Clear Sky Science · en
Metabolic reprogramming enhances oxidative stress resistance in differentiating cardiomyocytes
Why heart cells’ battle with oxygen matters
Every beat of your heart depends on specialized muscle cells called cardiomyocytes. As we grow, these cells face a dramatic life change: they move from a low-oxygen, sugar-fueled environment in the womb to an oxygen-rich world after birth. Oxygen is vital, but it also generates harmful by-products that can damage DNA and kill cells. This study explores how maturing heart cells rewire their metabolism so they can handle more oxygen, generate more energy, and still protect themselves from oxidative damage—insights that could inform future treatments for heart disease and regeneration.
From simple builders to power athletes
Immature heart cells, known as cardiomyoblasts, are small, dividing cells that rely mainly on breaking down glucose for energy. As they differentiate into mature cardiomyocytes, they stop dividing and grow larger, often fusing into polynucleated, elongated cells better suited for powerful contraction. Using high-resolution microscopy, the researchers watched this transformation over ten days in a laboratory cell model. Cell proliferation markers dropped, while the cells stretched and reorganized into fibrous, heart-like structures, confirming that they had entered a more adult-like, contractile state. 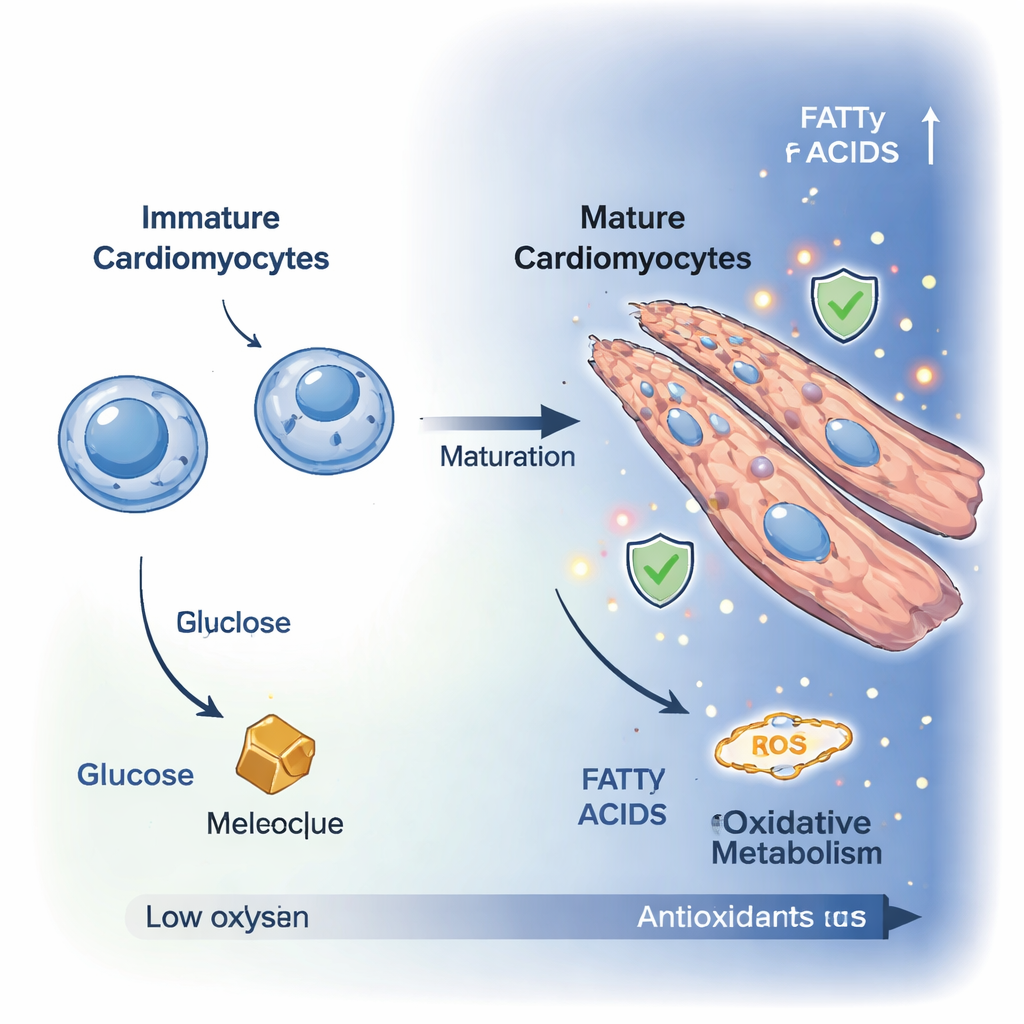
Switching fuel sources and wiring new energy routes
To understand what happens under the hood, the team measured hundreds of small molecules inside the cells and used advanced imaging of natural cell fluorescence to track energy use in real time. Together, these techniques showed a clear shift from a glycolytic state, where cells mainly burn glucose, to an oxidative state dominated by mitochondrial respiration. Levels of glucose and fructose fell, while intermediates of the tricarboxylic acid (TCA) cycle—such as citric acid, malate, and oxoglutarate—increased. Pathway analysis revealed stronger activity in routes that feed fuel into mitochondria, including the malate–aspartate shuttle and amino acid breakdown, indicating that mature cardiomyocytes broaden their fuel options and more efficiently channel energy into their power stations.
