Clear Sky Science · en
Nicorandil ameliorates neuropathic and inflammatory pain via TNF-α, IL6/MAPKERK1/2 and NO/cGMP signaling
Why easing nerve pain matters
Millions of people live with burning, shooting, or tingling pain that lingers long after an injury has healed. This “neuropathic” pain can be stubbornly resistant to common painkillers and often comes with side effects from long-term medication use. The study summarized here explores whether nicorandil—a heart drug already given to patients with chest pain—might also calm overactive pain pathways in the nervous system, and how it does so at the cellular level.
A heart medicine steps into pain research
Nicorandil is routinely prescribed to treat angina because it relaxes blood vessels. It does this through two main actions: donating small amounts of nitric oxide, a signaling molecule, and opening certain potassium channels in cells. Previous animal work hinted that nicorandil can also lessen pain by acting on nerve receptors involved in heat and chili-pepper sensations and by engaging the body’s own opioid system. In the present study, the researchers set out to map what happens downstream of these first effects, focusing on inflammation, oxidative stress (a kind of chemical “rusting”), and specific signaling routes inside nerve cells.
Testing nicorandil in models of lasting pain
To mimic long-lasting nerve pain, the team loosely tied the sciatic nerve in rats, a classic procedure that produces touch and cold sensitivity similar to human neuropathic pain. They also used the formalin test, in which a mild chemical irritant is injected into a paw to create a well-defined, longer-lasting inflammatory pain. Rats received nicorandil by mouth in two doses, and the scientists measured how strongly they reacted to light touch and cold on the injured paw, as well as how much they licked or flinched after formalin. Importantly, nicorandil did not make animals drowsy or unsteady in an open-field test, suggesting that any reduction in pain behavior was not simply due to sedation.
Dialing down chemical “alarm signals”
Nicorandil clearly lessened both nerve injury–induced pain and formalin-evoked pain. Treated rats tolerated touch and cold better and showed fewer flinches and shorter licking times. Blood tests revealed that nicorandil lowered levels of powerful inflammatory messengers—tumor necrosis factor alpha (TNF-α) and interleukin-6 (IL‑6)—as well as cyclooxygenase‑2, an enzyme that helps produce pain-related prostaglandins. It also reduced malondialdehyde, a marker of oxidative stress, indicating less chemical damage to cell membranes. Under the microscope, nerves from nicorandil-treated animals looked healthier: there was less structural damage and swelling in the sciatic nerve and the cluster of sensory nerve cells known as the dorsal root ganglion.
