Clear Sky Science · en
Various distribution of BMPs in different periosteal layers contributing to inconsistent osteoinductivity of DBM-based products
Why bone repair materials don’t always work the same
When surgeons fill a broken or missing piece of bone, they often turn to demineralized bone matrix (DBM)—processed donor bone that is supposed to kick‑start new bone growth. Yet in real life these products can behave very differently, even when they appear similar on the label. This study asks a simple but important question: could those differences arise because the healing proteins inside the bone are unevenly distributed in different layers of the same bone, and because of how we measure them in the lab?
The promise of bone’s own healing proteins
DBM works because it carries natural bone‑forming molecules called bone morphogenetic proteins, or BMPs. These proteins signal nearby stem cells to turn into cartilage and bone cells, helping a damaged area rebuild itself. Earlier work showed that DBM preparations with more BMPs tend to grow more bone, but also revealed enormous variation between commercial products and even between batches of the same product. The authors of this study suspected that one overlooked source of this variation is where, within a long bone like the femur, the starting material is taken from, and how the BMPs are extracted and measured.
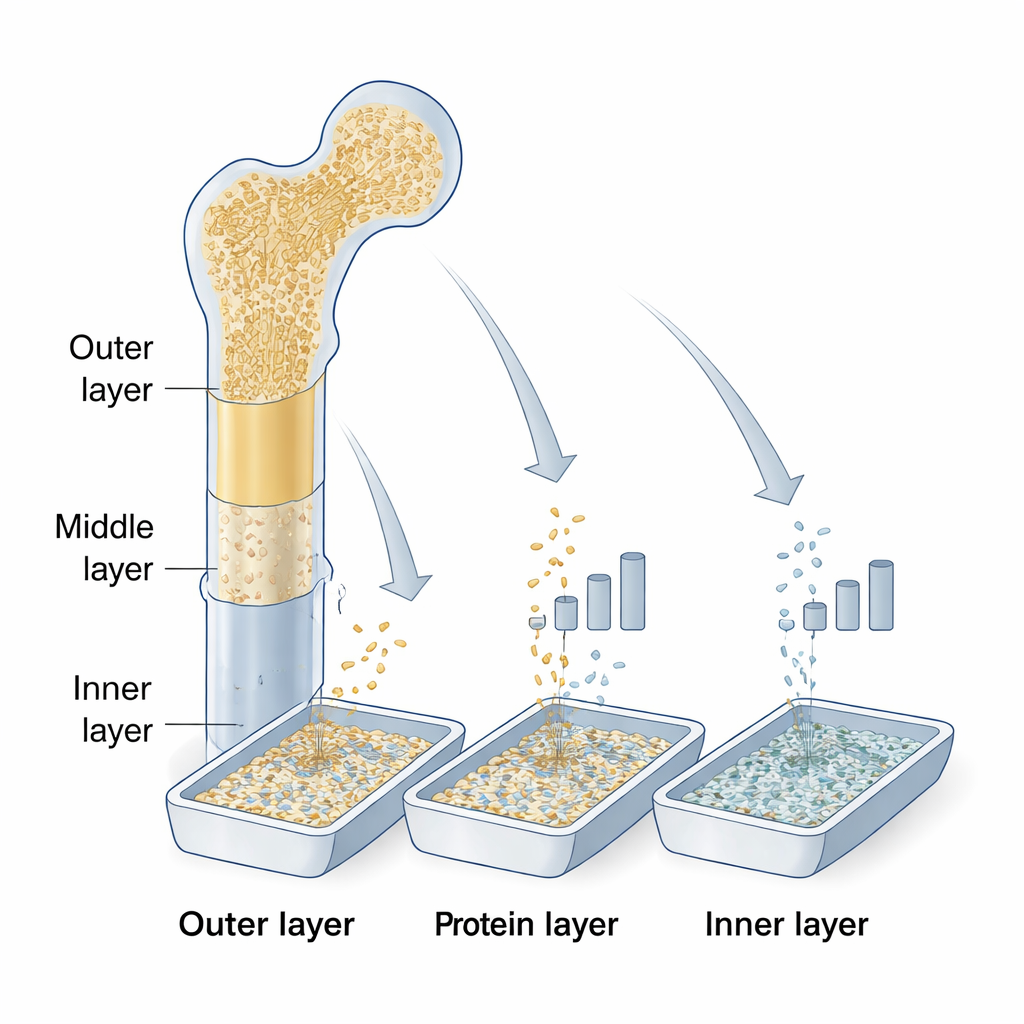
Peeling a bone into layers
Using a single human femur from a donor, the team sliced the hard outer shaft into three equal layers: an outer layer near the periosteum (the fibrous coat of the bone), a middle layer, and an inner layer near the endosteum and marrow side. They ground each layer into small particles. Some particles from the outer and middle layers were left mineralized as ordinary cortical bone, while others—plus all particles from the inner layer—were demineralized in acid to create DBM. This process removes most of the calcium but leaves behind the organic matrix that holds BMPs and other growth factors.
Measuring bone-building signals in the lab
To see how much BMP‑2 and BMP‑7 each sample contained, the scientists compared two common lab extraction methods. One used guanidine hydrochloride (GuHCl), a strong chemical that pulls proteins out of the bone matrix. The other used collagenase, an enzyme that digests collagen, the main structural protein of bone. After extraction, they measured both total protein and specific BMP levels using standard protein assays and highly sensitive antibody‑based tests (ELISAs), then compared the results across layers and methods.
