Clear Sky Science · en
Disruption of zinc homeostasis reduces histone acetylation levels in normal and tumor cells
When a Helpful Metal Turns Harmful
Zinc is best known as the nutrient in multivitamins that helps our immune system and wound healing. But inside our cells, zinc must be kept in a very narrow “just right” range. This study shows that when zinc levels in cells get too high, they can quietly switch off important genes by tightening how DNA is packaged, and even push cells toward a self‑cleaning process called autophagy. Because zinc supplements, anti-dandruff ingredients, and experimental cancer drugs can all alter zinc levels, understanding this hidden control system matters for everyday health and for future therapies.
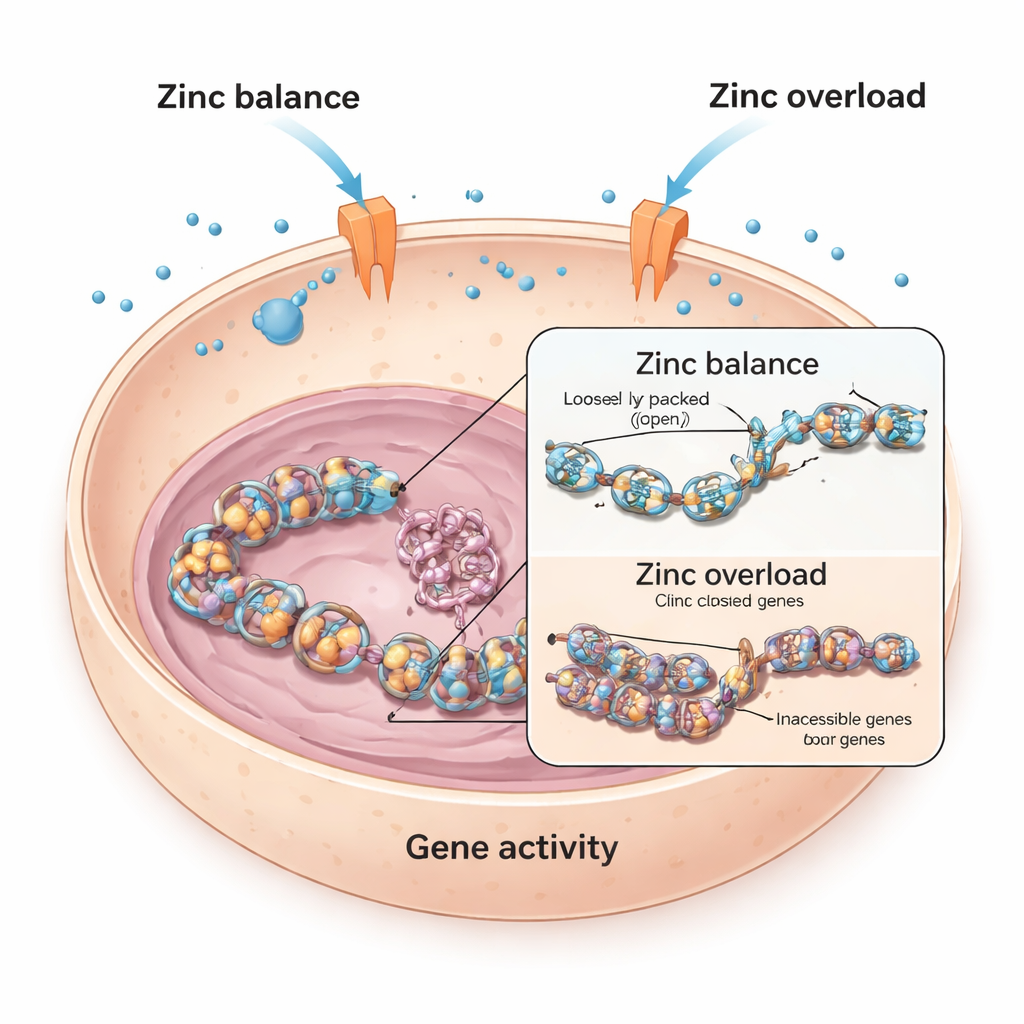
Balancing Zinc Inside Living Cells
Every cell relies on zinc, which helps thousands of proteins do their jobs. To avoid both shortage and overload, cells use special “gates” in their membranes, called zinc transporters, to move zinc in and out. The researchers began with heart muscle cells from mice and humans, asking what happens when these cells are exposed to extra zinc. They found that in mouse heart cells, high zinc levels strongly reduced the activity of a key heart‑related gene called Bmp4, which helps guide heart development and function. In contrast, human heart cells were much less affected, hinting that different cells defend themselves against zinc overload in different ways.
How Zinc Tightens DNA Packaging
To uncover how zinc silences genes like Bmp4, the team looked at histones, the spool‑like proteins around which DNA is wrapped. When small chemical tags called acetyl groups are added to histones, the DNA becomes looser and easier to read; when these tags are removed, DNA packs more tightly and genes are harder to switch on. The study showed that excess zinc in mouse heart cells led to a drop in histone acetylation at a specific site (known as H3K9), closely tracking the fall in Bmp4 activity. Drugs that block histone de‑acetylating enzymes could undo zinc’s effect and restore Bmp4, suggesting that zinc was tilting the balance of this chemical tagging system.
