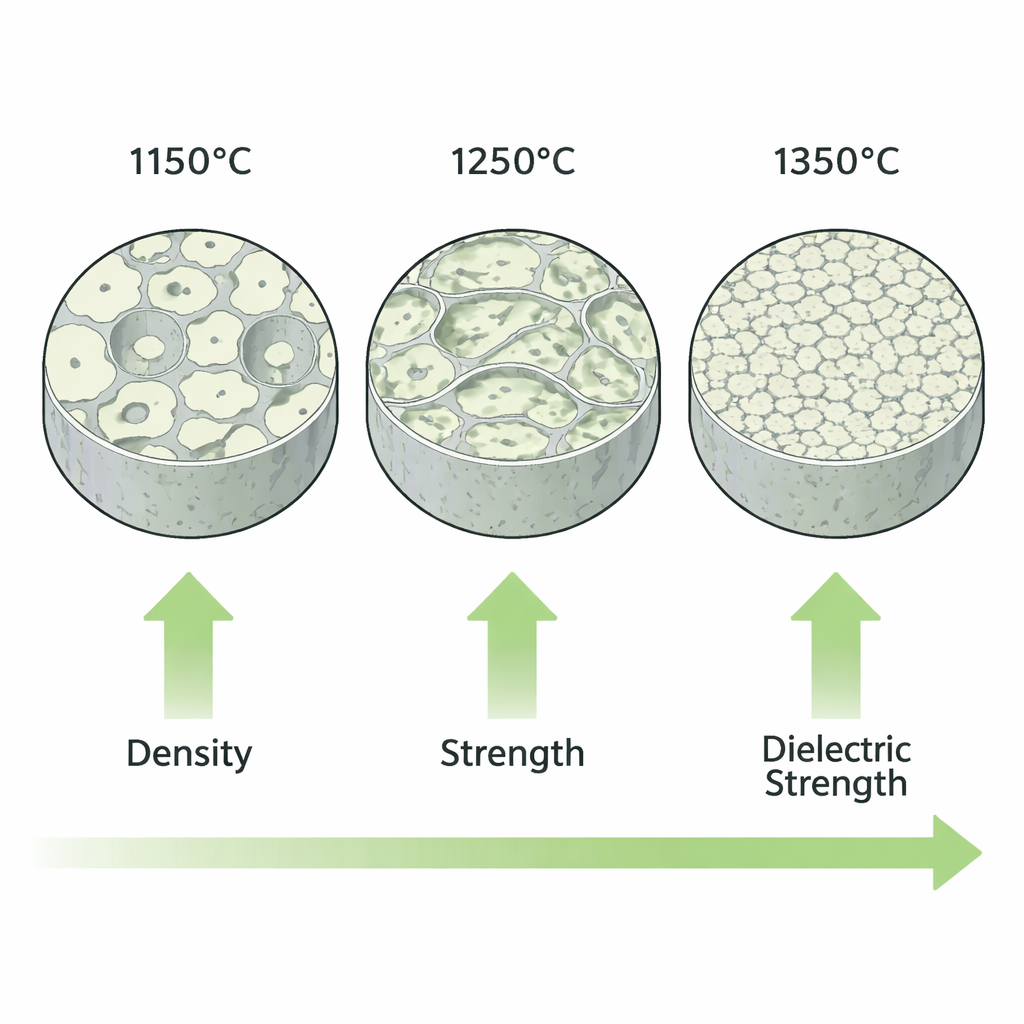
Strength and Insulation Improve with Heat
The researchers then connected these microscopic changes to real‑world performance. As the firing temperature increased from 1150 °C to 1350 °C, open pores inside the ceramic shrank from about 22% to roughly 12%, while density rose to 2.615 grams per cubic centimeter. With fewer and smaller pores, the compressive strength climbed to 420 megapascals—comparable to or better than many commercial mullite products made from pure raw materials at higher temperatures. The ability of the ceramic to resist electrical breakdown also improved, reaching a dielectric strength of 10.2 kilovolts per millimeter. This means the material can withstand high voltages without conducting electricity, an essential property for insulators used in power grids and electronic devices.
What This Means for Technology and the Environment
In everyday terms, this work demonstrates a way to turn a troublesome industrial sludge into a tough, heat‑resistant, and electrically insulating ceramic using relatively modest firing temperatures. By exploiting the fine‑scale mixing in diphasic gels, the team produced high‑quality 3:2 mullite from waste silica and a common aluminum salt, achieving strong, dense, and reliable parts suitable for electrical insulators and other advanced components. If scaled up, this approach could lower manufacturing costs, cut landfill waste, and help countries with limited resources create value‑added materials from their own industrial by‑products.
Citation: Negash, E.A., Mengesha, G.A., Tesfamariam, B. et al. Synthesis of 3:2 mullite ceramics from silica-enriched filter cake waste via diphasic gels method. Sci Rep 16, 5150 (2026). https://doi.org/10.1038/s41598-026-35281-3
Keywords: mullite ceramics, industrial waste reuse, diphasic sol-gel, electrical insulators, advanced ceramics