Clear Sky Science · en
Synthesis and characterization of g-C3N5/CuS/AgNPs nanocomposite as a Z-scheme photocatalyst for efficient methyl parathion degradation
Why this new water-cleaning material matters
Methyl parathion is a powerful pesticide that has helped protect crops, but it comes with a serious downside: even small amounts left in water can harm people and wildlife, damaging nerves, liver, and kidneys. It also breaks down into other toxic chemicals. This study reports a new light-driven material—a special blend of tiny particles—that can break down methyl parathion in water very quickly using visible light, offering a promising route toward safer drinking water and cleaner rivers near farms.
A stubborn farm chemical in our water
Methyl parathion belongs to a family of pesticides that disrupt the nervous system. Although it does not dissolve well in water, it can still reach ponds, rivers, and groundwater through runoff from fields. Once there, it is difficult to remove and can cause health problems ranging from headaches and nausea to severe organ damage. Traditional cleanup methods, such as using microbes, filtering through membranes, or adding chemicals, often take a long time, generate new waste, or are too costly for large-scale use. Scientists are therefore looking for methods that can directly destroy these molecules, rather than simply moving them from one place to another.
Using light to drive cleanup chemistry
One attractive option is photocatalysis, where light energy activates a solid material so that it can rip apart pollutant molecules. When light hits a suitable solid, it can create mobile charges—negatively charged electrons and positively charged “holes.” If these charges reach the surface before they recombine, they can react with oxygen and water to form aggressive short-lived species that attack pollutants. The challenge is to design a material that absorbs visible light efficiently, separates charges quickly, and offers a large surface where reactions can occur.

Building a three-part nano-sponge for light
In this work, the researchers built a three-part, or “ternary,” nanocomposite by combining: (1) g-C3N5, a carbon- and nitrogen-based solid that absorbs visible light; (2) flower-like copper sulfide (CuS) particles that provide large surface area and good charge mobility; and (3) tiny silver nanoparticles that help shuttle electrons and enhance light absorption. They first prepared g-C3N5 from a common laboratory compound, then grew CuS with a petal-like structure, and finally decorated this mixture with silver using a reducing agent. High-resolution electron microscopes showed g-C3N5 as plate-like pieces, CuS as clustered “flowers,” and silver as small spheres anchored on the surface. Surface-area measurements revealed that the combined material had a much larger reactive area than the individual components, and optical tests showed that its energy gap for absorbing light was reduced to about 1.5 eV, meaning it could use visible light very effectively.
How well the new material destroys methyl parathion
The team then tested how well this nanocomposite could remove methyl parathion from water under visible light. Light alone removed only about 2% of the pesticide in an hour, and the material in the dark removed almost none—showing that both light and the catalyst are required. By contrast, the full three-part material broke down about 95% of methyl parathion within one hour under visible light at a slightly acidic pH of 6 and a modest catalyst dose. Tests across different pH values, catalyst amounts, and starting pesticide levels showed that performance peaked at pH 6 and at an intermediate catalyst concentration; too much material caused clumping and reduced efficiency. Even when the starting pesticide concentration was increased, the material still removed most of it, though very high concentrations slowed the process as active sites on the particles became crowded.
Uncovering how the particles do the job
To understand the clean-up chemistry, the researchers added “scavenger” chemicals that selectively block certain reactive species. When they blocked hydroxyl radicals or superoxide species, the degradation of methyl parathion dropped sharply, revealing that these highly reactive forms of oxygen do most of the destructive work. Measurements of light emission and electrical resistance showed that the three-part material keeps electrons and holes apart longer than any of its single components, allowing more of these radicals to form. The authors propose a so-called Z-scheme pathway: under light, electrons and holes move along an energy “zigzag” between g-C3N5, CuS, and silver, recombining in a controlled way that leaves very strong oxidizing holes on g-C3N5 and strong reducing electrons on CuS. These then generate the radicals that attack and break the pesticide molecules into smaller, less harmful products.