Too Much Zinc Inside, Not Just Outside
The crucial step turned out to be zinc inside the cell’s watery cytoplasm, not just in the surrounding fluid. Using a fluorescent probe that lights up in the presence of free zinc, the researchers saw a surge of zinc inside mouse heart cells after treatment. When they used a zinc shuttle molecule to force zinc directly into cells, they observed the same fall in histone acetylation and gene activity, even with much lower zinc in the outside medium. In human heart cells, by contrast, the transporters at the cell membrane quickly adjusted: some zinc importers shut down and exporters ramped up, preventing a big rise in internal zinc and protecting histone acetylation. Mouse heart cells showed a more “miswired” transporter response, allowing more zinc to flood in and disrupt gene control.
From Gene Control to Self‑Cleaning in Normal and Tumor Cells
The investigators then asked whether this zinc‑driven tightening of DNA packaging is unique to heart cells. They tested several normal human cell types and cancer cell lines from the stomach, uterus, and ovary. When zinc was allowed to build up inside with the help of a shuttle, nearly all of these cells showed a clear drop in histone acetylation. In some tumor cells, such as gastric cancer cells, this happened at especially low zinc doses, hinting that certain cancers are unusually sensitive to zinc imbalance. The team also linked lowered acetylation to autophagy, a process where cells recycle worn‑out parts. Moderate internal zinc increases boosted autophagy, but very high zinc eventually weakened it, suggesting a narrow window where zinc might be used to fine‑tune this self‑cleaning pathway.
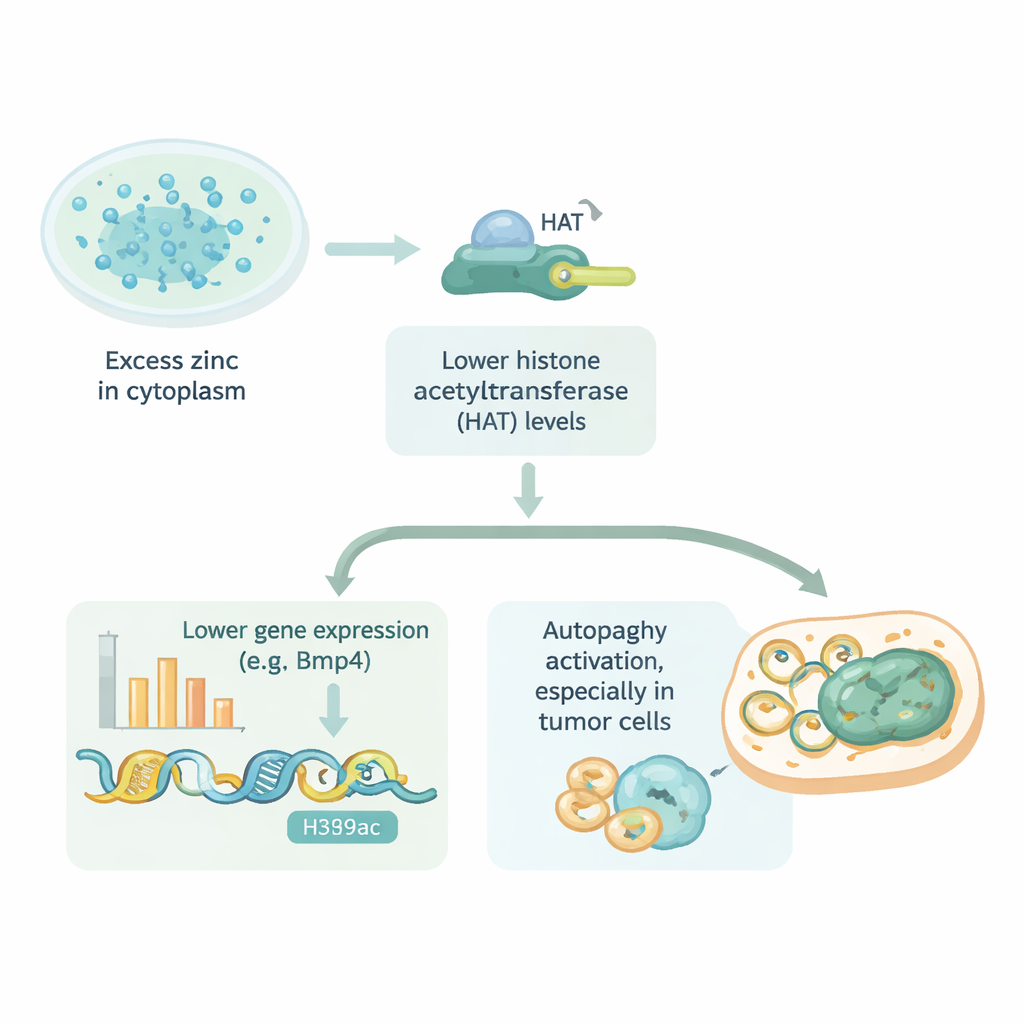
What This Means for Health and Treatment
Put simply, this work reveals that too much zinc inside cells can turn down genes by making DNA harder to access, and that this same pathway can influence how actively cells clean themselves up. The effect is not limited to heart cells; it appears across different normal and cancer cells, though some are better at defending against zinc overload than others. These insights suggest that the tiny zinc transport “gates” in cell membranes could be promising drug targets—either to protect healthy tissues from zinc‑related damage or to push vulnerable tumor cells toward death. While the findings come from cell culture experiments, they provide a fresh lens on how an everyday nutrient can shape gene activity and disease when its delicate balance is disturbed.
Citation: Xu, S., Hu, Y., Tang, C. et al. Disruption of zinc homeostasis reduces histone acetylation levels in normal and tumor cells. Sci Rep 16, 4983 (2026). https://doi.org/10.1038/s41598-026-35270-6
Keywords: zinc homeostasis, gene regulation, histone acetylation, autophagy, cancer cells