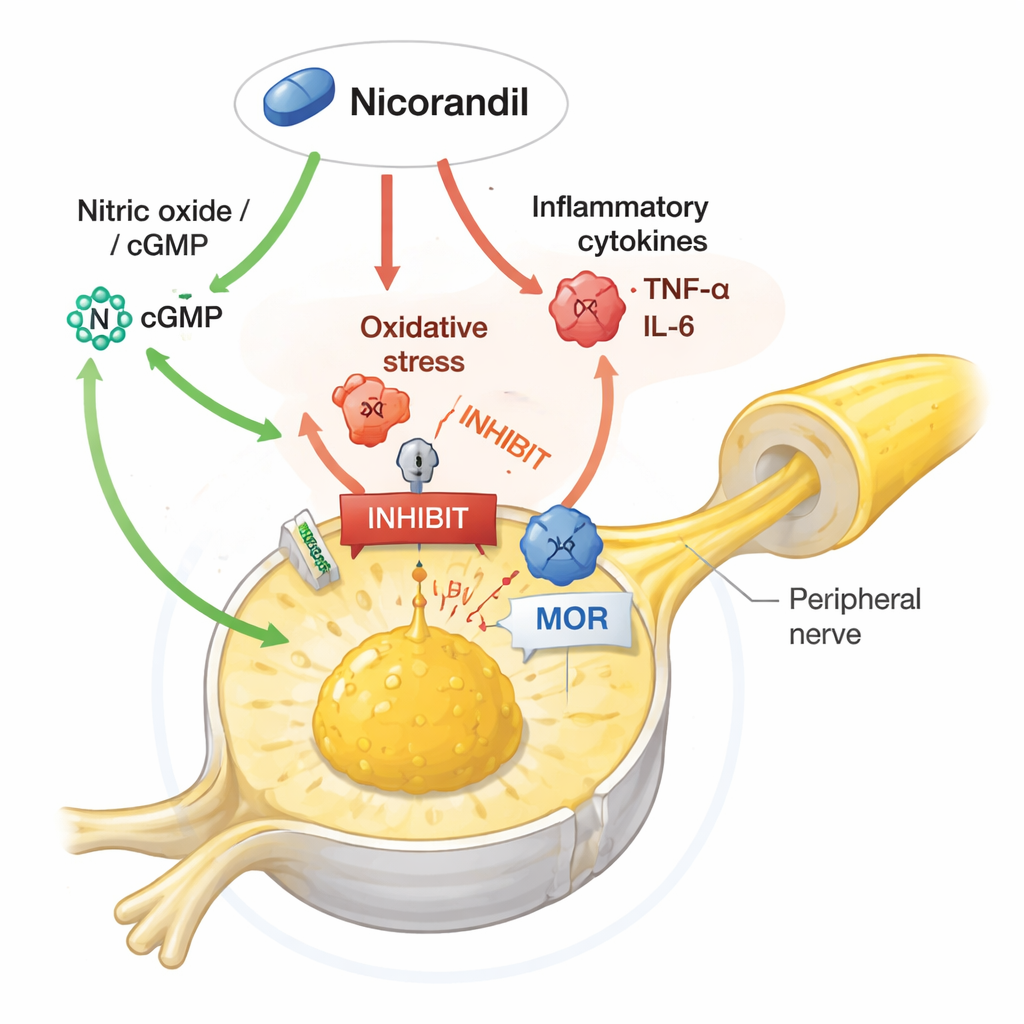
Untangling the signaling web inside pain nerves
The study went further by probing which molecular pathways were required for nicorandil’s benefits. When the researchers gave drugs that boosted nitric oxide or raised cyclic GMP (a messenger molecule linked to nitric oxide), they partially blunted nicorandil’s ability to reduce pain and to normalize key proteins. Nicorandil prevented the usual rise in ERK1/2, a member of the MAP kinase family that amplifies pain signals, and curbed levels of TRPV1, a channel that makes nerves more sensitive to heat and chemical irritants. Enhancing nitric oxide signaling or cyclic GMP reversed these protective changes, while blocking nitric oxide-making enzymes sometimes strengthened nicorandil’s pain relief. Surprisingly, blocking the potassium channels that nicorandil is famous for opening did not abolish its antinociceptive effects, suggesting that in this context, its pain-relieving power relies more on modulating nitric oxide, oxidative stress, inflammation, and opioid-related signaling than on its classic vascular action.
What this could mean for people in pain
Altogether, the findings paint nicorandil as a multitarget pain modulator: it calms overactive nerve pathways by reducing oxidative damage and inflammatory messengers, quieting ERK1/2 and TRPV1 inside sensory neurons, and interacting with nitric oxide and the body’s own opioid system. While these results come from rat models and short-term dosing, they suggest that a medicine already in clinical use for heart disease might one day be repurposed or adapted to help treat difficult neuropathic and inflammatory pain conditions. Future work in people will be needed to determine safe doses, long-term effects, and whether the same protective mechanisms operate in human nerves.
Citation: Badr, R.M., Abuiessa, S.A., Elblehi, S.S. et al. Nicorandil ameliorates neuropathic and inflammatory pain via TNF-α, IL6/MAPKERK1/2 and NO/cGMP signaling. Sci Rep 16, 4722 (2026). https://doi.org/10.1038/s41598-026-35272-4
Keywords: neuropathic pain, nicorandil, inflammation, oxidative stress, nitric oxide signaling