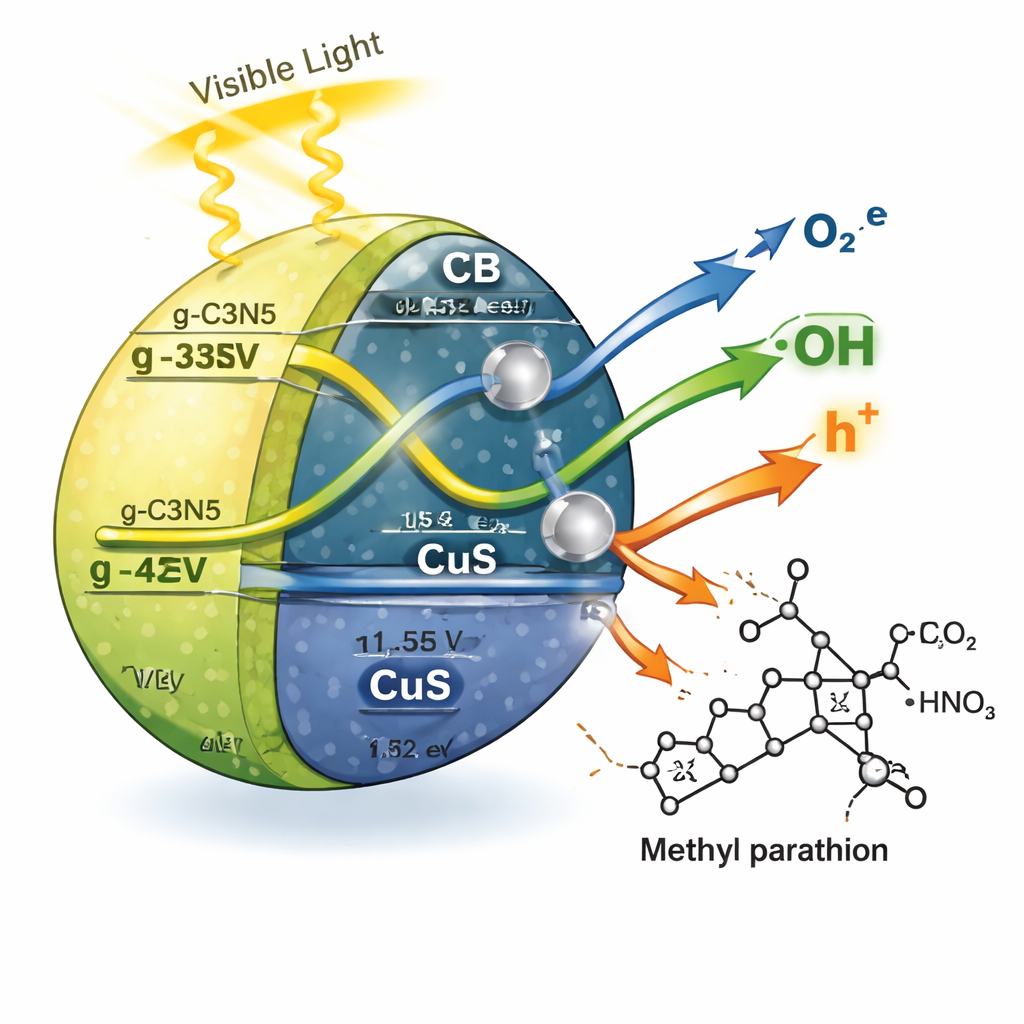
What this could mean for cleaner water
From a lay perspective, this study shows that it is possible to design tiny, robust particles that act like solar-powered cleaners for stubborn pesticides. The new g-C3N5/CuS/Ag nanocomposite removed nearly all methyl parathion from water in an hour under visible light and kept working well over several reuse cycles, suggesting it could be a practical tool for treating agricultural wastewater. Because it is relatively easy and low-cost to make, and because it uses light rather than large amounts of added chemicals, the approach could help safeguard drinking water and ecosystems in farming regions if it can be scaled up and integrated into treatment systems.
Citation: Teymourinia, H., Alshamsi, H.A., Gharagozlou, M. et al. Synthesis and characterization of g-C3N5/CuS/AgNPs nanocomposite as a Z-scheme photocatalyst for efficient methyl parathion degradation. Sci Rep 16, 6619 (2026). https://doi.org/10.1038/s41598-026-35254-6
Keywords: methyl parathion, photocatalyst, nanocomposite, water purification, pesticide degradation