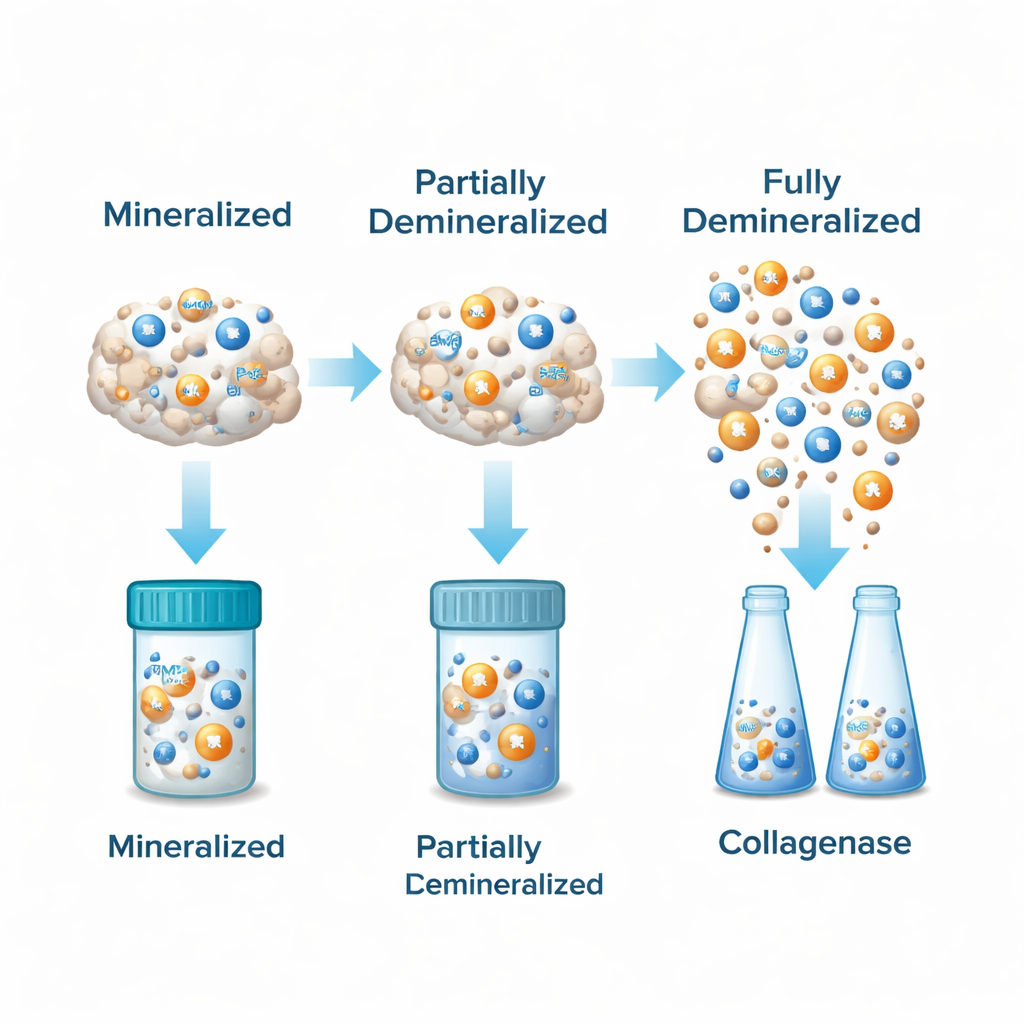
Uneven protein maps inside a single bone
Demineralization made a striking difference: for any given layer, DBM samples contained far more detectable BMP‑2 and BMP‑7 than the matching mineralized bone particles. Within the DBM group, the outer layer consistently had the highest levels of both BMPs, the middle layer had somewhat less, and the inner layer had the lowest amounts. This pattern held regardless of whether GuHCl or collagenase was used. The two BMPs rose and fell together in a strong linear relationship—samples rich in BMP‑2 almost always had more BMP‑7 as well—showing that these key growth signals tend to co‑vary within the bone.
When the test method changes the answer
The way proteins were extracted also mattered. GuHCl pulled out dramatically more BMP‑2 and BMP‑7 than collagenase, in some cases more than ten‑ to one hundred‑fold higher, even though collagenase sometimes yielded more total protein overall. This means that routine lab tests can underestimate or misjudge the bone‑forming power of a DBM product if they rely on a less efficient extraction method. Because clinical products are made from mixtures of bone from different layers and donors, such hidden variability in BMP content can translate directly into unpredictable performance when the material is implanted in patients.
What this means for patients and surgeons
For non‑specialists, the takeaway is straightforward: not all bone graft substitutes are created equal, even if they come from the same type of bone. The outer regions of long bones naturally contain more of the proteins that spark new bone growth, and strong chemical extraction reveals much more of these signals than gentler enzyme methods. These built‑in differences help explain why DBM products sometimes heal bone defects well and sometimes fall short. Better control over where donor bone is taken from and how its protein content is tested could make future bone graft materials more reliable and effective.
Citation: Zhao, Yj., Xue, Y., Sun, S. et al. Various distribution of BMPs in different periosteal layers contributing to inconsistent osteoinductivity of DBM-based products. Sci Rep 16, 5279 (2026). https://doi.org/10.1038/s41598-026-35269-z
Keywords: bone graft, demineralized bone matrix, bone morphogenetic protein, bone healing, orthopedic surgery