Living with more oxygen—and more stress
Burning fuel more efficiently in mitochondria comes at a price: higher production of reactive oxygen species (ROS), chemically aggressive molecules that can damage lipids, proteins, and DNA. The researchers found that as cardiomyocytes matured, their mitochondria became larger and more elongated, and markers of oxidative stress increased. Fluorescence-based imaging revealed more lipofuscin-like granules, a hallmark of oxidized lipids, and a nuclear probe showed higher ROS levels in differentiated cells. Metabolomics pointed to changes in glutathione metabolism, a central antioxidant system that uses the molecule glutathione—built from amino acids like glutamate and glycine—to neutralize ROS. These findings suggest that as heart cells ramp up oxidative metabolism, they also tune their antioxidant pathways to keep damage in check. 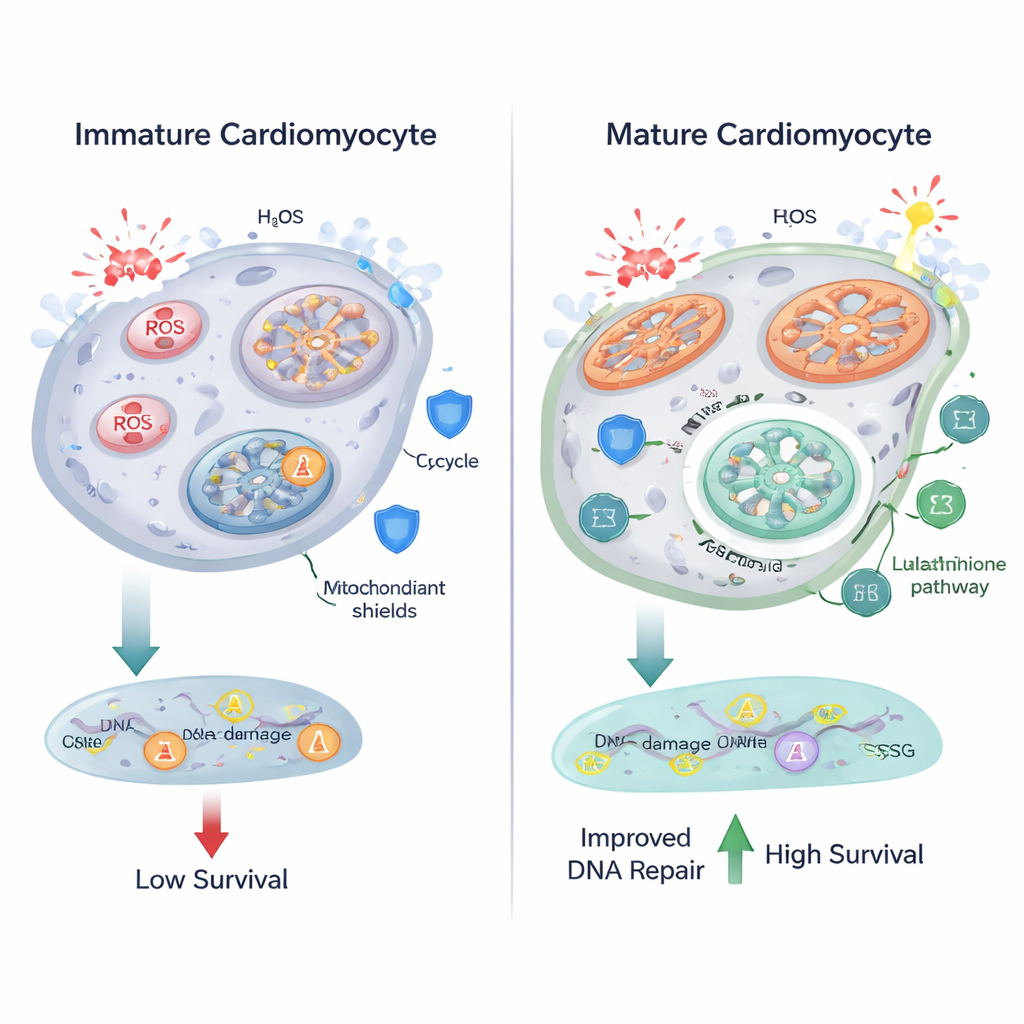
Damage contained, defenses strengthened
Surprisingly, despite higher ROS, differentiated cardiomyocytes did not show more DNA breaks than their immature counterparts when measured by γ-H2AX foci, a sensitive marker of DNA damage. When the team challenged cells with hydrogen peroxide, a strong oxidizing agent, both immature and mature cells accumulated ROS and DNA breaks—but mature cells fared better. They showed proportionally fewer DNA lesions relative to their higher ROS levels, were less prone to activating cell-death–linked signals such as PUMA, and displayed markedly higher survival rates. These results indicate that maturing cardiomyocytes not only adjust their metabolism but also upgrade their DNA repair and survival machinery, allowing them to withstand harsher oxidative conditions.
What this means for protecting the heart
In simple terms, this study shows that as heart cells grow up, they learn to run hotter without burning out. They switch from a sugar-heavy, low-oxygen lifestyle to an oxygen-intensive, mitochondria-driven one, and at the same time build better shields and repair teams to cope with oxidative stress. Understanding this finely tuned balance between energy production and self-protection could help scientists design strategies to make damaged adult heart tissue more resilient, or to guide stem cells into safer, more durable cardiomyocytes for regenerative therapies.
Citation: Novais, L.B., Rodrigues, B.R.I., Pereira, F.O.B. et al. Metabolic reprogramming enhances oxidative stress resistance in differentiating cardiomyocytes. Sci Rep 16, 5534 (2026). https://doi.org/10.1038/s41598-026-35263-5
Keywords: cardiomyocyte differentiation, oxidative stress, mitochondria, cell metabolism, heart disease